€30m raised for first-in-class in vivo cell reprogramming platform
Drug Discovery World
MARCH 19, 2024
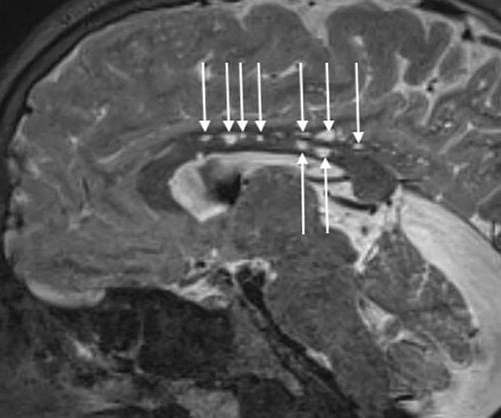
AT-108 is a first-in-class, off-the-shelf gene therapy that directly reprogrammes tumour cells into antigen-presenting dendritic cells, ultimately leading to a personalised anti-tumour immune response. We believe this breakthrough strategy will give rise to the next generation of cell therapies.









































Let's personalize your content