Apellis reports strong start for new vision loss drug
Bio Pharma Dive
MAY 5, 2023
Higher-than-expected sales of the geographic atrophy therapy Syfovre pushed shares in Apellis higher, and could add to speculation about a takeover.

Bio Pharma Dive
MAY 5, 2023
Higher-than-expected sales of the geographic atrophy therapy Syfovre pushed shares in Apellis higher, and could add to speculation about a takeover.
Pharmaceutical Technology
MAY 5, 2023
Sanofi Canada has received approval from Health Canada for its Dupixent (dupilumab injection) to treat eosinophilic esophagitis (EoE) in adults and in adolescent patients aged 12 years and above. It is being jointly developed by Sanofi and Regeneron under a global collaboration agreement. In Canada, it is commercialised by Sanofi Canada and Regeneron Canada.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Bio Pharma Dive
MAY 5, 2023
The biotech is the latest startup to capitalize on rising investor interest in targeted drugs that use radioisotopes to destroy tumors.

Pharmaceutical Technology
MAY 5, 2023
TFF Pharmaceuticals and the National Institute of Environmental Health Sciences (NIEHS) have signed an agreement for the development of dry powder formulations of high molecular weight hyaluronan (HMW-HA) for respiratory diseases. Under the collaborative research and development agreement (CRADA), respirable dry powder formulations of HMW-HA will be developed for prevention and treatment.
Bio Pharma Dive
MAY 5, 2023
The medicine, available in Europe but rejected two years ago by U.S. regulators, didn’t eliminate the need for blood transfusions among patients with myelodysplastic syndrome.

Pharmaceutical Technology
MAY 5, 2023
The European Medicines Agency’s (EMA) committee for medicinal products for human use (CHMP) has recommended full marketing authorisation (MA) for Gilead Sciences ’ Hepcludex (bulevirtide) to treat chronic hepatitis delta virus (HDV) and compensated liver disease in adult patients. In July 2020, the European Commission (EC) granted conditional marketing authorisation to bulevirtide for urgent access to HDV treatment.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Pharmaceutical Technology
MAY 5, 2023
Regeneron Pharmaceuticals has reported a 7% rise in total revenue to $3.16bn in the first quarter (Q1) of 2023 compared to $2.96bn in Q1 2022. During the first quarter, the generally accepted accounting principles (GAAP) diluted earnings per share (EPS) and non-GAAP diluted EPS stood at $7.17 and $10.09 respectively, representing declines of 17% and 12% respectively compared to Q1 2022.
Medical Xpress
MAY 5, 2023
When new COVID-19 vaccines were first administered two years ago, public health officials found an increase in cases of myocarditis, an inflammation of the heart muscle, particularly among young males who had been vaccinated with mRNA vaccines. It was unclear, however, what exactly was causing this reaction.
Pharmaceutical Technology
MAY 5, 2023
Spanish biopharmaceutical company Palobiofarma has collaborated with the Foundation for Prader Willi Research (FPWR) to develop its PBF-999 to treat Prader Willi syndrome (PWS). The company has received a new venture philanthropy award as a convertible loan from FPWR. It intends to utilise the funding from FPWR to partially fund its ongoing Phase II clinical trial of PBF-999, a dual adenosine A2a receptor antagonist/phosphodiesterase 10 (PDE-10) inhibitor.
Medical Xpress
MAY 5, 2023
There's new hope for potentially restoring vision in patients suffering from degenerative retinal disease, thanks to work by researchers at Université de Montréal.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Pharmaceutical Technology
MAY 5, 2023
The World Health Organization (WHO) announced the end of the public health emergency for Covid-19, marking the end of a declaration that had been in effect for more than three years. During a meeting on Friday, WHO officials pointed toward decreasing trends in infections, hospitalisations, and deaths stemming from Covid-19 in the international community.
Medical Xpress
MAY 5, 2023
Twelve people with persistent neurological symptoms after SARS-CoV-2 infection were intensely studied at the National Institutes of Health (NIH) and were found to have differences in their immune cell profiles and autonomic dysfunction. These data inform future studies to help explain persistent neurological symptoms in Long COVID. The findings, published in Neurology: Neuroimmunology & Neuroinflammation, may lead to better diagnoses and new treatments.

Pharmaceutical Technology
MAY 5, 2023
Switzerland-based Siegfried has purchased a 95% stake in DiNAMIQS, a biotechnology firm that focuses on the development and production of viral vectors for cell and gene therapies. Siegfried will leverage DiNAMIQS’ capabilities to commercial scale by constructing a facility that complies with good manufacturing practice standards. The facility will be situated with DiNAMIQS’ existing operations at Bio-Technopark in Zurich-Schlieren, Switzerland, and is anticipated to become operational in 2025.

Medical Xpress
MAY 5, 2023
Type I interferon, a protein that is important for the body's defense against viruses, plays a critical role in suppressing inflammation in mice infected with the schistosome parasite, Penn State immunology researchers have found. The discovery may lead to effective therapies for those suffering from schistosomiasis, second only to malaria as the most prevalent parasitic disease globally.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Pharmaceutical Technology
MAY 5, 2023
The US Food and Drug Administration (FDA) has accepted Optinose’s supplemental new drug application (sNDA) for Xhance (fluticasone propionate) to treat chronic rhinosinusitis, which is now under review. The drug-device combination product Xhance nasal spray combines a nasal anti-inflammatory drug with an exhalation delivery system (EDS). The EDS has been designed to deliver a topical anti-inflammatory high and deep into the sinuses and sino-nasal drainage tracts that are difficult to access.
XTalks
MAY 5, 2023
This week, the US Food and Drug Administration (FDA) approved the world’s first respiratory syncytial virus (RSV) vaccine. The shot, named Arexvy, is approved for adults aged 60 years and older and was developed by GlaxoSmithKline (GSK). GSK says the RSV vaccine will be available for older adults before this year’s RSV season, which usually starts before the winter months.

Pharmaceutical Technology
MAY 5, 2023
Hasten Biopharmaceutic has purchased the commercial rights for Rocephin in mainland China from Swiss healthcare firm Roche. There will now be a transition period between the two firms, following the completion of the deal earlier in 2023. Hasten also purchased Rocephin’s marketing authorisation and intellectual property including trademarks. Rocephin is a long-acting, broad spectrum cephalosporin antibiotic for parenteral use.
Medical Xpress
MAY 5, 2023
Sons of women with polycystic ovary syndrome (PCOS) are three times more likely to develop obesity, according to a study published in Cell Reports Medicine. According to the researchers from Karolinska Institutet the findings highlight a previously unknown risk of passing PCOS-related health problems across generations through the male side of a family.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
XTalks
MAY 5, 2023
In the US, food waste is a pervasive problem, with an estimated 30 to 40 percent of the country’s food supply going to waste each year. This amounts to around 133 billion pounds of food, worth an estimated $161 billion, being discarded annually. To combat this issue, companies like Full Harvest and others have emerged to sell surplus or imperfect produce that would otherwise be thrown away.
Medical Xpress
MAY 5, 2023
The immune system has a biological telecommunications system—small proteins known as interleukins that send signals among the leukocyte white blood cells to control their defense against infections or nascent cancer. Interleukin-6, or IL-6, is one of these key mediators of inflammation, and it can, as needed, provoke the immune system into attack against pathogens.

Intouch Solutions
MAY 5, 2023
AI Technology and the Impact to Paid Search Google has dominated paid search since the early 2010s, but Microsoft has shaken things up with ChatGPT (and Google has countered with Bard). These AI tools will change how users find information, and impact the level of engagement advertisers can expect from users. Let’s look at what these technologies are, where they’re going, and what advertising spaces they’ll disrupt.
Medical Xpress
MAY 5, 2023
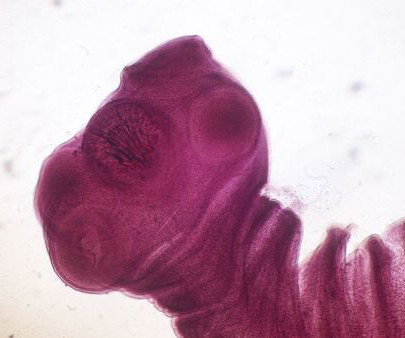
The pork tapeworm (Taenia solium) can grow to be more than 20 feet long inside the human intestine, where it attaches to the intestinal wall using the 4 suckers on its scolex, which is only 1 millimeter in diameter. The scolex looks somewhat like a head, but the organism has no mouth or digestive tract and simply absorbs nutrients through its body covering.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Pharma Times
MAY 5, 2023
Systems will support millions of patients with chronic respiratory conditions throughout Europe
Medical Xpress
MAY 5, 2023
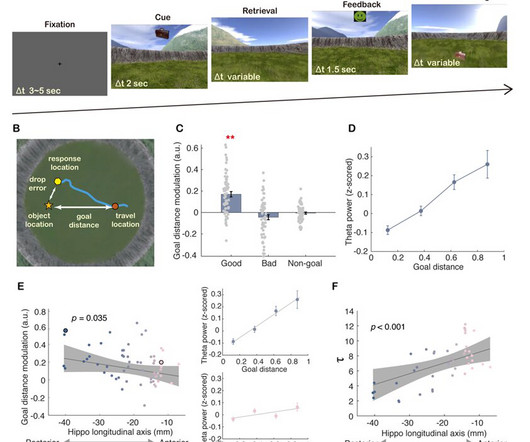
A research team led by Dr. Wang Liang from the Institute of Psychology of the Chinese Academy of Sciences has found that right hippocampal theta power hierarchically encodes the distance between the current position and a goal destination.

Cloudbyz
MAY 5, 2023
Executive Summary Risk-Based Monitoring (RBM) is an approach to clinical trial monitoring that optimizes the use of resources by focusing on trial risks, with the goal of ensuring patient safety, data quality, and regulatory compliance. This white paper provides an in-depth analysis of RBM in clinical trial operations, discussing its definition, methodology, benefits, challenges, and best practices for implementation.
Medical Xpress
MAY 5, 2023
Football players who have had repetitive head trauma and concussion are at heightened risk for chronic traumatic encephalopathy (CTE), an irreversible condition that leads to dementia. But not every case of cognitive decline means CTE, as illustrated by a new case study published by researchers from Mass General Brigham in Current Sports Medicine Reports.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

pharmaphorum
MAY 5, 2023
Digitisation is fundamental to unlocking a patient-centric industry Mike.

Medical Xpress
MAY 5, 2023
Overall, 18.7 percent of U.S. adults reported currently using any tobacco product in 2021, with most reporting combustible tobacco product use, according to research published in the May 5 issue of the U.S. Centers for Disease Control and Prevention Morbidity and Mortality Weekly Report.
BioPharma Reporter
MAY 5, 2023
Mekonos, a biotech platform developing cell therapies, has announced a new research collaboration with bit.bio, a synthetic biology company providing human cells for research, drug discovery and cell therapy.

Medical Xpress
MAY 5, 2023
For individuals with depression, the most common causes of hospitalization are endocrine, musculoskeletal, and vascular diseases, according to a study published online May 3 in JAMA Psychiatry.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content