Disc wins orphan drug tag for rare blood cancer
Pharmaceutical Technology
FEBRUARY 12, 2024
The humanised monoclonal antibody DISC-3405 is under investigation in a Phase I clinical trial, with data expected this year.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Pharmaceutical Technology
FEBRUARY 12, 2024
The humanised monoclonal antibody DISC-3405 is under investigation in a Phase I clinical trial, with data expected this year.

Pharmaceutical Technology
JANUARY 9, 2023
Eisai and Biogen have received approval for their antibody Leqembi (lecanemab-irmb) , 100mg/mL injection for intravenous use, from the US Food and Drug Administration (FDA) under the Accelerated Approval Pathway to treat Alzheimer’s disease (AD). The regulatory approval is based on the data obtained from the Phase II trial.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
JULY 21, 2022
AstraZeneca has signed a deal with the Federal Office of Public Health (FOPH) of Switzerland to deliver over 1,200 doses of antibody therapy, tixagevimab and cilgavimab combination (AZD7442), for Covid-19 prevention and treatment. In June 2020, these antibodies, discovered at Vanderbilt University Medical Center, were licensed to AstraZeneca.
Drug Discovery World
OCTOBER 11, 2023
Collaborators Boehringer Ingelheim and CDR-Life have commenced a Phase I evaluation of BI 771716, their antibody fragment-based treatment developed to preserve the vision of people with geographic atrophy (GA). The company’s first therapeutic candidate, CDR404, targets MAGE-A4 and is expected to enter the clinic in 2024.

Pharmaceutical Technology
FEBRUARY 14, 2023
Precision oncology firm Corbus Pharmaceuticals and CSPC Megalith Biopharmaceutical have entered an exclusive licensing agreement for the latter’s new clinical stage antibody drug conjugate (ADC), CRB-701 (SYS6002). We look forward to working with CSPC to advance clinical development of this asset and realise its full potential.”

XTalks
JANUARY 17, 2024
Oncology is recognized as having the largest drug pipeline of any therapeutic area. Given this hotbed of activity, innovation in the space to drive faster decisions and more efficient trials is intense. It’s very positive and it’s going to help move forward the pathway for new drugs.”
Drug Discovery World
MAY 1, 2024
DDW’s Megan Thomas speaks to industry experts and thought leaders about the potential for therapeutic antibodies in 2024. Many of the best-selling drugs in 2023 were monoclonal antibody (mAB) therapies, including Keytruda (pembrolizumab), Humira (adalimumab), and Dupixent (dupilumab) 1.
XTalks
MAY 2, 2022
This year, the monoclonal antibody lecanemab is approaching final stages of investigation as a treatment for targeting the A? The Phase IIb clinical trial focused on the long-term clinical outcomes and safety for patients with MCI and amyloid pathology (early AD). The main pathological markers of AD are beta-amyloid (A?)
Drug Discovery World
MARCH 10, 2023
Cancer Research UK and UCB have agreed a clinical development collaboration to advance two of UCB’s investigational oncology antibody candidates through clinical trials. If successful in clinical trials the investigational candidates may have the potential to offer cancer patients access to new targeted treatment options.

Drug Discovery World
MARCH 12, 2024
DDW’s Megan Thomas speaks to experts from the drug discovery industry about their predictions on what 2024 holds for our sector. Here, experts weigh in on the future of monoclonal antibodies (mAbs). Amanda Halford, President Bioprocess, Cytiva “In 2024, the emphasis will remain on novel mAbs, specifically antibody drug conjugates.
Bio Pharma Dive
JUNE 21, 2021
Final clinical trial results confirmed treatment dramatically reduced the risk of hospitalization and death among adults with mild-to-moderate COVID-19.

pharmaphorum
FEBRUARY 8, 2024
Gilead Sciences has said it will no longer develop its CD47-targeting antibody magrolimab in blood cancers after data suggested that patients taking the drug in clinical trials were more likely to die than those in control groups.

BioPharma Reporter
DECEMBER 13, 2021
Memo Therapeutics AG will receive CHF 10.5m (US $11.4m) from the Swiss government to clinically develop a SARS-CoV-2 antibody against COVID-19, allowing it to start Phase 1 studies in Q1, 2022.
Drug Discovery World
MARCH 4, 2024
In an early phase clinical trial, a combination of antibody-based medications targeting the immune system generated promising safety data and anti-tumour activity in individuals with advanced cancer. Both medications tested in the trial support immune responses against tumour cells.
Drug Discovery World
JULY 26, 2023
A Cancer Research UK-funded clinical trial has shown, for the first time, that a new class of antibody could benefit cancer patients whose existing treatments have stopped working. The drug, MOv18 IgE, was developed by researchers at King’s College London.
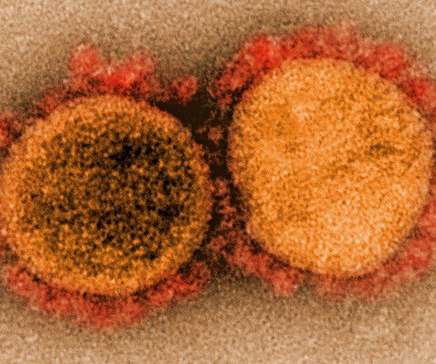
Bio Pharma Dive
JANUARY 20, 2021
Results from a clinical trial in nursing home residents and staff offered the first evidence Lilly's bamlanivimab could protect against, as well as treat, COVID-19
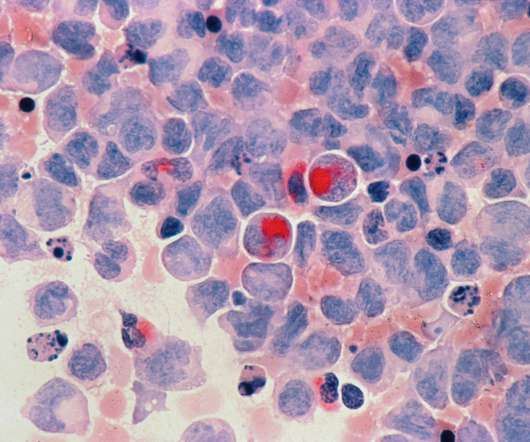
Medical Xpress
NOVEMBER 21, 2022
Over the past two decades, numerous complex cancer therapies called antibody drug conjugates (ADCs) have been tested in clinical trials and approved for use in patients. Their findings are published online in Cancer.

Roots Analysis
FEBRUARY 27, 2024
Ambrx and NovoCodex Biopharmaceuticals announced that they have formed a second collaboration to develop and commercialize Ambrx’s internally developed site-specific antibody drug conjugates. The antibody drug conjugates market is expected to grow at a CAGR of ~10% during the forecast period.

Medical Xpress
APRIL 10, 2023
Repeat radiation therapy combined with the monoclonal antibody drug bevacizumab extended progression-free survival in patients with recurrent glioblastoma, according to findings from a recent clinical trial published in the Journal of Clinical Oncology.

XTalks
MAY 4, 2023
A blood test can detect antibodies to gluten, but a definitive diagnosis requires a biopsy of the small intestine. As of now, the US Food and Drug Administration (FDA) has not approved any drugs for the treatment of celiac disease. How does Beyond Celiac spread awareness about research and clinical trials for celiac disease?

Pharmaceutical Technology
SEPTEMBER 16, 2022
The US Food and Drug Administration (FDA) has granted fast track designation to Jasper Therapeutics’ antibody, JSP191, for the treatment of severe combined immunodeficiency (SCID) patients who undergo allogeneic hematopoietic stem cell transplant. .
STAT News
DECEMBER 11, 2022
NEW ORLEANS — An antibody treatment developed by the Belgian drugmaker Argenx raised platelet counts and stopped bleeding episodes in patients with a rare autoimmune disorder that causes the body to attack and destroy its own blood-clotting platelets.
Drug Discovery World
APRIL 14, 2023
Oliver Schon, PhD, is VP Research & Development at BiVictriX Therapeutics, a UK-based emerging drug discovery and development company generating a pioneering first-in-class next-generation ADC anti-cancer therapeutics which exhibit superior selectivity towards cancer.
STAT News
JANUARY 10, 2023
SAN FRANCISCO — For months, drugmakers have been pleading with regulators to lower the bar for authorizing antibody drugs for Covid. The virus, they noted, had evolved fast enough to render every previous antibody obsolete.
Drug Discovery World
OCTOBER 25, 2023
Iksuda Therapeutics has dosed the first patient in a Phase I trial of antibody drug conjugate (ADC) IKS014 in patients with advanced solid tumours that express HER2. The post First patient dosed with antibody drug conjugate for HER2+ solid tumours appeared first on Drug Discovery World (DDW).

Drug Discovery World
MARCH 8, 2024
The top stories: Antibody therapy shows anti-tumour activity in advanced cancer In an early phase clinical trial, a combination of antibody-based medications targeting the immune system generated promising safety data and anti-tumour activity in individuals with advanced cancer.

STAT News
NOVEMBER 13, 2022
Roche reported negative results Monday from a pair of clinical trials investigating an antibody treatment for people with early-stage Alzheimer’s disease — another setback for the Swiss pharma giant’s decades-long effort to develop medicines against the memory-destroying disease.

Drug Discovery World
MARCH 8, 2024
Biopharmaceutical company Innovent Biologics has launched the first-in-human (FIH) Phase I clinical trial of IBI3002, a novel bispecific antibody targeting Interleukin 4 receptor α (IL-4Rα) and thymic stromal lymphopoietin (TSLP). IBI3002 has high-efficient dual-blocking function on both IL-4Rα and TSLP.
Drug Discovery World
DECEMBER 8, 2023
ONK Therapeutics and NAYA Biosciences have agreed to develop a combination therapy of ONK’s optimally-engineered natural killer (NK) cell therapies and NAYA’s FLEX-NK bispecific antibodies. ONK and NAYA plan to assess several combination therapies in preclinical cancer models in 2024 prior to subsequently exploring initiating clinical trials.

XTalks
JULY 25, 2022
Byondis announced that the US Food and Drug Administration (FDA) accepted their Biologics License Application (BLA) for [vic-]trastuzumab duocarmazine (SYD985), an investigational therapy being studied to treat HER2-positive unresectable locally advanced or metastatic breast cancer. Clinical Trial Results for [Vic-]Trastuzumab Duocarmazine

Pharmaceutical Technology
MAY 22, 2023
The Center for Drug Evaluation (CDE) of China’s National Medical Products Administration (NMPA) has accepted an investigational new drug application (IND) for SinoMab BioScience’s SM17 to treat asthma. SinoMab BioScience intends to commence a Phase I clinical trial in China to evaluate SM17’s safety profile.

XTalks
APRIL 22, 2022
Flow cytometric receptor occupancy assays are being increasingly used in preclinical and clinical studies. Both the areas of drug development and clinical trials are increasingly using in vitro assays to help determine the efficacy of an investigational therapeutic. What is Flow Cytometry?
STAT News
NOVEMBER 28, 2022
… A 65-year-old woman who was receiving a promising experimental treatment to slow the cognitive decline caused by her early Alzheimer’s disease recently died from a massive brain hemorrhage that some researchers link to the drug , Science reports. STAT reported on another such case last month.
XTalks
OCTOBER 16, 2020
The US Food and Drug Administration (FDA) has approved Inmazeb (atoltivimab, maftivimab and odesivimab-ebgn) as the world’s first treatment for Zaire ebolavirus (Ebola virus) infection in adult and pediatric patients, including newborns of mothers who have tested positive for the virus. Targeted Antiviral Treatment.

Drug Discovery World
APRIL 3, 2023
A team of researchers at the Keck School of Medicine of University of Southern California (USC) have found a drug with the potential for curbing painful hyperinflammation from osteoarthritis, according to results of an animal study. In trying to fix the problem, the immune system causes even more damage,” said Evseenko.

BioTech 365
FEBRUARY 6, 2022
Celltrion Submits Investigational New Drug (IND) Application to Initiate a Global Phase III Clinical Trial Evaluating an Inhaled COVID-19 Antibody Cocktail Therapy Celltrion Submits Investigational New Drug (IND) Application to Initiate a Global Phase III Clinical Trial Evaluating an Inhaled … Continue reading →

Drug Discovery World
NOVEMBER 1, 2023
Many drug discovery companies took the opportunity to share their latest cancer research findings. Datopotamab deruxtecan, Daiichi Sankyo and AstraZeneca The companies shared results from multiple trials of their TROP2 directed DXd antibody drug conjugate (ADC) datopotamab deruxtecan (Dato-DXd). Median PFS was 4.4
Drug Discovery World
AUGUST 9, 2023
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has granted Aleta Biotherapeutics a clinical trial authorisation (CTA) to evaluate biologic ALETA-001 in a Phase I/II clinical trial in patients with B-cell malignancies who are relapsed/refractory to CD19 CAR T cell therapy.
Pharmaceutical Technology
APRIL 14, 2023
Clinical-stage biopharmaceutical company TORL BioTherapeutics has raised $158m in a Series B financing round for advancing the development of new biologics for cancer treatment. TORL BioTherapeutics is a newly formed, US-based company focused on the development of novel antibody-based therapeutics for cancer patients. CLDN 18.2).
Drug Discovery World
JANUARY 3, 2023
A monoclonal antibody to prevent Covid-19 in vulnerable patients, AZD3152, has entered clinical trials less than 12 months after discovery. . An accelerated development programme by AstraZeneca means a new Covid-19 product could be available in the second half of 2023, subject to trial readouts and regulatory reviews. .
Drug Discovery World
AUGUST 29, 2023
Fast Track designation from the Food and Drug Administration (FDA) will accelerate the path to US submission for Alentis Therapeutics’ investigational monoclonal antibody. ALE.C04 is a first-in-class monoclonal antibody developed to specifically target exposed CLDN1 on cancer cells.

Drug Discovery World
OCTOBER 4, 2022
Currently, at least 35 glycoengineered antibodies, with their Fc fucose partially or entirely removed, have been investigated in animal models or clinical trials. It results in increased antibody-dependent cellular cytotoxicity (ADCC). How to lower the fucosylation on Fc.
Drug Discovery World
NOVEMBER 14, 2023
DDW’s Megan Thomas looks at how different diseases will benefit from the success of therapeutic antibodies. Cancer Monoclonal antibodies (mAbs) are a type of targeted drug therapy, and as such are often used to treat cancer. This is not only made clear by the fact that the global cancer mAbs market size was valued at $55.6

Drug Discovery World
MAY 25, 2023
It has been evaluated in more than 500 patients in clinical trials and demonstrated strong activity, including rapid, deep and durable responses and a favourable tolerability profile. The post Pyxis Oncology to acquire antibody therapeutics company Apexigen appeared first on Drug Discovery World (DDW).
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content