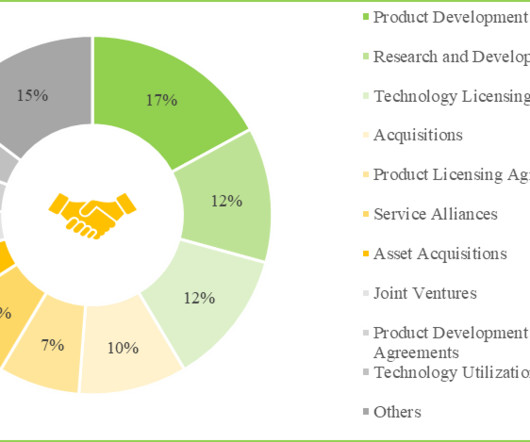
AvantGen Announces Licensing of Its Anti-SARS-CoV-2 Antibodies to IGM Biosciences for COVID-19 Therapy Development
The Pharma Data
JANUARY 11, 2021
a San Diego-based biotechnology company with an array of technology platforms for antibody discovery and optimization, and novel NK and T cell engager generation, today announced licensing of a panel of its anti-SARS-CoV-2 antibody clones to IGM Biosciences for COVID-19 therapy development.





























Let's personalize your content