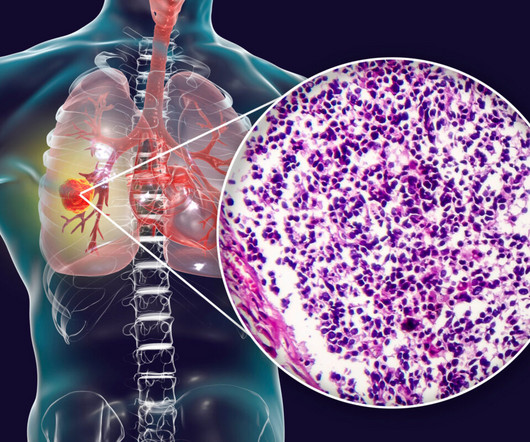
Amgen’s Tarlatamab Gets FDA Priority Review for Small Cell Lung Cancer
XTalks
DECEMBER 29, 2023
. “The FDA’s Priority Review designation for this application underscores the urgency to provide new treatment options for patients with advanced SCLC who have progressed following treatment with platinum-based chemotherapy,” said David M.













































Let's personalize your content