Food Allergy Awareness Week 2024: How can new treatments target an unmet need?
Pharmaceutical Technology
MAY 16, 2024
IgGenix is planning to initiate a clinical trial later this year investigating its peanut allergy monoclonal antibody IGXN001.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
MAY 16, 2024
IgGenix is planning to initiate a clinical trial later this year investigating its peanut allergy monoclonal antibody IGXN001.
STAT News
JANUARY 18, 2023
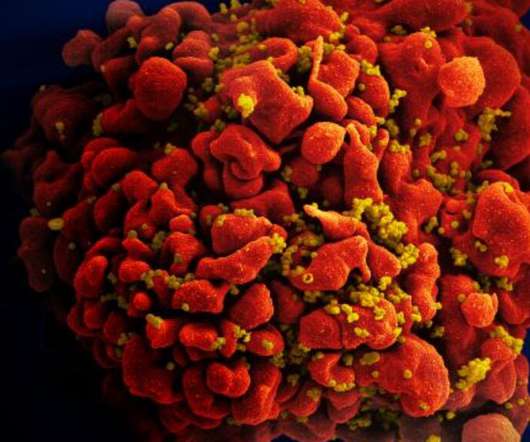
The National Institute of Allergy and Infectious Diseases reported Wednesday that a Phase 3 clinical trial of a vaccine was stopped because the vaccine was ineffective at preventing HIV infection. Yet another experimental HIV vaccine has failed. Read the rest…
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Deltaclinical
JANUARY 13, 2023
Home visits for clinical trials are becoming increasingly popular, as they offer a number of distinct benefits for both the patient and the provider. Improved patient retention in the study: When patients are more comfortable and less burdened by the study, they are more likely to stay enrolled in the trial.

Deltaclinical
JANUARY 13, 2023
Home visits for clinical trials are becoming increasingly popular, as they offer a number of distinct benefits for both the patient and the provider. Improved patient retention in the study: When patients are more comfortable and less burdened by the study, they are more likely to stay enrolled in the trial.

Deltaclinical
JANUARY 13, 2023
Home visits for clinical trials are becoming increasingly popular, as they offer a number of distinct benefits for both the patient and the provider. Improved patient retention in the study: When patients are more comfortable and less burdened by the study, they are more likely to stay enrolled in the trial.

Deltaclinical
JANUARY 13, 2023
Home visits for clinical trials are becoming increasingly popular, as they offer a number of distinct benefits for both the patient and the provider. Improved patient retention in the study: When patients are more comfortable and less burdened by the study, they are more likely to stay enrolled in the trial.
AuroBlog - Aurous Healthcare Clinical Trials blog
APRIL 13, 2023
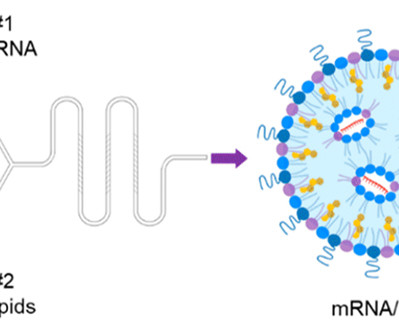
More treatments – and faster-acting treatments – for peanut allergies are urgently needed, and a new study outlines a promising drug technology based around a tiny nanoparticle just a few billionths of a meter in size. In tests on mice, the nanoparticle reversed peanut allergies and prevented them from developing in the first place.

Pharma Mirror
DECEMBER 1, 2021
Rockville, Emmes today announced that it conducted the data and statistical analysis for the third iteration of the Adaptive COVID-19 Treatment Trial (ACTT-3), using its proprietary Advantage eClinical system. The post Emmes Announces Completion of Third ACTT Clinical Trial for COVID-19 appeared first on Pharma Mirror Magazine.
Antidote
SEPTEMBER 1, 2023
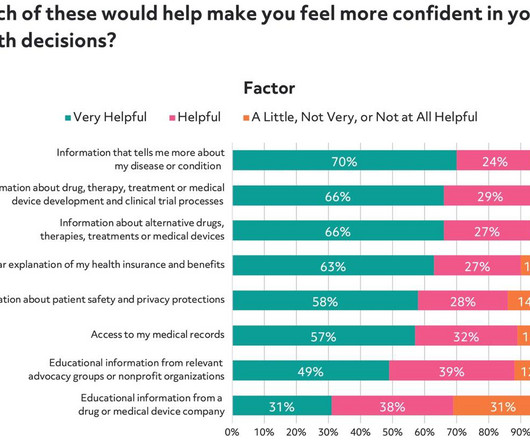
As part of our efforts to better understand patients and their motivations to participate in clinical trials, Antidote and SCORR Marketing partnered on a survey to gain a deeper understanding of how patients think about the medical research process.

BioTech 365
OCTOBER 29, 2021
Global Peanut Allergy Clinical Trials Review, H2 2021 Report – ResearchAndMarkets.com Global Peanut Allergy Clinical Trials Review, H2 2021 Report – ResearchAndMarkets.com DUBLIN–(BUSINESS WIRE)–The “Peanut Allergy – Global Clinical Trials Review, H2, 2021” clinical trials has been added to ResearchAndMarkets.com’s (..)

Antidote
NOVEMBER 20, 2023
In both 2018 and 2023, Antidote partnered with SCORR marketing to conduct surveys intended to increase the understanding of patients’ perceptions of clinical trials. Our 2018 survey collected data from nearly 4,000 individuals to gain insight into what matters most when considering a clinical trial.

Clinical Trial Podcast
SEPTEMBER 26, 2022
To answer these two questions, regulatory authorities around the world require medical product manufacturers to conduct Phase I, also known as early phase clinical trials. The post Complexity in Early Phase Clinical Trials with Dr. The post Complexity in Early Phase Clinical Trials with Dr. Oren Cohen?
Scienmag
NOVEMBER 18, 2021
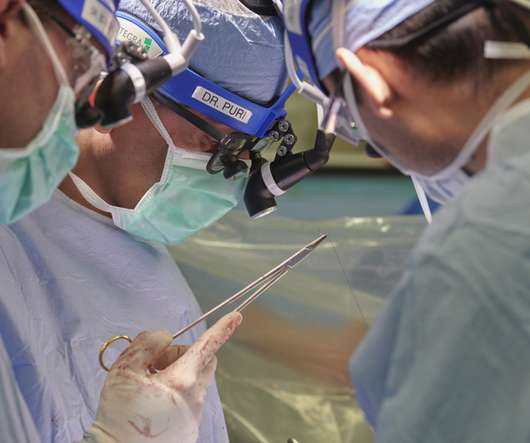
Louis and Massachusetts General Hospital/Harvard Medical School in Boston have received a seven-year, $22 million grant from the National Institute of Allergy and Infectious Diseases (NIAID) of the National Institutes of Health (NIH) to help lead a multicenter clinical trial evaluating whether a novel immunosuppressant can […].

JAMA Internal Medicine
JULY 16, 2023
This randomized clinical trial aims to determine whether a direct oral penicillin challenge is noninferior to the standard of care of penicillin skin testing followed by an oral challenge in patients with a low-risk penicillin allergy.

Rethinking Clinical Trials
NOVEMBER 29, 2022
The NIH Pragmatic Trials Collaboratory is excited to welcome the INSPIRE Demonstration Project to its portfolio of innovative, large-scale pragmatic clinical trials embedded in healthcare systems. Although fewer than 5% of such patients have an antibiotic-resistant infection, more than half receive extended-spectrum antibiotics.

Medical Xpress
FEBRUARY 28, 2023
A four-year clinical trial led by Edwin Kim, MD, at the UNC School of Medicine, has found that an increased dosage of a unique type of peanut allergy immunotherapy continues to show promise for children.
Drug Discovery World
MAY 18, 2023
A clinical trial of an experimental universal influenza vaccine developed by researchers at the National Institute of Allergy and Infectious Diseases’ (NIAID) Vaccine Research Center (VRC) has begun enrolling volunteers at Duke University in Durham, North Carolina. The trial will enrol up to 50 healthy volunteers aged 18 to 49.

Scienmag
JANUARY 20, 2022
A clinical trial funded by the National Institutes of Health has found that giving peanut oral immunotherapy to highly peanut-allergic children ages 1 to 3 years safely desensitized most of them to peanut and induced remission of peanut allergy in one-fifth. The immunotherapy consisted of a daily oral dose of peanut flour for 2.5

Drug Discovery World
JULY 19, 2022
An early-stage clinical trial has been launched in the US investigating a vaccine against Nipah virus infection. . The clinical trial has been launched by the National Institute of Allergy and Infectious Diseases (NIAID) to evaluate an investigational vaccine to prevent infection with Nipah virus.

Trialfacts
JANUARY 23, 2024
More Study Details About the Study A peanut allergy can be severe and life-threatening if not treated promptly. Peanut allergies are one of the most common food allergies in children in the USA, with more than 2% of children being affected. Participants will receive a new potential treatment for their peanut allergy at no cost.
NY Times
DECEMBER 11, 2020
British health officials recommended that people with severe allergy reactions not be given the vaccine. Such reactions to vaccines are rare, even in people who have allergies to food or bee stings.

Drug Discovery World
OCTOBER 13, 2022
New research has shown that standard cat allergy treatment can be enhanced to make it more effective and faster acting, and the benefits last for a year after treatment ends. The findings of the study, supported by the National Institutes of Health, were published in the Journal of Allergy and Clinical Immunology. .

XTalks
NOVEMBER 6, 2020
Canadian clinical-stage biotech company Symvivo Corporation has developed an oral COVID-19 vaccine that entered clinical trials this week. The first healthy volunteer was dosed with the vaccine in Australia as part of the bacTRL-Spike COVID-19 Phase I clinical trial. COVID-19 Clinical Trials.
Scienmag
APRIL 21, 2021
Credit: NIAID A Phase 2/3 trial to evaluate a new fully-human polyclonal antibody therapeutic targeted to SARS-CoV-2, called SAB-185, has begun enrolling non-hospitalized people with mild or moderate cases of COVID-19.

Medical Xpress
MARCH 6, 2023
A clinical trial testing a freeze-dried, temperature-stable experimental tuberculosis (TB) vaccine in healthy adults found that it was safe and stimulated both antibodies and responses from the cellular arm of the immune system. A non-temperature stable form of the candidate previously had been tested in several clinical trials.

Rethinking Clinical Trials
JANUARY 24, 2023
INSPIRE is studying the effectiveness of a personalized clinical decision support program in improving antibiotic prescribing for non–critically ill patients hospitalized with abdominal infections or skin and soft tissue infections.

Scienmag
MAY 25, 2021
May 25, 2021 — Critical Path Institute (C-Path) today announced its schedule for the Design of Clinical Trials in New-Onset Type 1 Diabetes: Regulatory Considerations for Drug Development Workshop, to be held virtually June 15-16. Together […].

Medical Xpress
JANUARY 17, 2023
A promising approach to control Staphylococcus aureus bacterial colonization in people—using a probiotic instead of antibiotics—was safe and highly effective in a Phase 2 clinical trial. aureus colonization in trial participants without harming the gut microbiota, which includes bacteria that can benefit people.

Rethinking Clinical Trials
JUNE 14, 2023
Updated study snapshots and ethics and regulatory documentation are now available for the IMPACt-LBP and INSPIRE trials, both NIH Pragmatic Trials Collaboratory Demonstration Projects. The project is supported within the NIH Collaboratory under an award from the National Institute of Allergy and Infectious Diseases.

The Pharma Data
MARCH 31, 2022
A Phase 2 clinical trial evaluating various additional COVID-19 booster shots has begun enrolling adult participants in the United States. “This trial will help us understand if we can use prototype and variant vaccines alone or together to shift immune responses to cover existing and emerging COVID-19 variants.”

Scienmag
MAY 21, 2021
The findings of a clinical trial by Trinity College Dublin researchers of treatment for atopic dermatitis have been published today in The Lancet journal (Friday, 21st May, 2021). Results of the clinical trial at […].
STAT News
SEPTEMBER 12, 2022
National Institutes of Health is launching a Phase 3 clinical trial to determine the efficacy of tecovirimat — the smallpox antiviral better known as TPOXX — for use in treating monkeypox, as current data on its effectiveness against the virus is limited , The Hill writes. The TPOXX was approved by the U.S.

Medical Xpress
DECEMBER 22, 2022
The study analyzed data from two phase 3 clinical trials and involved an international group of researchers, including those from Children's Hospital of Philadelphia (CHOP).
Drug Discovery World
SEPTEMBER 25, 2023
Enrolment in a Phase I trial of a new investigational universal influenza vaccine candidate has begun at the National Institutes of Health’s Clinical Center in Bethesda, Maryland, US. It is an adaptation of an earlier universal flu vaccine candidate, FluMos-v1, which began first-in-human testing in 2021 and is still undergoing trials.
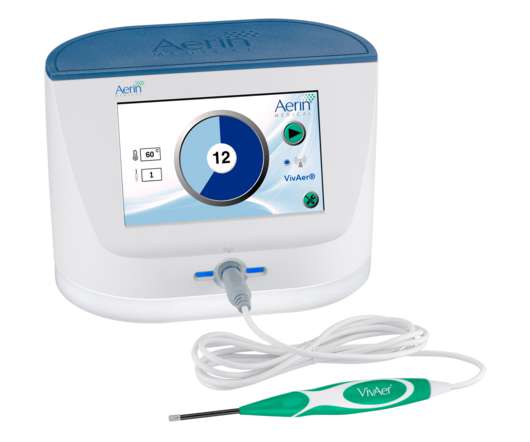
XTalks
JULY 6, 2022
Their 48-month clinical study has revealed that VivAer can substantially ease the symptoms of NVC and improve nasal breathing. The four-year results from the VivAer clinical trial were published in the International Forum of Allergy & Rhinology.
Medical Xpress
JANUARY 30, 2023
A newly published paper in The Lancet shows that an experimental vaccine against Marburg virus (MARV) was safe and induced an immune response in a small, first-in-human clinical trial.
XTalks
SEPTEMBER 21, 2023
announced that the first participant has been dosed in a new Phase I trial for an innovative HIV vaccine candidate, VIR-1388. The trial’s primary objective is to assess the safety of VIR-1388 and its capacity to stimulate a specific immune response against HIV in study participants. Vir Biotechnology, Inc.,
Drug Discovery World
NOVEMBER 29, 2023
At sites in the US, Japan, and the EU, the clinical trial will evaluate the safety, tolerability, and efficacy of leniolisib in 15 children, one to six years of age, who have a confirmed APDS diagnosis. The post Trials investigate targeted APDS treatment in a paediatric setting appeared first on Drug Discovery World (DDW).
The Pharma Data
APRIL 3, 2021
The CoVIg-19 Plasma Alliance today announced that the Phase 3 Inpatient Treatment with Anti-Coronavirus Immunoglobulin (ITAC) clinical trial sponsored and funded by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health (NIH), did not meet its endpoints. About the ITAC Trial.

Scienmag
SEPTEMBER 8, 2020
million USD to IVI to support clinical trial site preparedness in four African and Asian countries to potentially support future COVID-19 Phase III efficacy vaccine trials. Following successful completion […].

The Pharma Data
JANUARY 7, 2021
This clearance is a pivotal regulatory milestone that further supports Camallergy’s leadership and capabilities in the field of peanut allergy immunotherapy with the only cGMP-certified manufacturing facility in Europe producing peanut oral immunotherapy,” said Sherden Timmins, Chief Technology Officer. 8, 2021 07:00 UTC. About Camallergy.

The Pharma Data
NOVEMBER 16, 2020
November 16, 2020 — An independent data and safety monitoring board (DSMB) overseeing the Phase 3 trial of the investigational COVID-19 vaccine known as mRNA-1273 reviewed trial data and shared its interim analysis with the trial oversight group on Nov. 37% of trial volunteers are from racial and ethnic minorities.

Rethinking Clinical Trials
JUNE 6, 2023
The purpose of the trial is to reduce unnecessary broad-spectrum antibiotic use in non-intensive care unit inpatients. The 12-month trial is currently in month 5, and the team has already seen a downward inflection in broad-spectrum antibiotic use.

The Pharma Data
AUGUST 7, 2020
The governing body claims this proposal could have positive effects on mitigating burdensome requirements for gene therapy clinical trials in the future. In this light, Pharma IQ’s weekly round-up focuses on advancing therapies and clinical trials to combat Covid-19.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content