Three trends in the antibody-drug conjugate (ADC) market
Drug Discovery World
JUNE 1, 2023
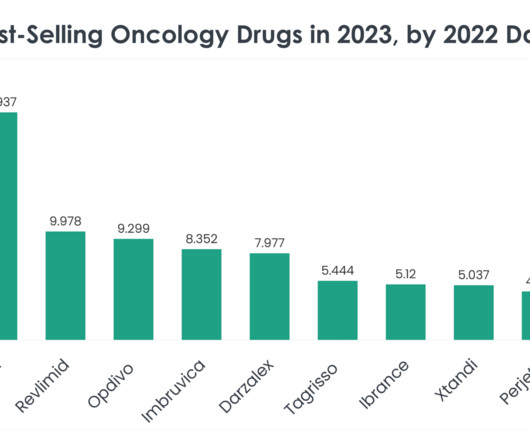
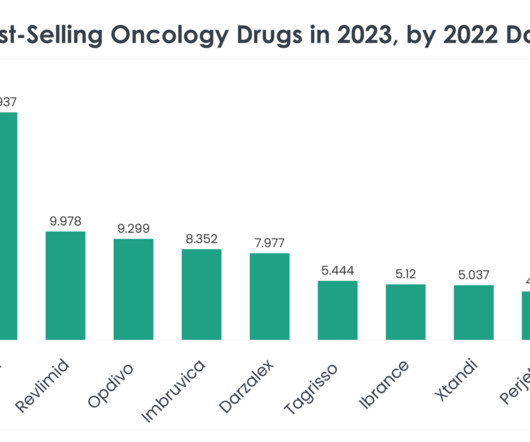
Antibody-drug conjugates (ADCs) are biopharmaceutical products in which a monoclonal antibody (mAB) is linked to a small molecule drug with a stable linker 1. A Nature publication confirmed that there are currently 12 FDA-approved ADCs on the market, and nine of these secured FDA approval in the past six years 2.



























Let's personalize your content