First gene therapies for sickle cell disease secure FDA approval
Pharmaceutical Technology
DECEMBER 11, 2023
The FDA has approved Vertex/CRISPR’s Casgevy and bluebird bio’s Lyfgenia gene therapies for the red blood cell disorder.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Pharmaceutical Technology
DECEMBER 11, 2023
The FDA has approved Vertex/CRISPR’s Casgevy and bluebird bio’s Lyfgenia gene therapies for the red blood cell disorder.

Bio Pharma Dive
MARCH 19, 2024
clearance of Lenmeldy, for a rare and inherited metabolic disease, triggers an additional payout related to Kyowa Kirin’s recent deal to acquire the once high-flying gene therapy developer.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
SEPTEMBER 30, 2022
After several setbacks, bluebird bio bounces back with two major FDA gene therapy approvals. Last month, Zynteglo (betibeglogene autotemcel), or beti-cel, was approved as a one-time potentially curative gene therapy for patients with beta-thalassaemia who require regular blood transfusions.

Pharmaceutical Technology
MAY 26, 2023
Krystal Biotech has received approval from the US Food and Drug Administration for topical gene therapy VYJUVEK to treat dystrophic epidermolysis bullosa (DEB) in adults and in children aged six months and above. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.
Pharmaceutical Technology
JUNE 30, 2023
BioMarin’s Roctavian, a one-time therapy for severe haemophilia A, has secured FDA approval following previous rejections and delays.

Pharmaceutical Technology
MARCH 19, 2024
The US FDA has granted approval to Orchard Therapeutics' Lenmeldy for specific forms of metachromatic leukodystrophy (MLD).

Bio Pharma Dive
MAY 22, 2023
Cleared to treat dystrophic epidermolysis bullosa, Vyjuvek is the sixth gene therapy for an inherited disease to gain clearance in the U.S.

Bio Pharma Dive
MAY 20, 2022
The brain-infused treatment for a rare neurological disease would be one of only three gene therapies commercially available in the EU. PTC plans to ask for FDA approval later this year.

pharmaphorum
MARCH 26, 2024
Nanoscope Therapeutics is on the brink of filing for FDA approval of what could be the first gene therapy for incurable eye disease retinitis pigmentosa (RP) that can be used regardless of underlying genetic mutations.
Fierce Pharma
DECEMBER 8, 2023
Vertex Pharmaceuticals and CRISPR Therapeutics have won an historic FDA approval for their sickle cell disease gene therapy Casgevy. Groundbreaking? Game-changing? Transformational?

BioSpace
APRIL 25, 2024
Pfizer will go toe-to-toe with CSL Behring following the FDA’s Friday approval of its hemophilia B gene therapy Beqvez and will launch a warranty program based on the durability of response.

Bio Pharma Dive
JUNE 29, 2023
After a prolonged journey, the medicine, known as Roctavian, is now cleared for certain patients with hemophilia A, the more common form of the rare bleeding disorder.

BioSpace
MARCH 18, 2024
Orchard Therapeutics on Monday secured the FDA’s first approval for an autologous gene therapy to treat the rare metabolic disease metachromatic leukodystrophy in children.

Bio Pharma Dive
DECEMBER 19, 2022
The OK caps a long development journey for the treatment, which was previously turned back by the agency. Ferring doesn’t expect it to be available until the second half of next year, however.
Bio Pharma Dive
NOVEMBER 22, 2022
The treatment, which is for the less common “B” form of the bleeding disorder, will be sold in the U.S. by maker CSL for $3.5

Fierce Pharma
MARCH 18, 2024
Kyowa Kirin’s bet on Orchard Therapeutics has paid off, furnishing the Japanese drugmaker with a newly approved gene therapy in the U.S. | The FDA on Monday gave a thumbs up to Lenmeldy as the first gene therapy in the U.S. for kids with certain types of metachromatic leukodystrophy (MLD).

BioSpace
MARCH 19, 2024
Two days after winning FDA approval, Orchard Therapeutics on Wednesday provided its U.S. launch plans for metachromatic leukodystrophy gene therapy Lenmeldy, which has a wholesale acquisition cost of $4.25 million for the one-time treatment.

BioSpace
MARCH 26, 2024
Nanoscope Therapeutics nabbed a victory on the changed primary endpoint of its Phase IIb vision loss gene therapy trial Tuesday, teeing the biotech up to file for FDA approval of the candidate this year.
pharmaphorum
DECEMBER 19, 2022
Ferring Pharma ’s Adstiladrin is a non-replicating (cannot multiply in human cells) adenoviral vector-based gene therapy. Dr Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, said: “This approval provides healthcare professionals with an innovative treatment option for [these] patients.”.

Drug Discovery World
JUNE 23, 2023
Sarepta Therapeutics’ Elevidys has become the first gene therapy for Duchenne muscular dystrophy (DMD) to gain marketing authorisation in the US. It is contraindicated in patients with any deletion in exon 8 and/or exon 9 in the DMD gene.
Pharmaceutical Technology
FEBRUARY 10, 2023
4D Molecular Therapeutics (4DMT), the California-based biotechnology company focused on developing gene therapies for rare and large market diseases, has had the FDA place a clinical hold onto its Fabry disease (FD) gene therapy program (4D-310).

XTalks
DECEMBER 13, 2023
The US Food and Drug Administration (FDA) has approved the first gene therapies for the treatment of sickle cell disease, approving two on the same day. The landmark approvals were awarded to bluebird bio’s Lyfgenia (lovo-cel) and Vertex Pharmaceuticals and CRISPR Therapeutics’ jointly developed Casgevy (exa-cel).

Bio Pharma Dive
SEPTEMBER 17, 2022
The therapy, called Skysona and cleared to treat cerebral adrenoleukodystrophy, is the product of more than a decade of work by Bluebird. It will cost $3 million.

BioSpace
MAY 21, 2023
Krystal Gets First FDA Approval for Redosable Gene Therapy, Rare Skin Disease 5/22/2023

Fierce Pharma
MARCH 2, 2023
With 2nd chance at FDA approval, BioMarin preps for hemophilia gene therapy launch in US zbecker Thu, 03/02/2023 - 16:28

Pharmaceutical Technology
NOVEMBER 9, 2022
On November 2, the Institute for Clinical and Economic Review (ICER) released its updated evidence aimed at measuring the clinical effectiveness and cost of the two haemophilia gene therapies. Known by the brand name Roctavian, BioMarin’s haemophilia A therapy valoctocogene roxaparvovec could be fairly priced in the range of $1.95–1.96

Bio Pharma Dive
APRIL 24, 2023
Submission of the application comes weeks after rivals Vertex and CRISPR filed their gene editing medicine for the disease with the agency.

Bio Pharma Dive
JUNE 30, 2023
The agency granted a long-awaited clearance on Thursday, but unexpected aspects of the hemophilia treatment’s label had some investors worried about its commercial prospects.

BioSpace
JUNE 21, 2023
Elevidys, authorized Thursday to treat ambulatory patients 4 to 5 years of age, is the first in vivo gene therapy to win the FDA’s accelerated approval. It is also the first such therapy for DMD.

BioSpace
DECEMBER 7, 2023
Friday’s FDA approval of Vertex-CRISPR’s Casgevy and bluebird bio’s Lyfgenia has immediately revealed startling differences between these two gene therapies: price and a black-box warning.

BioPharma Reporter
MARCH 19, 2024
Food and Drug Administration has cleared AGC Biologicsâ Milan site to begin manufacturing of Orchard Therapeuticsâ Lenmeldy (atidarsagene autotemcel), a gene therapy for early-onset metachromatic leukodystrophy (MLD).

BioSpace
JUNE 29, 2023
After an initial rejection, BioMarin has finally secured the FDA’s approval for Roctavian, the first gene therapy in the U.S. for the most common form of the bleeding disorder.

Drug Discovery World
DECEMBER 11, 2023
The US Food and Drug Administration (FDA) has approved the first cell-based gene therapies for the treatment of sickle cell disease (SCD), Casgevy and Lyfgenia. Casgevy, from Vertex Pharmaceuticals and CRISPR Therapeutics, is also the first FDA-approved treatment to use CRISPR gene editing technology.

BioSpace
DECEMBER 7, 2023
Along with CRISPR/Cas9-based Casgevy—developed by Vertex Pharmaceuticals and CRISPR Therapeutics—the regulator on Friday approved bluebird bio’s Lyfgenia, a second gene therapy for sickle cell disease.

STAT News
NOVEMBER 22, 2022
The Food and Drug Administration on Tuesday approved the first gene therapy to treat people with hemophilia B, an inherited bleeding disorder. The one-time treatment, called Hemgenix, was developed by the Dutch biotech company UniQure and will be marketed by CSL Behring, an Australian pharmaceutical company.

BioSpace
NOVEMBER 22, 2022
The FDA approved CSL Behring and uniQure’s Hemgenix, a one-time gene therapy developed for adults with Hemophilia B,the companies announced Tuesday afternoon.

Fierce Pharma
DECEMBER 19, 2023
Despite bluebird bio winning a recent FDA nod for its muc | Bluebird's FDA approval for sickle cell disease gene therapy Lyfgenia left something to be desired because the agency rejected a request for a priority review voucher. Without that voucher, bluebird is moving fast to try to fund its launches.

Drug Discovery World
MAY 29, 2023
The US Food and Drug Administration has approved Krystal Biotech’s Vyjuvek, a herpes-simplex virus type 1 (HSV-1) vector-based gene therapy, for the treatment of dystrophic epidermolysis bullosa (DEB). The post FDA approves first gene therapy for rare skin disorder appeared first on Drug Discovery World (DDW).
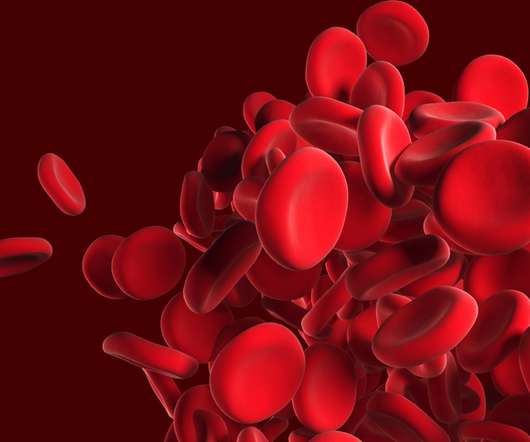
BioPharma Reporter
AUGUST 18, 2022
Food and Drug Administration (FDA) has approved bluebird bioâs Zynteglo (betibeglogene autotemcel), also known as beti-cel: a one-time gene therapy custom-designed to treat the underlying genetic cause of beta thalassemia in adult and pediatric patients who require regular red blood cell (RBC) transfusions.

BioSpace
MARCH 21, 2024
by ITF Therapeutics, joins a growing market that includes recently approved gene therapy Elevidys and corticosteroid Agamree. Duvyzat, which will be sold in the U.S.

Pharmaceutical Technology
MAY 22, 2023
The US Food and Drug Administration (FDA) has granted approval for Krystal Biotech’s Vyjuvek (beremagene geperpavec-svdt) to treat dystrophic epidermolysis bullosa (DEB) in patients aged six months and above. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.
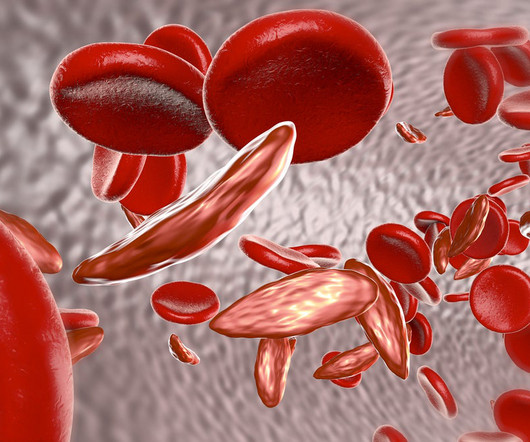
Pharmaceutical Technology
APRIL 13, 2023
Two gene therapies up for approval this year for sickle cell disease could be cost effective in some cases at a $2 million price point, based on a draft evidence report published by the Institute for Clinical and Economic Review (ICER). Also known as lovo-cel, bluebird bio’s product is a lentiviral gene therapy.

Pharmaceutical Technology
AUGUST 7, 2023
HuidaGene claims its gene therapy candidate was superior to FDA-approved Luxturna in preclinical studies.

BioPharma Reporter
MARCH 6, 2023
Sarepta Therapeutics would seem to be edging closer to accelerated FDA approval of its gene therapy for Duchenne muscular dystrophy (DMD).
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content