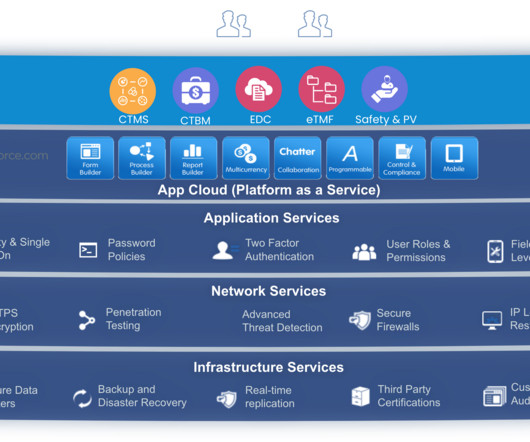
The FDA’s Technical Rejection Criteria for Study Data: Does Your eCTD Submission Comply?
Camargo
AUGUST 22, 2021
Effective September 15, 2021, the FDA will begin enforcing the TRC for study data. The correct STF file-tags are used for datasets and corresponding data definition files. How Are the Study Data Standards Enforced? What Are the Relevant Technical Rejection Criteria Documents? Validation Code. Criteria Being Verified. Code 1734.











Let's personalize your content