J&J files lung cancer bispecific amivantamab for FDA approval
pharmaphorum
DECEMBER 4, 2020
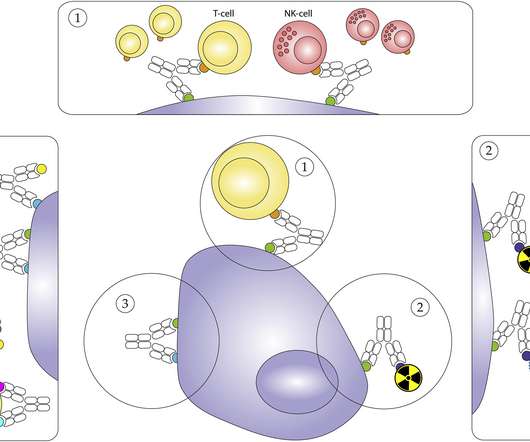
Johnson & Johnson has filed its bispecific antibody amivantamab to the FDA, hoping to muscle into the big market for drugs that are used to treat EGFR-positive non-small cell lung cancer (NSCLC). The post J&J files lung cancer bispecific amivantamab for FDA approval appeared first on.













































Let's personalize your content