Bayer signals ‘significant’ layoffs in plan to shrink bureaucracy
Bio Pharma Dive
JANUARY 18, 2024
The company said it has secured agreement from labor representatives in Germany for job cuts that will shed “many managerial employees.

Bio Pharma Dive
JANUARY 18, 2024
The company said it has secured agreement from labor representatives in Germany for job cuts that will shed “many managerial employees.

Pharmaceutical Technology
JANUARY 18, 2024
The endocannabinoid inhibitor SYT-510 will be investigated in healthy patients to assess its safety, tolerability, and pharmacokinetics.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Bio Pharma Dive
JANUARY 18, 2024
Called Advanced Medicine Partners, the new company will offer specialized cell and gene therapy manufacturing services to biotech and pharma clients.

AuroBlog - Aurous Healthcare Clinical Trials blog
JANUARY 18, 2024
Conventional wisdom would have us believe the journey to a long and healthy life begins with 10,000 steps. Each and every day. For those living a more sedentary lifestyle, it’s a goal that can take some effort to maintain. We’ve also known for some time it’s also almost certainly wrong.

Bio Pharma Dive
JANUARY 18, 2024
The biotech will receive $500 million upon FDA approval of its rare disease drug acoramidis after selling 5% sales royalties to two investment firms.

AuroBlog - Aurous Healthcare Clinical Trials blog
JANUARY 18, 2024
Even as the central government is expected to present its interim budget for 2024-25 in less than three weeks, the Medical Technology Association of India (MTaI) has urged the Union Finance Ministry to reduce the customs duty on certain devices. “India’s current tariff duty structure on medical device imports is very high.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Pharmaceutical Technology
JANUARY 18, 2024
The proceeds will fund a clinical trial of Comanche’s siRNA therapy in pregnant preeclamptic women that will start later in the year.

Outsourcing Pharma
JANUARY 18, 2024
Inceptua Group and Cycle Pharmaceuticals partnership will give access to Free Goods Partnership that gets medicine to patients with extremely rare genetic conditions, which they would be unable to access otherwise.

Pharmaceutical Technology
JANUARY 18, 2024
Windtree and Lee's Pharmaceutical signed an agreement for istaroxime to treat acute heart failure and cardiogenic shock in Greater China.
Antidote
JANUARY 18, 2024
Antidote’s mission is to connect patients with medical research so medical breakthroughs can take place. A key part of our strategy is partnering with nonprofits and patient advocacy groups. We provide our clinical trial matching technology, Antidote Match, at no cost to these organizations, to help patients and caregivers discover new research opportunities and connect to clinical trials quickly and easily.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations
Pharmaceutical Technology
JANUARY 18, 2024
Ratio Therapeutics has secured $50m in a Series B financing round, a significant move aimed at advancing radiotherapies for cancer treatment.

BioPharma Reporter
JANUARY 18, 2024
The Cell and Gene Therapy Catapult (CGT Catapult), an independent organization specialising in the advancement of cell and gene therapies, has revealed that the UK has remained an attractive destination for commercial trials, with this type of clinical trial accounting for 81% of trials in 2023 and 80% in 2022.

Pharmaceutical Technology
JANUARY 18, 2024
Sun Pharma has signed a definitive merger agreement for the acquisition of all outstanding shares of Taro Pharmaceutical for $347.73m.

Outsourcing Pharma
JANUARY 18, 2024
With innovative and flexible lab and manufacturing solutions being a critical success factor in drug development and delivery, SmartLabs, was delighted to announce the raise of $48 million series C financing.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Pharmaceutical Technology
JANUARY 18, 2024
Expected to close on 22 January 2024, the German biotech’s offering follows the $1.7bn-topping deal with Moderna struck in late 2023.

XTalks
JANUARY 18, 2024
This episode of the Xtalks Food Podcast features an interview with with Eric Taipale, CTO of Sentera, a global technology leader for in-season data, analytics and insights for growers. Taipale discussed how Sentera’s technology specifically caters to the needs of growers and the agricultural community. He shared the benefits that generative AI provides to agriculture and the role that synthetic data generation has in accelerating technology adoption for the food industry.

Pharmaceutical Technology
JANUARY 18, 2024
The AI Governance Alliance has called for stronger collaboration to create a framework for AI, as industry leaders gather in Davos.

Pharmaceutical Commerce
JANUARY 18, 2024
In an interview with Pharma Commerce editor Nicholas Saraceno, Bill Roth, General Manager, Managing Partner, Blue Fin Group, discusses changes that will come with the pharma giant's new platform.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

Pharmaceutical Technology
JANUARY 18, 2024
Xenetic has entered agreements with the University of Virginia (UVA) in the US to advance the systemic DNase programme.
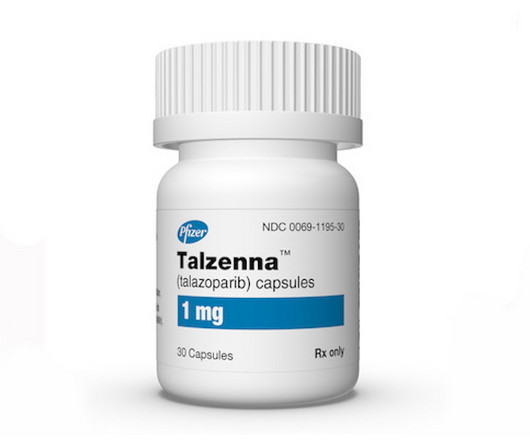
pharmaphorum
JANUARY 18, 2024
Pfizer’s PARP inhibitor Talzenna has become the first targeted treatment for HER2-negative locally advanced or metastatic breast cancer with BRCA1/2 mutations to be recommended by NICE for routine NHS use.

Fierce Pharma
JANUARY 18, 2024
Six months after England’s National Institute for Health and Care Excellence (NICE) spurned Pfizer’s breast cancer treatment Talzenna (talazoparib) because it was not cost effective, the agency has | Six months after England’s National Institute for Health and Care Excellence spurned Pfizer’s breast cancer treatment Talzenna because it was not cost effective, the agency has reversed course following the company’s offer to slash the price.

pharmaphorum
JANUARY 18, 2024
Medtech company DermaSensor gets FDA approval for a handheld device, powered by AI, that can be used to detect skin cancer at the point of care

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Fierce Pharma
JANUARY 18, 2024
In its efforts to probe high U.S. drug prices, the Senate health committee has already heard from the heads of Moderna, Eli Lilly, Novo Nordisk and Sanofi. | The Senate Committee on Health, Education, Labor, and Pensions will vote on issuing subpoenas at the end of the month after the Johnson & Johnson and Merck CEOs refused to testify at an upcoming hearing.

pharmaphorum
JANUARY 18, 2024
This page explores the significant impact of artificial intelligence (AI) on the advancement of gene therapy development within the biopharmaceutical field.

Fierce Pharma
JANUARY 18, 2024
It took 17 years for Sun Pharmaceutical and Taro Pharmaceutical to finally tie the knot. | It took 17 years for Sun Pharmaceutical and Taro Pharmaceutical to finally tie the knot. On Thursday, the companies revealed their long-awaited merger, with Sun of Mumbai, India, agreeing to purchase outstanding shares in the Israeli drugmaker for $43 each. The deal nets out to $348 million for a 48% premium on Taro's share price of $29.

pharmaphorum
JANUARY 18, 2024
AI-powered patient risk assessment company C2-Ai and the Sickle Cell Society have won an NHS contest to find solutions that can tackle health inequality

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.
Pharma Times
JANUARY 18, 2024
Green light concerns generalised myasthenia gravis therapy

pharmaphorum
JANUARY 18, 2024
pharmaphorum editor-in-chief Jonah Comstock is back from San Francisco, where he attended the annual JP Morgan Healthcare Conference. In today’s podcast episode, guest host Eloise McLennan interviews him about what he saw and heard at the show.

Fierce Pharma
JANUARY 18, 2024
Roche's TIGIT antibody has posted a positive phase 3 result that probably won't lead anywhere. In a win for Daiichi Sankyo, a U.S. agency has invalidated a Seagen patent. | Roche's TIGIT antibody has posted a positive phase 3 result that probably won't lead anywhere. In a win for Daiichi Sankyo, a U.S. agency has invalidated a Seagen patent.

pharmaphorum
JANUARY 18, 2024
India’s Sun Pharma has signed an agreement to take full control of Israeli generic drugmaker Taro Pharmaceutical Industries, taking it into private hands.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content