Lifera, Sanofi and Arabio boost vaccine manufacture in Saudi Arabia
Pharmaceutical Technology
JULY 6, 2023
Lifera, Arabio and Sanofi have signed a memorandum of understanding (MOU) to bolster manufacturing of vaccines in Saudi Arabia.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Pharmaceutical Technology
JULY 6, 2023
Lifera, Arabio and Sanofi have signed a memorandum of understanding (MOU) to bolster manufacturing of vaccines in Saudi Arabia.

Pharmaceutical Technology
MARCH 8, 2024
SK bioscience has broken ground on a significant expansion of its vaccine manufacturing facility, L HOUSE, in South Korea.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
DECEMBER 19, 2023
BioNTech has inaugurated a new messenger RNA (mRNA) vaccine manufacturing facility in Kigali, Rwanda, which will have BioNTainers.

Pharmaceutical Technology
DECEMBER 15, 2023
Biovac has received an investment and advisory package from the IFC to support vaccine manufacturing and pandemic preparedness in Africa.

BioSpace
SEPTEMBER 26, 2023
The COVID-19 pandemic highlighted a need for local production of vaccines. Now, German pharma company BioNTech has said it will start manufacturing vaccines in Africa.
Bio Pharma Dive
APRIL 1, 2021
One vaccine batch being manufactured for J&J by Emergent Biosolutions failed quality checks, reportedly ruining material sufficient for up to 15 million doses.
Bio Pharma Dive
APRIL 26, 2021
The French pharma has agreed to "fill and finish" up to 200 million vials of Moderna's shot at its New Jersey plant, the third such manufacturing deal it struck with other vaccine developers.
BioPharma Reporter
NOVEMBER 7, 2023
The Australian biotech GPN Vaccines has expanded a partnership with compatriot contract development and manufacturing organization (CDMO) BioCina to scale up the manufacture of its lead candidate vaccine against pneumococcal diseases such as pneumonia, meningitis and febrile bacteraemia.

Pharmaceutical Technology
NOVEMBER 17, 2023
in funding from the Bill & Melinda Gates Foundation for the bulk manufacturing of needle-free vaccines. Micron Biomedical has received $23.6m

Bio Pharma Dive
JANUARY 29, 2021
A deal to fill vials of Pfizer and BioNTech's vaccine could be the first of several Novartis signs to help others make COVID-19 treatments.

Pharma Mirror
SEPTEMBER 22, 2021
Nantes, France, Naobios, a CDMO (Contract Development and Manufacturing Organization) providing bioprocess development and GMP production of clinical batches of viral vaccines BSL2/BSL3, oncolytic viruses and viral vectors, today announces the next phase in its partnership with FluGen, Inc., The company’s.

Pharmaceutical Technology
NOVEMBER 14, 2022
BioNTech’s Singapore affiliate BioNTech Pharmaceuticals Asia Pacific has signed an agreement with Novartis Singapore Pharmaceutical Manufacturing to acquire a GMP-certified manufacturing site in the country. . It also has the potential for extension into the manufacturing of other drug classes such as cell therapies. .

XTalks
SEPTEMBER 1, 2023
GlaxoSmithKline (GSK) has been pumping large sums of cash into expanding its vaccine manufacturing operations and today, it announced an investment of €250 ($272 million) into building a new unit for freeze-drying vaccines at its Wavre campus in Belgium. Both vaccines will be manufactured at the new unit in Belgium.

Pharmaceutical Technology
DECEMBER 4, 2023
GC Biopharma has concluded the construction activities at an mRNA manufacturing plant at its vaccine production site in South Korea.

Pharmaceutical Technology
JANUARY 20, 2023
In 2020, Moderna made a net loss of $747 million while its investigational mRNA vaccines were under development. However, during the Covid-19 pandemic, the US-based biotech rose to prominence as it was one of the first companies to develop Covid-19 vaccines with its mRNA technology. billion in Covid-19 vaccine sales.
Pharmaceutical Technology
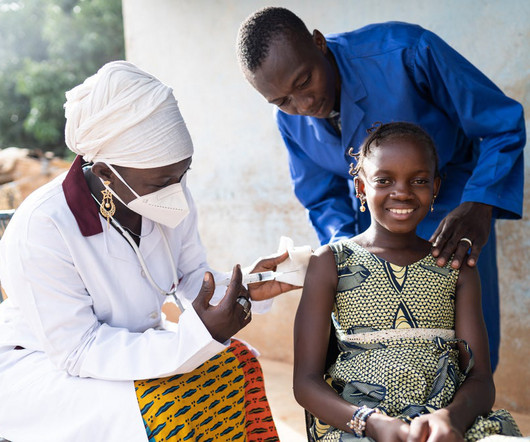
APRIL 13, 2023
The expanded licence will include the development of vaccines and therapeutic proteins beyond Covid-19 for human and animal health markets in Africa. During the Covid-19 pandemic, vaccination rates of many countries in Africa were significantly trailing the rest of the world.

Pharma Mirror
MAY 31, 2023
In the face of increasing global health challenges, the development and production of vaccines have become paramount. One critical aspect is the ability to scale up manufacturing processes to meet the growing demand for viral vector-based vaccines.

BioPharma Reporter
APRIL 7, 2022
US company Catalent is acquiring the UKâs Vaccine Manufacturing and Innovation Centre in Harwell: a facility which had been planned to support COVID-19 and wider vaccine production and which is still under construction.

Pharmaceutical Technology
APRIL 4, 2024
The vaccines will be developed using Touchlight’s doggybone DNA technology, which speeds up the manufacturing process.

Pharmaceutical Technology
APRIL 14, 2023
Ghana’s Food and Drug Authority (FDA) has approved R21/Matrix-M malaria vaccine in children aged 5 to 36 months, marking the first regulatory clearance for the University of Oxford-developed vaccine in any country in the world. Children between the ages of five and 36 months are at highest risk of death from malaria.

Pharmaceutical Technology
MARCH 31, 2023
Moderna has finalised an agreement with the government of the Republic of Kenya to establish an mRNA manufacturing facility in the country. The company is also committed to establishing mRNA manufacturing facilities in Australia, Canada, the US and the UK.

Pharmaceutical Technology
AUGUST 3, 2023
AAHI is developing a novel RNA vaccine that overcomes manufacturing and storage limitations to increase accessibility.

BioPharma Reporter
JANUARY 20, 2022
A new vaccine manufacturing plant has been opened in Cape Town by South Africa president Cyril Ramaphosa this week.
Pharmaceutical Technology
JUNE 6, 2023
Oragenics has secured funding from the Canadian bioresearch consortium CQDM to develop a variant-agnostic protein antigen for use in its Covid-19 intranasal vaccine. In March 2023, Oragenics and Inspirevax entered an exclusive global licence deal to develop the former’s lead intranasal Covid-19 vaccine candidate, NT-CoV2-1.

pharmaphorum
AUGUST 10, 2023
Manufacturing gene therapies, oncolytic viruses, and vaccine candidates: In conversation with ABL Mike.Hammerton Thu, 10/08/2023 - 08:00 Bookmark this

Fierce Pharma
MARCH 1, 2024
Moderna is laying off employees within its manufacturing unit, with the move tied to a resizing of its COVID production work. | Moderna is laying off some employees within its manufacturing unit after shaving COVID production costs. The company previously disclosed plans to rightsize its COVID vaccine production footprint.
Bio Pharma Dive
AUGUST 26, 2020
Catalent, which is already helping the British pharma make its vaccine, will now add key services from a Maryland gene therapy plant.
Pharmaceutical Technology
FEBRUARY 23, 2023
GenScript ProBio has announced a strategic collaboration with RVAC Medicines to manufacture GMP-grade plasmid DNA (pDNA) for the latter’s RVM-V001, an mRNA Covid-19 vaccine candidate. Under the agreement, GenScript ProBio will offer GMP plasmid manufacturing service for the RVM-V001 programme.
Bio Pharma Dive
SEPTEMBER 15, 2020
The Serum Institute of India, a go-to partner for coronavirus vaccine makers, will help Novavax boost capacity to levels matching its larger rivals.

Pharmaceutical Technology
NOVEMBER 2, 2023
While things may have slowed down a little since the rapid approvals of the COVID-19 vaccines, the excitement around RNA-LNP is still going strong.
BioPharma Reporter
DECEMBER 14, 2021
Moderna and the Australian Government have announced an agreement in principle to build a mRNA vaccine manufacturing facility in Victoria, Australia: providing up to 100 million doses a year.
Pharmaceutical Technology
JULY 29, 2022
Pfizer (New York, New York) and Moderna’s (Cambridge, Massachusetts) Omicron-specific Covid-19 vaccines are in late-stage development, have demonstrated good efficacy against the variant and will likely be available to the public later this year to provide an additional booster and increase demand for injectable manufacturing.

Pharmaceutical Technology
AUGUST 2, 2022
Samsung Biologics and GreenLight Biosciences have completed the initial commercial-scale engineering run for their messenger ribonucleic acid (mRNA) Covid-19 vaccine under their manufacturing collaboration. This will enable GreenLight to commercially deliver the mRNA vaccine.
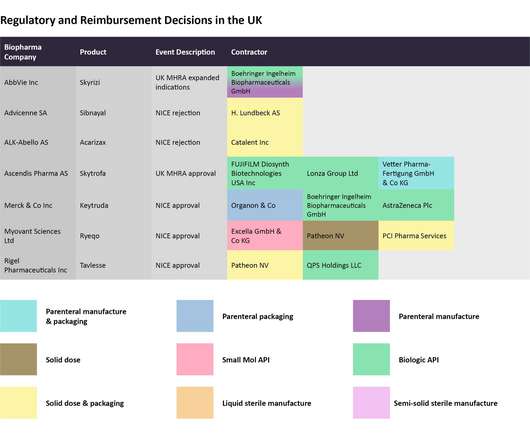
Pharmaceutical Technology
DECEMBER 12, 2022
In this last 2022 edition of the series, which started in June , Pharmaceutical Technology is tracking major trial announcements and decisions by regulators and reimbursement agencies that have occurred since mid-October, as well as their potential impact on manufacturing plans. Iomab-B met the durable complete remission endpoint.

BioSpace
NOVEMBER 24, 2020
Manufacturing capacity is often decried as a major bottleneck in administering COVID-19 vaccines to a global population of 7.8 For mRNA vaccines, the bottleneck is the availability of raw materials, not bioreactors.
BioPharma Reporter
AUGUST 21, 2022
Bavarian Nordic has entered into an agreement with Grand River Aseptic Manufacturing (GRAM), a US based contract manufacturer, for fill and finish of the Jynneos smallpox/monkeypox vaccine in the country.

Pharmaceutical Technology
JULY 14, 2022
Regulatory decisions have a ripple effect, first affecting the pharma sponsors, and then the companies tasked to manufacture the drug. Pharmaceutical Technology looks at drugs and biologics with recent regulatory verdicts that will likely impact manufacturing volumes. Covid-19 vaccines stay in the spotlight.
Bio Pharma Dive
JANUARY 26, 2021
The French drugmaker, having hit delays with its own coronavirus shot, agreed to manufacture over 100 million doses of Pfizer and BioNTech's vaccine for supply in Europe.
Pharmaceutical Technology
NOVEMBER 21, 2022
RVAC Medicines has signed a master research partnership agreement with the Agency for Science, Technology and Research (A*STAR) for analysing and developing solutions to build messenger ribonucleic acid (mRNA) manufacturing and analytics expertise in Singapore. Topic sponsors are not involved in the creation of editorial content.

XTalks
MAY 27, 2021
To help boost production of its COVID-19 vaccine, Moderna has enlisted the services of Samsung Biologics , a contract development and manufacturing organization (CDMO) based in South Korea. The vaccine doses are intended for countries outside the US, with manufacturing planned to commence in the third quarter of 2021.

Fierce Pharma
NOVEMBER 2, 2023
The days of booming COVID-19 vaccine sales, which sent Moderna into the ranks of pharma's top companies, have | The company is focusing on scaling back its manufacturing efforts to cope with the lack of COVID-19 vaccine demand.

Pharmaceutical Technology
NOVEMBER 24, 2022
South African bio-pharmaceutical firm Biovac and the International Vaccine Institute (IVI) in South Korea have signed a licencing and technology transfer agreement for developing and manufacturing an oral cholera vaccine (OCV).

XTalks
NOVEMBER 23, 2021
Canadian pharma and biotech contract development and manufacturing organization (CDMO) Biovectra has announced plans for building one of the first state-of-the-art mRNA vaccine and biomanufacturing facilities in Canada. Related: Medicago’s Plant-Based COVID-19 Vaccine Enters Human Trials. million, respectively.
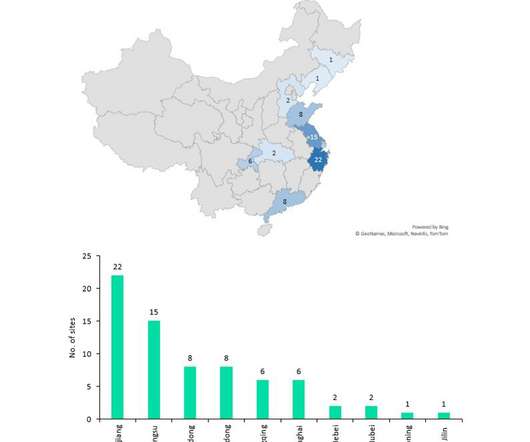
Pharmaceutical Technology
NOVEMBER 22, 2022
Despite China producing a significant proportion of the world’s API supply (mostly small molecule), it manufactures relatively few biosimilar and innovator drugs and no cell and gene therapies for the western markets of Europe and the US despite investments and an increasing number of startups to improve innovative manufacture.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content