Orphagen’s ACC therapy receives FDA rare pediatric disease status
Pharmaceutical Technology
JANUARY 17, 2023
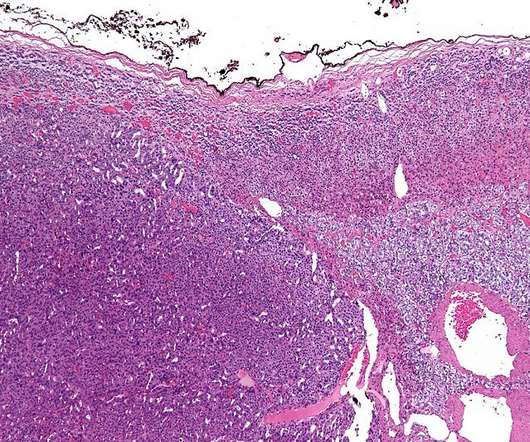
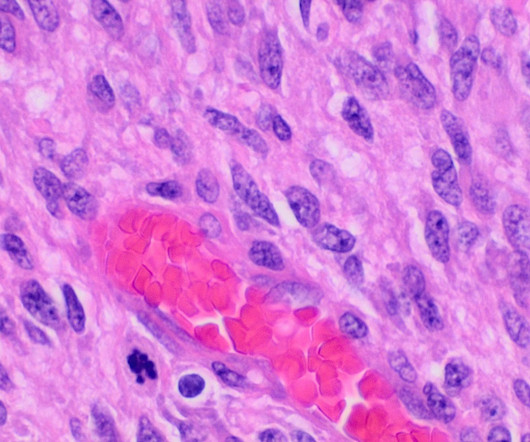
Orphagen Pharmaceuticals has received a rare pediatric disease designation (RPDD) for OR-449 from the US Food and Drug Administration (FDA) to treat paediatric adrenocortical carcinoma (ACC). In paediatric ACC, the transcription factor is commonly amplified at the gene level.















Let's personalize your content