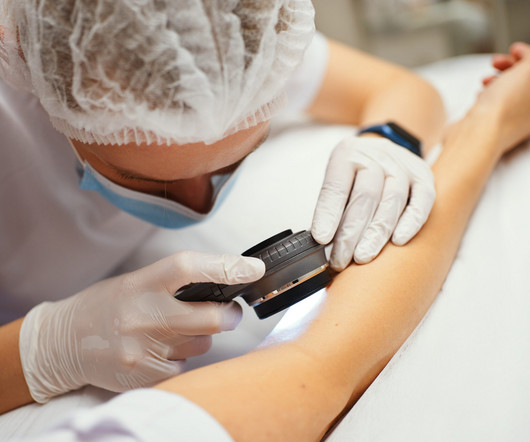
Filsuvez Topical Gel Gets FDA Approval for Epidermolysis Bullosa
XTalks
JANUARY 2, 2024
For instance, Vyjuvek , the first FDA-approved gene therapy for DEB, is priced at $24,250 per vial. a biotech company specializing in the development and commercialization of genetic medicines for rare diseases, announced FDA approval for Vyjuvek for the treatment of DEB.

















Let's personalize your content