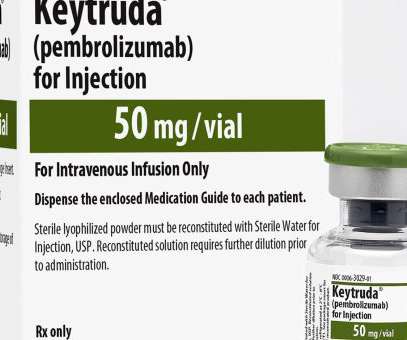
FDA Approves Expanded Indication for Merck’s KEYTRUDA® (pembrolizumab) in Locally Advanced Cutaneous Squamous Cell Carcinoma (cSCC)
The Pharma Data
JULY 8, 2021
KEYTRUDA is a humanized monoclonal antibody that blocks the interaction between PD-1 and its ligands, PD-L1 and PD-L2, thereby activating T lymphocytes which may affect both tumor cells and healthy cells. 1%) as determined by an FDA-approved test, with disease progression on or after platinum-containing chemotherapy.





















Let's personalize your content