Health Canada approves Jazz’s cannabis derived seizure therapy
Pharmaceutical Technology
NOVEMBER 24, 2023
Epidiolex has been approved as an adjunct therapy for seizures associated with three rare forms of epilepsy in patients aged two and older.

Pharmaceutical Technology
NOVEMBER 24, 2023
Epidiolex has been approved as an adjunct therapy for seizures associated with three rare forms of epilepsy in patients aged two and older.
Drug Discovery World
NOVEMBER 24, 2023
The University of Queensland’s re-engineered clamp platform has produced a vaccine that is equally safe and virus-neutralising as an approved Covid vaccine considered among the best in its class. The pivotal proof-of-concept testing clears the way to progress the Clamp2 technology in a range of research programmes. Project leader, Associate Professor Keith Chappell, said the preliminary clinical trial results were an ‘exciting reward’ for the team’s continued dedication and the community’s wi
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
NOVEMBER 24, 2023
Bayer announced the launch of a Berlin, Germany-based manufacturing facility to meet future demands from the US, China and Europe.

Pharma Times
NOVEMBER 24, 2023
Around 3,200 new cases of cervical cancer are diagnosed in the UK every year - News - PharmaTimes
Pharmaceutical Technology
NOVEMBER 24, 2023
Vertex Pharmaceuticals has obtained approval from EC) for the label expansion of Kaftrio plus ivacaftor to treat children with CF.
Pharma Times
NOVEMBER 24, 2023
The findings could benefit patients living with neurodegenerative conditions - News - PharmaTimes
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

XTalks
NOVEMBER 24, 2023
In the world of hot beverages, Postum occupies a unique place. Originating as a coffee alternative, this drink has carved out a niche among those seeking a caffeine-free option. To understand its enduring appeal, let’s delve into its history, ingredients and its status in today’s beverage market. The Birth of Postum Postum was born out of necessity.
Pharmaceutical Technology
NOVEMBER 24, 2023
Access and buy GlobalData’s databook on the latest filings and grants activity for Johnson & Johnson here.
Outsourcing Pharma
NOVEMBER 24, 2023
Calliditas Therapeutics AB announced today (November 24) that its partner Everest Medicines has revealed that China's National Medical Products Administration (NMPA) has approved Nefecon.

Pharmaceutical Technology
NOVEMBER 24, 2023
Access and buy GlobalData’s databook on the latest filings and grants activity for F. Hoffmann-La Roche here.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

pharmaphorum
NOVEMBER 24, 2023
International Health Partners' Christmas campaign: Working with pharma for patients worldwide Nicole.
Pharmaceutical Technology
NOVEMBER 24, 2023
Access and buy GlobalData’s databook on the latest filings and grants activity for Bristol-Myers Squibb here.
Outsourcing Pharma
NOVEMBER 24, 2023
The Janssen Pharmaceutical Companies of Johnson & Johnson announced last week (November 23) the submission of a type 2 extension of indication application to the European Medicines Agency (EMA).

Pharmaceutical Technology
NOVEMBER 24, 2023
With more approved drugs coming through than ever before, the need to avoid confusing names to ensure patient safety is important.

pharmaphorum
NOVEMBER 24, 2023
How to optimise commercial-scale ATMP manufacturing through automation Mike.

Pharmaceutical Technology
NOVEMBER 24, 2023
The expansion comes as Novo is trying to maintain strong growth in T2D after the launch of Eli Lilly’s Zepbound this month.
Drug Patent Watch
NOVEMBER 24, 2023
Annual Drug Patent Expirations for RINVOQ Rinvoq is a drug marketed by Abbvie and is included in one NDA. It is available from one supplier. There are twenty-six patents protecting… The post New patent for Abbvie drug RINVOQ appeared first on DrugPatentWatch - Make Better Decisions.
Pharmaceutical Technology
NOVEMBER 24, 2023
Access and buy GlobalData’s databook on the latest filings and grants activity for Novartis here.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
Drug Patent Watch
NOVEMBER 24, 2023
Annual Drug Patent Expirations for XYOSTED+%28AUTOINJECTOR%29 Xyosted (autoinjector) is a drug marketed by Antares Pharma Inc and is included in one NDA. It is available from one supplier. There are… The post New patent for Antares Pharma drug XYOSTED (AUTOINJECTOR) appeared first on DrugPatentWatch - Make Better Decisions.

Pharmaceutical Technology
NOVEMBER 24, 2023
China’s NMPA has approved ArkBio IND application for its respiratory syncytial virus (RSV) neutralising antibody, AK0610.

Drug Discovery World
NOVEMBER 24, 2023
Patrick Speedie , Co-Founder & Co-CEO of IN-PART , explores why a global approach is needed for the best science to prosper and the lessons industry can take from the Covid-19 pandemic. The Covid-19 pandemic unequivocally demonstrated the power of industry-academia collaboration, with the fastest ever development, testing and approval of a life-saving vaccine.
Pharmaceutical Technology
NOVEMBER 24, 2023
Ellipses Pharma has secured ODD from the US FDA for its next generation EP0031/A400, to potentially treat solid tumours.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
Outsourcing Pharma
NOVEMBER 24, 2023
The Janssen Pharmaceutical Companies of Johnson & Johnson announced last week (November 23) the submission of a type 2 extension of indication application to the European Medicines Agency (EMA).
Pharmaceutical Technology
NOVEMBER 24, 2023
Access and buy GlobalData’s databook on the latest filings and grants activity for Amgen here.

Drug Discovery World
NOVEMBER 24, 2023
AstraZeneca has announced that the National Institute for Health and Care Excellence (NICE) has recommended Imfinzi (durvalumab) in combination with gemcitabine and cisplatin (chemotherapy) for NHS use in England and Wales for the first-line treatment of adults with locally advanced, unresectable, or metastatic biliary tract cancer (BTC). 1 There remains a significant unmet need for new treatment options in BTC, a group of rare and aggressive cancers that occur in the bile ducts and gallbladder,
Pharmaceutical Technology
NOVEMBER 24, 2023
Lupin has introduced the fixed-dose triple combination drug (FDC), Vilfuro-G, to manage chronic COPD in India.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.
pharmaphorum
NOVEMBER 24, 2023
NHS Confederation, ABPI say break down barriers to partnerships and change the lives of millions Nicole.
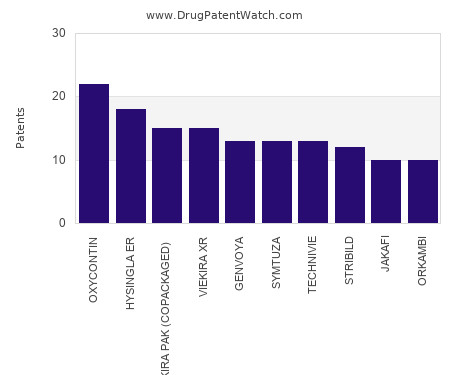
Drug Patent Watch
NOVEMBER 24, 2023
This chart shows the drugs with the most patents in Croatia. Patents must be filed in each country (or, in some cases regional patent office) where patent protection is desired.… The post Which pharmaceutical drugs have the most drug patents in Croatia? appeared first on DrugPatentWatch - Make Better Decisions.

pharmaphorum
NOVEMBER 24, 2023
Intellectual property: Striking the balance Mike.
Let's personalize your content