Juvisé licenses J&J’s MS drug Ponvory for marketing outside North America
Pharmaceutical Technology
MARCH 27, 2024
The market for multiple sclerosis therapies is expected to be worth over $41bn by 2030, according to GlobalData.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

AuroBlog - Aurous Healthcare Clinical Trials blog
MARCH 20, 2023
Empowering of the Central Drugs Standard Control Organisation (CDSCO) through centralisation of drug licensing may not be the path to improve the quality of drugs manufactured in the country both for domestic and international markets, says SME Pharma Industries Confederation (SPIC), the apex organisation of small and medium pharma industries in the (..)
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharmaceutical Technology
JANUARY 23, 2023
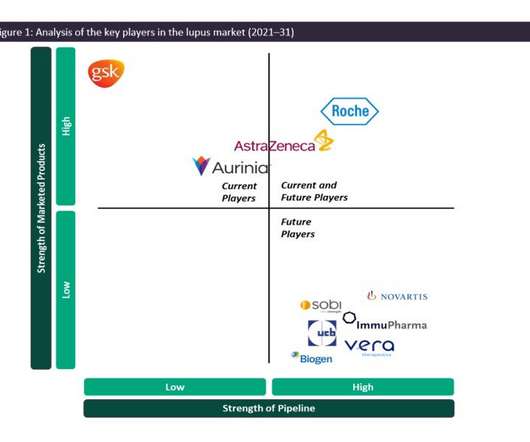
As such, the SLE and LN marketplace is dominated by generics, and GSK’s Benlysta and AstraZeneca’s Saphnelo are the only drugs that have gained marketing approval specifically for SLE in more than 50 years. In previous years, Benlysta has managed to grow the lupus market in terms of value, having generated approximately $492.9m

Bio Pharma Dive
AUGUST 1, 2023
Some on Wall Street believe the New York-based biotechnology company is now less likely to get acquired after agreeing to sell partial rights to its only marketed product.
Pharmaceutical Technology
JUNE 19, 2023
Bio-Thera Solutions and Biomm have entered a licensing and supply agreement for Bio-Thera’s BAT2206, a ustekinumab biosimilar. Biomm will gain exclusive rights for the distribution and marketing in Brazil of the proposed biosimilar to Janssen’s Stelara.
Pharmaceutical Technology
JANUARY 23, 2023
Takeda has signed an exclusive licence agreement with HUTCHMED (China) and its subsidiary HUTCHMED to develop and market the latter’s fruquintinib. The China National Medical Products Administration (NMPA) approved fruquintinib for marketing in September 2018. The submission is planned to be completed in the first half of this year.

BioPharma Reporter
APRIL 2, 2024
Ipsen is expanding its oncology pipeline by securing the global licensing rights to an antibody-drug conjugate (ADC) for solid tumors from Sutro Biopharma, its first drug in the category.

Drug Discovery World
JUNE 1, 2023
Antibody-drug conjugates (ADCs) are biopharmaceutical products in which a monoclonal antibody (mAB) is linked to a small molecule drug with a stable linker 1. A Nature publication confirmed that there are currently 12 FDA-approved ADCs on the market, and nine of these secured FDA approval in the past six years 2.
Pharmaceutical Technology
JULY 28, 2022
Melinta Therapeutics has signed a licence agreement with Cidara Therapeutics to facilitate a strategic collaboration for marketing the latter’s rezafungin in the US. The collaboration comes after Cidara filed a New Drug Application (NDA) for rezafungin to treat candidemia and invasive candidiasis.
Bio Pharma Dive
DECEMBER 14, 2021
Fitusiran, which Sanofi licensed from Alnylam, could finally get to market after trial delays and safety concerns slowed its progress. But gene therapies and other new medicines might provide competition.

BioPharma Reporter
OCTOBER 31, 2023
The French biotech company Vect-Horus has licensed its technology to the Danish big pharma Novo Nordisk to deliver drug cargoes to undisclosed disease targets in three programs.
Drug Discovery World
AUGUST 2, 2023
Renaissance Pharma has announced its first development programme focused on Hu14.18, a humanised anti-GD2 monoclonal antibody (mAb), licensed from St Jude Children’s Research Hospital for the treatment of newly diagnosed high-risk neuroblastoma. ” The post Renaissance Pharma licenses Hu14.18
Pharmaceutical Technology
OCTOBER 23, 2023
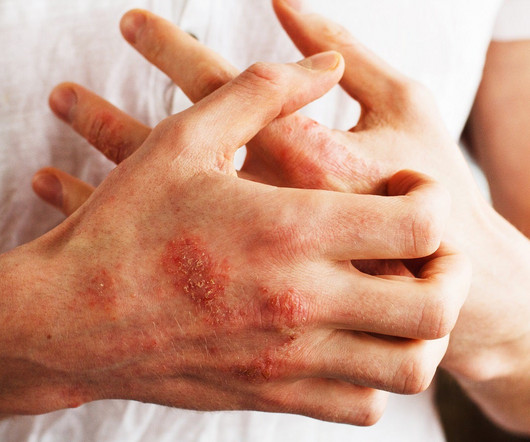
On 2 October, Eli Lilly announced that the US Food and Drug Administration (FDA) had issued a complete response letter (CRL) regarding the biologic license application (BLA) for its anti-interleukin (IL)-13 biologic lebrikizumab for the treatment of adults and adolescents (12 and older) with moderate-to-severe atopic dermatitis (AD).

Pharmaceutical Technology
MAY 22, 2023
Biogen and Eisai have submitted a marketing authorisation application (MAA) to the UK’s medicines and healthcare products regulatory agency (MHRA) for lecanemab to treat early Alzheimer’s disease (AD). The regulator has also designated the therapy for the innovative licensing and access pathway (ILAP).

Pharmaceutical Technology
APRIL 27, 2023
The European Commission’s (EC) long-anticipated pharma reform plans in the European Union have finally been unveiled , indicating a focus on improving access to medicines across the bloc while cutting down on market exclusivity. Outside of these steps, the reform targets drug shortages and pharma industry’s environmental impact.
Pharmaceutical Technology
MAY 3, 2023
Aurinia Pharmaceuticals has received marketing authorisation from the Swiss Agency for Therapeutic Products (Swissmedic) for Lupkynis (voclosporin), along with a background immunosuppressive therapy. Lupkynis has also received orphan drug designation in Switzerland. This promotes podocyte stability in the kidneys.

BioPharma Reporter
SEPTEMBER 25, 2023
MC2 Therapeutics has acquired the exclusive licensing rights for Regranionâs RGRN-305, a new treatment for hidradenitis suppurativa (HS).

Bio Pharma Dive
MARCH 13, 2023
The deal builds on a licensing agreement between Sanofi and Provention to market the first-of-its-kind treatment, called Tzield and approved to slow the disease’s progression.
Pharmaceutical Technology
MAY 24, 2023
Y-mAbs Therapeutics has received marketing authorisation for Danyelza (naxitamab-gqgk) 40mg/10mL injection from the Brazilian Health Regulatory Agency, Agência Nacional de Vigilância Sanitária, to treat high-risk neuroblastoma. The therapy is given three times a week and repeated every four weeks.

BioPharma Reporter
OCTOBER 26, 2023
The biopharmaceutical industry witnessed a 400% growth in total deal value of antibody-drug conjugate (ADC) licensing agreements from 2017-2022 and reached a peak of $16.6 billion in 2022, reveals data and analytics company GlobalData.

Fossil Remedies
OCTOBER 16, 2022
In a pharma business, you have a distribution network like a pharma manufacturing company, pharma marketing company, C&F agents, wholesalers/distributors/stockiest, retailers, and pharmacies. To launch the best PCD pharma franchise company, you will have to acquire the necessary licenses from authorities. Wholesale Drug License.

pharmaphorum
JANUARY 9, 2023
Swiss biotech Stalicla has continued to diversify its pipeline from a focus on autism therapies with a deal to license Novartis’ mavoglurant, in clinical trials for cocaine use disorder (CUD) as well as neurodevelopmental disorders (NDD). The market potential of the CUD and NDD indications alone could top 2 billion euros ($2.12

Bio Pharma Dive
APRIL 13, 2022
Antares Pharma's auto-injector technology is of particular interest to Halozyme, which believes that the various markets to which it could be applied represent billions of dollars in potential sales.
Pharmaceutical Technology
JUNE 5, 2023
Pint-Pharma will obtain and maintain all marketing authorisations and will commercialise BESREMi in the region. It holds orphan drug designation in the US to treat PV. In 2019, the European Medicines Agency approved the therapy, which has also been licensed in Japan, Taiwan and South Korea.
Pharmaceutical Technology
DECEMBER 26, 2022
LegoChem Biosciences and Amgen have signed a multi-target research collaboration and license agreement to develop antibody-drug conjugates (ADC). Till date, the company has signed a total of 12 ADC licensing deals, worth more than $5bn.

BioTech 365
JUNE 9, 2021
World Pancreatic Cancer Market Spotlight 2021-2031: Key Marketed and Pipeline Drugs, Events, Clinical Trials, Regulations, Disease Prevalence, Licensing and Acquisition, Revenue – ResearchAndMarkets.com World Pancreatic Cancer Market Spotlight 2021-2031: Key Marketed and Pipeline Drugs, Events, Clinical Trials, Regulations, Disease Prevalence, (..)

BioTech 365
JUNE 9, 2021
Global Osteoarthritis and Osteoarthritis Pain Market 2021-2031: Key Marketed and Pipeline Drugs, Events, Clinical Trials, Regulations, Disease Prevalence, Licensing and Acquisition, Revenue – ResearchAndMarkets.com Global Osteoarthritis and Osteoarthritis Pain Market 2021-2031: Key Marketed and Pipeline Drugs, Events, Clinical Trials, Regulations, (..)

Pharmaceutical Technology
MAY 10, 2023
Gilead Sciences has emerged victorious in a legal battle with the US government over patents surrounding the HIV pre-exposure prophylaxis (PrEP) drugs Descovy and Truvada following a federal jury’s verdict on May 9. Gilead markets both Truvada and its successor Descovy.

BioTech 365
JANUARY 27, 2022
Attention Deficit Hyperactivity Disorder (ADHD) Market Spotlight Report 2021: 10-year Disease Prevalence Forecast, and Licensing and Acquisition Deals, Drug-specific Revenue Forecasts – ResearchAndMarkets.com Attention Deficit Hyperactivity Disorder (ADHD) Market Spotlight Report 2021: 10-year Disease Prevalence Forecast, and Licensing and Acquisition (..)

BioTech 365
JANUARY 27, 2022
Systemic Hypertension Market Spotlight Report 2021: 10-Year Disease Prevalence Forecast, Clinical Trials, Regulatory Events, Licensing and Acquisitions, and Drug-specific Revenue Forecasts – ResearchAndMarkets.com Systemic Hypertension Market Spotlight Report 2021: 10-Year Disease Prevalence Forecast, Clinical Trials, Regulatory Events, Licensing (..)

BioTech 365
JANUARY 27, 2022
Glioblastoma (GBM) Market Spotlight Report 2021: 10-Year Disease Prevalence Forecast, Clinical Trials, Regulatory Events, Licensing and Acquisitions, and Drug-specific Revenue Forecasts – ResearchAndMarkets.com Glioblastoma (GBM) Market Spotlight Report 2021: 10-Year Disease Prevalence Forecast, Clinical Trials, Regulatory Events, Licensing and (..)

pharmaphorum
NOVEMBER 23, 2021
Sanofi has bolstered its push into mRNA-based therapies with a new licensing deal – but not as might be expected with some up-and-coming biotech company. Sanofi is the first pharma company to partner with Baidu on its LinearDesign AI platform, which has been developed specifically to optimise the sequence of mRNA-based vaccines and drugs.

BioTech 365
JANUARY 27, 2022
Atrial Fibrillation (AF) Market Spotlight Report 2021: 10-Year Disease Prevalence Forecast, Clinical Trials, Regulatory Events, Licensing and Acquisitions, and Drug-Specific Revenue Forecasts – ResearchAndMarkets.com Atrial Fibrillation (AF) Market Spotlight Report 2021: 10-Year Disease Prevalence Forecast, Clinical Trials, Regulatory Events, (..)

BioPharma Reporter
JANUARY 6, 2023
Synaffix, a Netherlands based company providing clinical-stage platform technology for the development of antibody-drug conjugates (ADCs), has signed off on two new licensing deals this week.

BioTech 365
JANUARY 27, 2022
Systemic Lupus Erythematosus (SLE)Market Spotlight Report 2021: 10-Year Disease Prevalence Forecast, Clinical Trials, Regulatory Events, Licensing and Acquisitions, and Drug-specific Revenue Forecasts – ResearchAndMarkets.com Systemic Lupus Erythematosus (SLE)Market Spotlight Report 2021: 10-Year Disease Prevalence Forecast, Clinical Trials, (..)

pharmaphorum
DECEMBER 7, 2021
AstraZeneca has snapped up another Ionis-discovered drug for its pipeline, this time phase 3 candidate eplontersen for the disease transthyretin amyloidosis (ATTR). billion in milestones depending on sales levels if the antisense-based drug reaches the market.

Pharmaceutical Technology
FEBRUARY 14, 2023
Precision oncology firm Corbus Pharmaceuticals and CSPC Megalith Biopharmaceutical have entered an exclusive licensing agreement for the latter’s new clinical stage antibody drug conjugate (ADC), CRB-701 (SYS6002). The US Food and Drug Administration (FDA) has cleared the investigational new drug (IND) application for CRB-701.

BioPharma Reporter
DECEMBER 6, 2021
Australiaâs Noxopharm has in-licensed novel RNA tech developed by Hudson Institute of Medical Research: focusing on RNA drug discovery and mRNA vaccine manufacture via Noxopharmâs subsidiary Pharmorage.
Roots Analysis
FEBRUARY 26, 2024
Biologics constitute a majority of the top selling drugs and represent one of the fastest growing segments of the overall pharmaceutical industry. In fact, the share of biologics in the overall pharmaceutical contract manufacturing market has increased from 16% in 2006 to over 25% in 2017.

BioTech 365
JANUARY 27, 2022
Hereditary Angioedema Market Spotlight Report 2021: 10-year Disease Incidence and Prevalence Forecasts, Probability of Success, Licensing and Asset Acquisition Deals, Drug-specific Revenue Forecasts – ResearchAndMarkets.com Hereditary Angioedema Market Spotlight Report 2021: 10-year Disease Incidence and Prevalence Forecasts, Probability of Success, (..)
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?



Let's personalize your content