GPCR-targeting drugs: A renewed focus on a ubiquitous group of proteins
Bio Pharma Dive
MARCH 7, 2023
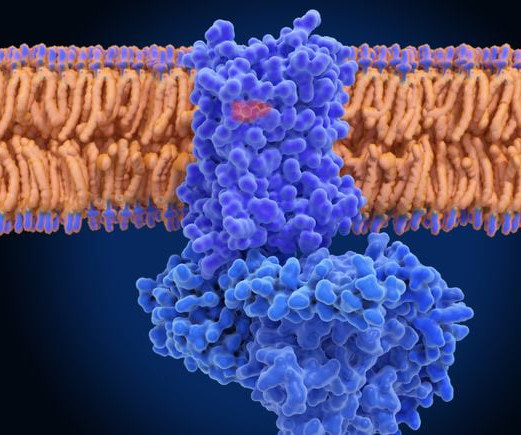
As many as a third of FDA-approved drugs target GPCRs in some fashion, with uses ranging from treating cancer to pain. But biotech startups say there is still room to develop more.


































Let's personalize your content