Sanders takes aim at US drug prices of Novo’s Ozempic and Wegovy
Pharmaceutical Technology
APRIL 25, 2024
A Senate Committee is investigating the “outrageously high” US price tags of the blockbuster diabetes and obesity drugs.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 tag us-drug-pricing
tag us-drug-pricing 
Pharmaceutical Technology
APRIL 25, 2024
A Senate Committee is investigating the “outrageously high” US price tags of the blockbuster diabetes and obesity drugs.

Fierce Pharma
MARCH 20, 2024
Kyowa Kirin and its subsidiary Orchard Therapeutics have broken new boundaries in pricing, too. After scoring an FDA nod Monday for the first gene therapy to treat the rare genetic disease metachromatic leukodystrophy (MLD) in the U.S.,
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
SEPTEMBER 30, 2022
Spark Therapeutics’ Luxturna, indicated for inherited retinal disease (IRD), was the first gene therapy to be approved, in 2017, with a price tag of $850,000 for each eye. bluebird then broke its own pricing record when the $3m Skysona was approved, and now faces the unique challenge of selling the world's two most expensive drugs.
Pharmaceutical Technology
FEBRUARY 10, 2023
On November 22, 2022, the FDA approved CSL Behring’s Hemgenix (etranacogene dezaparvovec), the first gene therapy treatment for hemophilia B, with a staggering manufacturer price of $3.5 million, according to GlobalData’s Price Intelligence (POLI), making it the most expensive drug in the world.
Pharmaceutical Technology
JUNE 21, 2023
per dose, it is the most expensive single-use gene therapy in the US. The high price tag may be hard to defend , but with lifetime treatment costs for haemophilia B reaching as high as $23m for some, the single-dose treatment could prove far more cost-effective. Haemophilia B is a rare condition.
Pharmaceutical Technology
MAY 19, 2023
DMTs for MS have a high price tag, particularly in the US. According to GlobalData’s recent Multiple Sclerosis: Seven-Market Drug Forecast and Market Analysis to 2030 – Update report, which identified 19 DMTs marketed for MS in the US, the average annual cost of therapy increased by 9% between 2020 and 2022.

World of DTC Marketing
JUNE 16, 2021
This includes about one in five who report that they have not filled a prescription or took an over-the counter drug instead, and about one in ten who say they have cut pills in half or skipped a dose. The public sees profits made by pharmaceutical companies as the largest factor contributing to the price of prescription drugs.
STAT News
OCTOBER 6, 2022
And this is especially true for us, since we hit the proverbial pause button yesterday to observe ancient rituals. Please feel free to join us. Even a drug-price watchdog, the Institute for Clinical and Economic Review, deemed the $2.8 Rise and shine, everyone, another busy day is on the way.

pharmaphorum
NOVEMBER 16, 2022
The advancements made in drug development over the last decade have seen the arrival of a number of treatments that offer one-time cures and more effective therapies for conditions in smaller patient populations. However, alongside this progress, Ben Hargreaves finds that there is also a conundrum over how to price these therapies.
Pharmaceutical Technology
DECEMBER 15, 2022
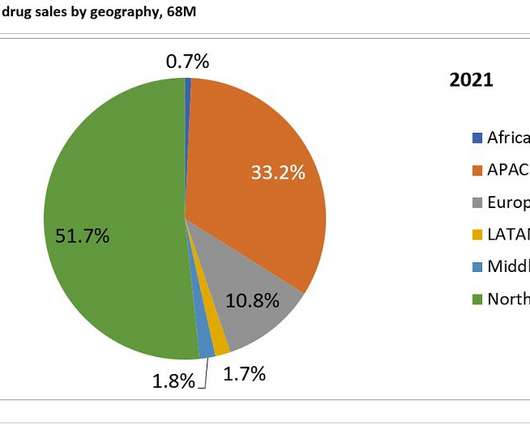
In an expanded version of the sales forecast from GlobalData’s June 2022 publication, Cardiomyopathies: Global Drug Forecast and Market Analysis , the global cardiomyopathies market is expected to grow from $3.07bn last year to $9.63bn in 2031. of sales in 2021 and 2031 respectively. of global cardiomyopathies sales in 2021 and 31.2%
Pharmaceutical Technology
JUNE 29, 2022
The US Food and Drug Administration (FDA) put a high-profile bluebird bio trial for sickle cell disease on partial clinical hold, and advisory panels deliberated over decisions involving gene therapies for amyotrophic lateral sclerosis (ALS), cerebral adrenoleukodystrophy (CALD), and beta-thalassemia.

pharmaphorum
OCTOBER 3, 2022
Amylyx Pharmaceuticals’ Relyvrio – a new treatment for amyotrophic lateral sclerosis (ALS) approved by the FDA on Thursday, after review of the data from its phase 2 trial – was the next day set at a list price of $158,000 per year in the US, sparking outcry. ALS affects over 30,000 people in the US.
pharmaphorum
DECEMBER 24, 2021
The anti-amyloid drug, which also claimed a breakthrough tag from the FDA in July, is being developed for the treatment of early-stage Alzheimer’s disease. The $56,000-a-year price tag for the drug, which has now been cut in half in the US, also hasn’t helped the drugmakers make a case for Aduhelm with US payers.
Pharmaceutical Technology
JULY 14, 2022
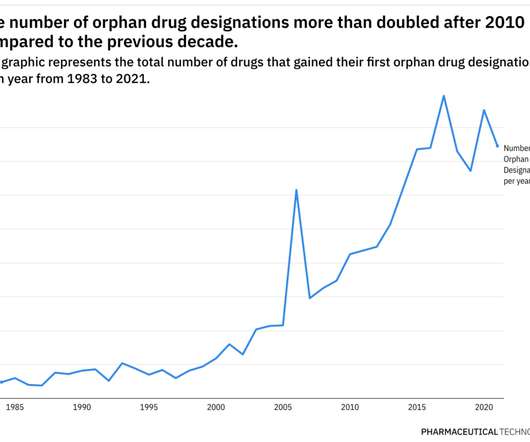
The commercial investment required to research and develop an innovative drug, prove its safety and efficacy, and bring it to market is staggering. Most famously, the US passed its Orphan Drug Act in 1983, providing innovators with financial motivation to develop orphan drugs and meet the needs of these forgotten patients.

XTalks
DECEMBER 13, 2023
The US Food and Drug Administration (FDA) has approved the first gene therapies for the treatment of sickle cell disease, approving two on the same day. Casgevy is also the first ever CRISPR/Cas9-based therapy approved in the US. Vertex-CRISPR’s Casgevy has a US list price of $2.2 million after discounts.

Pharmaceutical Technology
FEBRUARY 22, 2023
Amgen could be the winner of a high-stakes patent spat with Sanofi as tensions run high weeks before the March 27 US Supreme Court hearing. The legal dispute is centered around rival cholesterol-lowering drugs produced by the two companies, which were approved in the summer of 2015. Both drugs come with a high price tag.

pharmaphorum
AUGUST 26, 2022
The US biotech claimed conditional marketing approval from the European Commission for Roctavian (valoctocogene roxaparvovec) as a treatment for adults with severe haemophilia A, who don’t have a history of developing the antibodies that inhibit the activity of blood-clotting protein Factor VIII. million spread over five years.

XTalks
JULY 12, 2021
The US Food and Drug Administration’s (FDA) recent approval of Biogen’s Alzheimer’s drug Aduhelm (aducanumab) was met with a lot of backlash, prompting the health agency to narrow its recommendation of the drug for mild forms of the disease.

pharmaphorum
NOVEMBER 19, 2020
Novartis/Genentech’s eye drug Lucentis could be the next big blockbuster to face competition from cheaper biosimilars after its US patent expired this year – and Samsung Bioepis and Biogen are closing in after the FDA accepted a filing for their cut-price rival.

pharmaphorum
JUNE 8, 2021
The FDA “has failed in its responsibility to protect patients and families from unproven treatments with known harms” in approving Biogen’s Alzheimer’s disease drug Aduhelm. It said the drug was the first to treat the underlying mechanism in Alzheimer’s rather than addressing symptoms.
Drug Discovery World
DECEMBER 20, 2022
The therapy was also recently approved by the FDA for patients in the US. . Thank you to all for the role you have played in helping us reach this regulatory milestone in Europe.” . World’s most expensive drug. A one-time treatment with etranacogene dezaparvovec costs US$3.5

Rethinking Clinical Trials
JUNE 7, 2023
The drugs were administered open label and all patients were given a low dose of aspirin. The study drug was mailed directly to participants, and at the end of the trial participants were instructed to destroy the drug. The price for Apixaban was $600 per bottle/$6M, and the price for warfarin was $50K.
XTalks
JUNE 9, 2021
Biogen has been working vigorously over the past couple of years to get its potentially disease-modifying drug Aduhelm (aducanumab) approved for Alzheimer’s disease, and it has finally happened. This Monday, the US Food and Drug Administration (FDA) granted accelerated approval to the drug for the treatment of the disease.

Pharmaceutical Technology
MARCH 17, 2023
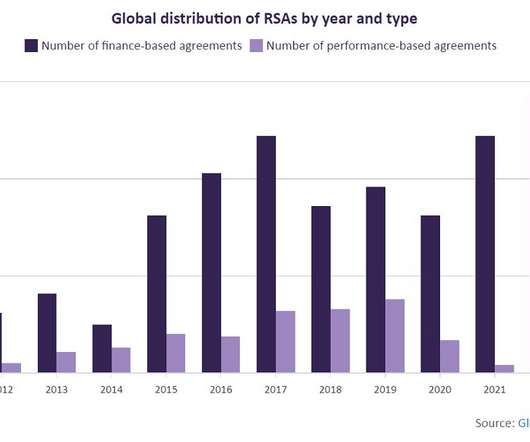
These agreements have been particularly propelled by the recent rise in inflationary pressures and pharmaceutical market instability, pushing healthcare systems to find new ways to reduce risks associated with new drugs. Thus, a higher risk score reflects a more hostile market access environment and drug launch in a geographical area.

pharmaphorum
AUGUST 21, 2022
There is still a pressing need for antidepressant therapies that can start to work more quickly than current tricyclic or selective serotonin reuptake inhibitor drugs can take weeks to elevate mood, placing severely affected patients at risk of self harm before they kick in. Photo by Sydney Sims on Unsplash.

Drug Discovery World
MAY 22, 2023
While the global economic landscape faces a major crisis and biotech is not recession-proof, cell and gene therapy (CGT) has continued to progress with acquisitions, breaking ground on new facilities and approvals across the year – not limited to the breakout of the world’s most expensive drug. But are these price tags a barrier to access?
XTalks
MARCH 22, 2023
To celebrate this milestone, we’re taking a look back over the last 20 years in the pharmaceutical industry by highlighting some of the most talked-about mergers, blockbuster drugs, trends in FDA approvals and what’s next. Within 2009 and 2010, Swiss pharmas Roche and Novartis acquired Genentech and Alcon , respectively.

pharmaphorum
MARCH 27, 2022
Both CAR-Ts are directed at BCMA, a biomarker for multiple myeloma also targeted by GlaxoSmithKline’s approved antibody-drug conjugate Blenrep (belantamab mafodotin) and J&J’s experimental bispecific antibody teclistamab, which was submitted for approval in Europe last month.

XTalks
FEBRUARY 8, 2024
Pharma commercials at Super Bowl 2024 will include ones for newly approved menopause drug Veozah and a unique partnership between antacid giant TUMS and sports betting company Draft Kings. After all, nearly half of women in the US going through menopause may experience moderate to severe VMS.

Drug Discovery World
JANUARY 30, 2024
Joel Eichmann, Co-Founder and Managing Director, Green Elephant Biotech, agrees that prices need to come down significantly for CGT to be widely available. Pricing and reimbursement continues to be a challenge we address as a field. Pricing and reimbursement continues to be a challenge we address as a field.
pharmaphorum
JUNE 2, 2022
R&D into orphan drugs is growing alongside the number of approved treatments, providing treatments for rare diseases that previously did not have any. This is why the action by the US in 1983 and subsequently the European Commission Regulation No. The price is right. It is estimated that between 3.5

XTalks
NOVEMBER 24, 2022
In a pivotal approval, the US Food and Drug Administration (FDA) has given the nod to a new gene therapy called Hemgenix (etranacogene dezaparvovecfor) for the treatment of adults with the genetic blood disorder hemophilia B (congenital Factor IX deficiency). With a list price of $3.5

pharmaphorum
AUGUST 10, 2020
Roche is hoping to undercut hugely expensive rivals after the FDA approved its oral spinal muscular atrophy (SMA) drug Evrysdi (risdiplam). But Spinraza costs $750,000 in the first year of treatment and about half that price annually from then on. The post Roche takes on pricey rivals as FDA approves SMA drug appeared first on.

XTalks
SEPTEMBER 27, 2022
With a list price of $3 million and a recent accelerated US Food and Drug Administration (FDA) approval, bluebird bio’s gene therapy Skysona (elivaldogene autotemcel, eli-cel), has officially become the world’s most expensive drug. Together, this amounts to about 40 cases a year in the US. Related: Bluebird’s $2.8M

pharmaphorum
APRIL 14, 2022
US cost-effectiveness watchdog ICER has handed bluebird bio some good news ahead of its FDA advisory committee meeting for rare blood disorder gene therapy beti-cel in June, by endorsing its proposed $2.1 million price tag. million if paid through an outcomes-based contract for patients with sustained transfusion independence.”

pharmaphorum
JUNE 12, 2022
The FDA still has to deliver its own verdict on the applications, and doesn’t always follow its advisors’ lead, but with two unanimous decisions bluebird looks set up for its first approvals in the US. Beti-cel has already been approved for marketing in Europe as Zynteglo, with a price tag of around $1.8

XTalks
APRIL 29, 2022
Eli Lilly announced that its obesity drug tirzepatide has scored favorably in a late-stage clinical trial, with results showing that people who took the drug lost an average of 50 pounds, or 21 percent, of their body weight compared to placebo. In the US, over 73 percent of Americans are obese or overweight, with one third being obese.

pharmaphorum
JANUARY 9, 2023
” Zolgensma is one of the most expensive therapies available, with a price tag of around $2.1 million in the US and just under €2 million in Europe, which the company says is justified by its dramatic benefits when given as a one-shot therapy to patients with SMA.

pharmaphorum
APRIL 5, 2022
Kimmtrak (tebentafusp) has been cleared to for the treatment of HLA-A*02:01-positive adult patients with unresectable or metastatic uveal melanoma (mUM), mirroring its label in the US. It’s the first treatment for mUM in the EU, as well as the first BiTE drug to treat a solid tumour. ” Image by Pexels from Pixabay .

XTalks
JUNE 8, 2021
With a price tag of over $2.5 million, Zolgensma is currently the world’s most expensive drug. It was first approved in the US in 2019 to treat children below the age of two with SMA. They also advised use of Zolgensma for infants with SMA type 1 before they develop symptoms.

XTalks
AUGUST 19, 2022
Bluebird bio’s gene therapy Zynteglo (betibeglogene autotemcel, beti-cel) has been awarded a much anticipated approval from the US Food and Drug Administration (FDA) for the treatment of adult and pediatric patients with beta thalassemia who need regular blood transfusions. Zynteglo’s hefty price tag of $2.8

pharmaphorum
NOVEMBER 28, 2022
Substra is the software that powered the MELLODDY platform, a collaboration in which Amgen, Astellas, AstraZeneca, Bayer, Boehringer Ingelheim, GSK, Janssen, Merck, Novartis, and Servier all agreed to share drug discovery data — but not with each other directly, nor with Owkin.

pharmaphorum
MAY 17, 2021
Empaveli’s label covers treatment-naïve PNH patients as well as those who want to switch from Soliris or Ultomiris, and is based on the phase 3 PEGASUS trial in 80 patients which compared the drug to Soliris over a 16-week period. The post Apellis set to take on Alexion as FDA clears PNH drug Empaveli appeared first on.

XTalks
FEBRUARY 27, 2024
Iovance Biotherapeutics’ Amtagvi (lifileucel) won US Food and Drug Administration (FDA) approval last week for the treatment of advanced melanoma, making it the first individualized tumor-infiltrating lymphocyte (TIL) therapy and the first T-cell therapy for a solid tumor to win US regulatory approval.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content