Vicore’s lung disease digital therapy gets breakthrough tag
pharmaphorum
MARCH 20, 2024
Vicore gets FDA breakthrough status for Almee, a cognitive behavioural therapy (CBT) digital health therapy used to support people with pulmonary fibrosis

 tag lung-diseases
tag lung-diseases 
pharmaphorum
MARCH 20, 2024
Vicore gets FDA breakthrough status for Almee, a cognitive behavioural therapy (CBT) digital health therapy used to support people with pulmonary fibrosis

Rethinking Clinical Trials
MARCH 1, 2023
S1800A was a Phase II randomized study of ramucirumab plus pembrolizumab versus standard of care for NSCLC patients previously treated with immunotherapy performed within the Lung-MAP platform. They are both approved and used widely in lung cancer, and combining two existing agents is logistically and clinically simpler.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Rethinking Clinical Trials
OCTOBER 11, 2023
Murray, PhD NIH Associate Director for Prevention and Director, NIH Office of Disease Prevention Moderator: Jonathan C. Moyer, PhD Statistician, NIH Office of Disease Prevention Slides Keywords Implementation; Study design; Hybrid; Clustered; DECIPHeR Key Points People often contest that hybrid designs are not as rigorous as they should be.

pharmaphorum
DECEMBER 23, 2021
Daiichi Sankyo has been granted breakthrough status by the FDA for patritumab deruxtecan, a HER3-targeted antibody-drug conjugate (ADC) in clinical trials for lung cancer. After EGFR therapy fails, platinum-based chemo has limited efficacy, generally preventing disease progression or death for between four and six months.
pharmaphorum
APRIL 27, 2022
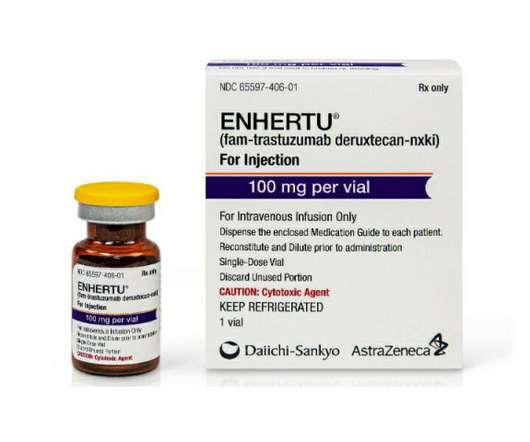
A breakthrough designation recognises that a drug could offer a substantial improvement over available therapies for serious or life-threatening diseases. The post Enhertu gets breakthrough tag in HER2-low breast cancer appeared first on. billion upfront to license rights to the drug in a deal that could be worth up to $6.9
XTalks
OCTOBER 26, 2021
The treatment came with an initial price tag of $39,000 per vial, which amounts to a total of $575,00 each year. Seizures occur due to the buildup of neurotoxins The condition can also cause other issues such as paralysis, insomnia and problems relating to the heart and lung. This leads to lowering of ALA and PBG levels.

pharmaphorum
JULY 25, 2022
That alliance also yielded $6 billion blockbuster Darzalex as well as Rybrevant (amivantamab), J&J’s first bispecific antibody which is used to treat EGFR-mutated non-small cell lung cancer (NSCLC). Most patients diagnosed with myeloma will relapse over the course of their disease, so the disease is still considered incurable.
Let's personalize your content