Xyphos and Poseida to develop allogeneic cell therapies for cancer
Pharmaceutical Technology
MAY 2, 2024
Xyphos Biosciences and Poseida Therapeutics have signed a deal to develop new allogeneic cell therapies for treating cancer.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 allogene
allogene 
Pharmaceutical Technology
MAY 2, 2024
Xyphos Biosciences and Poseida Therapeutics have signed a deal to develop new allogeneic cell therapies for treating cancer.

Bio Pharma Dive
MARCH 12, 2024
Allogene’s partnership with Arbor is designed to help the company stand out in what’s quickly become one of the industry’s hottest areas of drug research.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
APRIL 23, 2024
Discover the groundbreaking patent by Allogene Therapeutics for CD19-specific CARs resistant to rituximab binding. Learn about the innovative methods and features of these engineered immune cells.
Bio Pharma Dive
OCTOBER 7, 2022
The biotech believes the two Phase 2 trials initiated Thursday can support approval applications for what could be the first so-called allogeneic cancer cell therapy.
Bio Pharma Dive
JANUARY 10, 2022
A "chromosomal abnormality" that led regulators to halt Allogene's trials was judged to be unrelated to its technology, a finding with important implications for the field of donor-derived cell therapies.

Bio Pharma Dive
OCTOBER 7, 2021
Researchers found evidence of a "chromosomal abnormality" in one patient treated with Allogene's CAR-T cell therapy, spurring the clinical hold and an investigation.

Pharmaceutical Technology
MARCH 20, 2024
Cellectis presented promising data with UCART20x22, which represents a pioneering allogeneic dual CAR T-cell product designed to target two established antigens.

Bio Pharma Dive
SEPTEMBER 22, 2022
The French drugmaker terminated a nearly decade-old deal that helped Allogene emerge as the sector's largest "off-the-shelf" cell therapy developer.

BioSpace
APRIL 14, 2024
Roche’s Genentech subsidiary is terminating for undisclosed reasons its 2021 contract with Adaptimmune for the development of allogeneic T-cell therapeutics.

Roots Analysis
MAY 21, 2024
The cells used in the cell therapy can be from the patient’s own body (autologous) or from a donor (allogeneic). In allogeneic cell therapies, the cells are engineered ( to elicit a desired therapeutic response ) and amplified in-vitro. The modified cells can be preserved in a cell bank or administered to the patients.

Pharmaceutical Technology
FEBRUARY 8, 2024
Cemacabtagene ansegedleucel is under clinical development by Allogene Therapeutics and currently in Phase II for Follicular Lymphoma.

Pharmaceutical Technology
JULY 18, 2023
The development of allogeneic (allo) cell therapies has been a major focus of the biopharmaceutical industry for the last several years.

Pharmaceutical Technology
FEBRUARY 8, 2024
Cemacabtagene ansegedleucel is under clinical development by Allogene Therapeutics and currently in Phase II for Refractory Chronic Lymphocytic Leukemia (CLL).

Pharmaceutical Technology
FEBRUARY 8, 2024
Cemacabtagene ansegedleucel is under clinical development by Allogene Therapeutics and currently in Phase II for Relapsed Chronic Lymphocytic Leukemia (CLL).

Pharmaceutical Technology
FEBRUARY 8, 2024
Cemacabtagene ansegedleucel is under clinical development by Allogene Therapeutics and currently in Phase II for Primary Mediastinal B-Cell Lymphoma.

BioSpace
OCTOBER 6, 2022
Allogene Therapeutics is launching what it calls the first pivotal Phase II trial of an allogeneic CAR T product in the industry. The ALPHA2 trial will evaluate ALLO-501A in LBCL.

Pharmaceutical Technology
FEBRUARY 8, 2024
Cemacabtagene ansegedleucel is under clinical development by Allogene Therapeutics and currently in Phase II for Diffuse Large B-Cell Lymphoma.

Pharmaceutical Technology
FEBRUARY 8, 2024
Cemacabtagene ansegedleucel is under clinical development by Allogene Therapeutics and currently in Phase II for Marginal Zone B-cell Lymphoma.
Pharmaceutical Commerce
MAY 3, 2024
Collaboration will be oncology based—centered around allogeneic CAR-T cell therapy product candidates—and will provide Poseida with up to $550 million if certain milestones are met.
Drug Discovery World
MAY 2, 2024
Allogene Therapeutics has received a $15 million grant from the California Institute for Regenerative Medicine (CIRM) to support the development of ALLO-316, an AlloCAR T investigational product for the treatment of advanced or metastatic renal cell carcinoma (RCC).
Bio Pharma Dive
JANUARY 4, 2024
The company plans to restructure and will start a new late-stage study testing whether its “off-the-shelf” therapy can boost cure rates when used early on.
Bio Pharma Dive
FEBRUARY 24, 2022
Allogene, once the clear leader in development of a more convenient option to personalized cancer cell therapies, now is competing with CRISPR Therapeutics and other fast-emerging rivals.

pharmaphorum
FEBRUARY 15, 2024
The 6th Allogeneic Cell Therapies Summit, driving potency, efficacy & durability of universal cell therapies. Coming to Boston, MA this June 12th-14th 2024.
Drug Discovery World
APRIL 12, 2024
The company plans to initiate a Phase I study of the investigational allogeneic off-the-shelf Tr1 Treg therapy for this indication in the second quarter of 2024. ” “Allogeneic stem cell transplantation is the only curative treatment for many advanced blood cancers and genetic and acquired diseases.
Drug Discovery World
APRIL 28, 2023
The funding has been awarded to enable the two organisations to carry out a project – valued at £400,000 ($499,000) – which aims to create a broadly-applicable iPSC-derived allogenic immunotherapy platform. The post UK-Singapore alliance focuses on allogeneic cancer immunotherapy appeared first on Drug Discovery World (DDW).
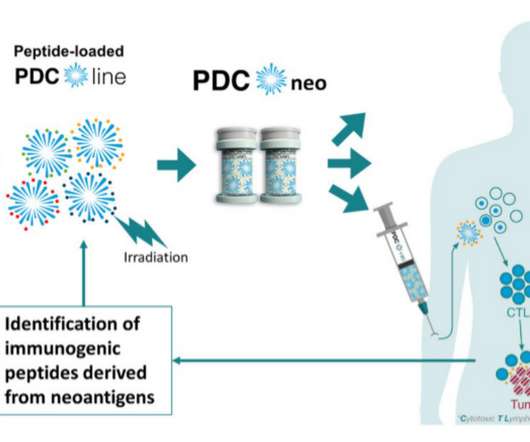
Medical Xpress
FEBRUARY 3, 2023
A new research perspective was published in Genes & Cancer on January 30, 2023, entitled, "Leveraging a powerful allogeneic dendritic cell line towards neoantigen-based cancer vaccines."
Fierce Pharma
DECEMBER 19, 2022
Atara makes history with world-first nod for allogeneic T-cell therapy Ebvallo. fkansteiner. Mon, 12/19/2022 - 14:06.
Drug Discovery World
JULY 27, 2023
The Phase I study was a first-in-human trial to investigate safety, immunogenicity, and survival in patients with advanced NSCLC (defined as metastatic or locally advanced disease) when administered the allogeneic dendritic cell (DC) vaccine VAC2.

Drug Discovery World
MAY 5, 2023
The acquisition will provide Eterna with a pipeline of allogeneic immuno-oncology products under development for the treatment of haematologic and solid tumours. The post Eterna acquires Exacis’ allogeneic immuno-oncology platform appeared first on Drug Discovery World (DDW).
BioSpace
JUNE 8, 2022
Precision BioSciences' clinical program exploring allogeneic CAR T therapy may well produce a market pioneer after initial data showed its ability to deliver a 100% overall response rate.
BioSpace
NOVEMBER 11, 2023
The investigational allogeneic CAR-T therapy P-BCMA-ALLO1 appears to be more effective following strong immunosuppressive preconditioning.
Drug Discovery World
FEBRUARY 10, 2023
Pierre Fabre will commercialise and distribute the first approved allogeneic T cell immunotherapy in Europe following the transfer of the European Commission marketing authorisation of Ebvallo (tabelecleucel) from Atara Biotherapeutics. To read this content in full, you need to login. Not yet a DDW member?

BioSpace
AUGUST 4, 2021
Gilead Sciences’ Kite Pharma has entered into a collaboration and licensing agreement with biotech startup Appia Bio, which will focus on the joint research and development of allogeneic cell therapies for cancer.

Bio Pharma Dive
JULY 6, 2023
Five years after helping launch Allogene Therapeutics, Pfizer is backing another developer of “off-the-shelf” cell therapies in a deal that gives the drugmaker the chance to acquire a multiple myeloma treatment.
BioSpace
NOVEMBER 9, 2021
Eric Ostertag, CEO of Poseida Therapeutics, believes the future of CAR-T programs is with allogeneic approaches and not autologous.

BioSpace
DECEMBER 14, 2020
Clinical-stage biotechnology company Allogene Therapeutics and start-up company Overland Pharmaceuticals are forming a new joint company named Allogene Overland Biopharm.

BioPharma Reporter
AUGUST 11, 2022
has received regulatory approval - in the form of Good Manufacturing Practice (GMP) certification - to commercially produce allogeneic cell therapy drug products for distribution in Europe, from the European Medicines Agency (EMA). Charles River Laboratories, International Inc.
BioPharma Reporter
NOVEMBER 30, 2021
The EMA has validated Atara Biotherapeutics' Marketing Authorization Application (MAA) for tabelecleucel (tab-cel), the first time an off-the-shelf allogeneic T-cell therapy will be evaluated by any regulatory agency in the world.

Bio Pharma Dive
AUGUST 5, 2021
Appia Bio, which launched with over $50 million in Series A funding earlier this year, will provide preclinical research for two CAR-iNKT candidates, which Kite will pick up should they advance past early clinical testing.
Bio Pharma Dive
AUGUST 3, 2022
The pharma has now signed two alliances with allogeneic drug developers since September, signalling its interest in CAR-T treatment alternatives.

BioSpace
OCTOBER 7, 2021
Allogene placed its study on hold after sending a notification to the U.S. FDA about a chromosomal abnormality in its ALPHA2 study patient.

pharmaphorum
OCTOBER 8, 2021
A phase 1 trial of Allogene’s of-the-shelf CAR-T for lymphoma has been placed on hold by the FDA, after a “chromosomal abnormality” was seen in a patient receiving the cell therapy. Off the shelf or allogeneic cell therapies are derived from a bank of cells and can be manufactured and administered in bulk like drugs.
Bio Pharma Dive
NOVEMBER 4, 2020
Results the company will present at a medical meeting, while early, don't appear to meet the high bar set by rival CAR-T cell therapies from Bristol Myers Squibb and Johnson & Johnson.
BioSpace
FEBRUARY 10, 2021
Precision BioSciences recently received a notice of allowance for a new patent for PBCAR19B, its stealth cell, CD19 allogeneic CAR T candidate for non-Hodgkin lymphoma.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content