Positive in vitro results for Imutex’s FLU-v
Pharma Times
OCTOBER 1, 2022
Data further supports the continued development of FLU-v as a broad-spectrum influenza vaccine
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharma Times
OCTOBER 1, 2022
Data further supports the continued development of FLU-v as a broad-spectrum influenza vaccine

Pharmaceutical Technology
MAY 12, 2023
BioNTech has ended its research collaboration with Matinas after its oral mRNA vaccine failed to demonstrate preclinical activity. Whilst the formulation had been successful in vitro , the oral administration in mice did not elicit activity. Matinas announced the news in a May 10 statement.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

BioPharma Reporter
JANUARY 28, 2021
In vitro studies found the Pfizer/BioNTech COVID-19 vaccine elicits antibodies against both the UK and South African virus strains.

BioPharma Reporter
JANUARY 25, 2021
An in vitro study shows Moderna's existing mRNA COVID-19 vaccine provides protection against strains that have emerged from South Africa and the UK. But it does suggest reduced protection against the South Africa strain, and so the company is also developing a new booster designed for maximum efficacy against this variant.

Pharmaceutical Technology
SEPTEMBER 19, 2022
AstraZeneca Vaccines and Immune Therapies executive vice-president Iskra Reic said: “Evusheld has already made an important difference around the world helping prevent Covid-19 infections in vulnerable populations who can’t mount an adequate response to Covid-19 vaccination. 5 variant, the company noted. 5 variant, the company noted.

Scienmag
JANUARY 25, 2022
Vaccination against COVID-19 did not affect fertility outcomes in patients undergoing in-vitro fertilization (IVF), a new study has found. The findings, which were published in Obstetrics & Gynecology (the Green Journal), add to the growing body of evidence providing reassurance that COVID-19 vaccination does not affect fertility.

Scienmag
DECEMBER 11, 2021
Published today, one of the earliest, peer-reviewed studies looking into the Omicron variant of COVID-19 suggests that people previously infected with COVID, and those vaccinated, will have some, “stronger than basic” defence against this new strain of concern.
pharmaphorum
MARCH 4, 2021
UK chancellor Rishi Sunak has announced a budget loaded with initiatives designed to kick-start the UK’s economy as it recovers from the coronavirus pandemic, with vaccine development, pharma and life sciences playing a key role. A scheme providing £500 payments to people self-isolating has been extended in England until the summer.

XTalks
MARCH 1, 2021
(NYSE: PFE ) and BioNTech SE (Nasdaq: BNTX ) have announced that they are beginning a trial to evaluate the safety and efficacy of a third booster dose for their COVID-19 vaccine (BNT162b2), as well as new vaccine variants. Related: Could Pfizer and BioNTech’s COVID-19 Vaccine be Stored at Standard Freezer Temperatures?
Drug Discovery World
MARCH 22, 2023
Download this eBook to learn more about process development and manufacturing in the mRNA space to enable successful scale-ups of mRNA production for therapeutic applications.
Pharmaceutical Technology
JUNE 1, 2023
The vaccine is self-administered through an intranasal spray, and is based on the company’s GlycoTarge platform. In April 2020, Pneumagen announced positive anti-viral activity results from three in vitro studies into preventing coronavirus infections, but the program has not yet been studied in humans. million ($4.75
XTalks
JANUARY 27, 2021
COVID-19 vaccine maker Moderna announced this week that results from a study show that the shot is effective against some of the new circulating variants of SARS-CoV-2. In a news release from Moderna, the company reported that the administration of its COVID-19 vaccine induced antibody titers against both variants.
XTalks
JANUARY 15, 2021
Ortho Clinical Diagnostics received emergency use authorization (EUA) from the US Food and Drug Administration (FDA) for its VITROS SARS-CoV-2 antigen test for detection of active COVID-19 infection. Related: COVID-19 Vaccine and Drug Development Coverage. active virus).
Pharmaceutical Technology
MAY 24, 2023
Morphogenesis’s technologies include Immune Fx (IFx) personalised cancer vaccines and tumour microenvironment (TME) modulators. Morphogenesis’ lead personalised cancer vaccine, IFx-Hu2.0, Morphogenesis’ lead personalised cancer vaccine, IFx-Hu2.0, The company is also advancing its mRNA vaccine, IFx-Hu3.0,

Drug Discovery World
MARCH 22, 2023
Topics covered in the eBook include: Expert views on mRNA development and manufacturing Advantages of mRNA processes compared to traditional vaccine manufacturing Best practices for scale up of in vitro transcription for mRNA production The future of the mRNA therapeutics field, and more Download now.

Drug Discovery World
MARCH 14, 2023
The success of messenger RNA or mRNA vaccines in the fight against the global SARS-CoV-2 crisis brings a new era of vaccine development. In vitro transcription, or IVT, is a complex reaction, requiring key components like an RNA polymerase, nucleotides, and a DNA template to make each mRNA.
The Pharma Data
AUGUST 15, 2021
Even as several safe and effective COVID-19 vaccines are being administered to people worldwide, scientists are still hard at work developing different vaccine strategies that could provide even stronger or longer-lasting immunity against SARS-CoV-2 and its variants.

Drug Discovery World
OCTOBER 2, 2023
The Nobel Assembly at Karolinska Institutet has awarded the 2023 Nobel Prize in Physiology or Medicine jointly to Katalin Karikó and Drew Weissman for their discoveries that enabled the development of mRNA vaccines against Covid-19. The discovery had profound significance for using mRNA as therapy. Image credit: Ill.

XTalks
APRIL 2, 2021
Pfizer released new COVID-19 vaccine trial results this week from its ongoing clinical studies, which include data showing that its COVID-19 mRNA vaccine is 100 percent effective in children between the ages of 12 and 15 and has a 91 percent efficacy against variants with protection lasting at least six months.

XTalks
MARCH 27, 2024
In vitro diagnostics, or IVD, is a field where precision, reliability and robust partnerships propel advancement. Dr. Dhiman has 24 years of clinical research experience in the immunogenetics of vaccines and infectious disease diagnostics. She did her clinical microbiology fellowship at Mayo Clinic in Rochester, Minnesota.

The Pharma Data
SEPTEMBER 17, 2020
This is the focus of vaccines in development and convalescent plasma therapy. The majority of current candidate vaccines aim to induce an antibody response against the spike protein. “As The test targets antibodies against the spike protein. Food and Drug Administration (FDA). said Thomas Schinecker, CEO Roche Diagnostics.

The Pharma Data
MAY 17, 2021
The European Commission’s Medical Device Coordination Group (MDCG) sent a notice to in vitro diagnostic (IVD) makers and their authorized representatives reminding them of their obligation to assess the impact of COVID-19 variants on the performance of their tests. . Posted 17 May 2021 | By Michael Mezher .

pharmaphorum
JANUARY 17, 2023
Research into mRNA dates back to the 1970s, but with the approval of both Moderna and BioNTech/Pfizer’s vaccines, this approach has finally been validated. At the end of last year, the UK government entered into a partnership with the company, which saw Moderna agree to provide 250 million vaccine doses per year over the course of a decade.

pharmaphorum
JANUARY 22, 2021
So far, neither variant seems to be associated with more severe COVID-19 symptoms, although there has been some preliminary research suggesting the SA strain could allow reinfection with SARS-CoV-2, and also be less susceptible to vaccines. The UK strain – known as B.1.1.7

XTalks
JUNE 27, 2022
CerTest Biotec specializes in the development and manufacturing of in vitro diagnostic medical devices and has expertise in the detection of human diseases. As such, the smallpox vaccine can be used to protect against monkeypox — according to the WHO, the vaccine is 85 percent effective against monkeypox infection.

Scienmag
SEPTEMBER 18, 2020
Antibody-like proteins that capture and neutralize SARS-CoV-2 Scientists have used a new high-speed, in vitro selection method to isolate 9 antibody-like proteins (ALPs) that bind to the SARS-CoV-2 virus – 4 of which also exhibited neutralizing activity – within 4 days, according to a new study.

pharmaphorum
DECEMBER 16, 2020
Soon the ABPI and the entire industry were facing the ripple effects of COVID-19, including a significant slowdown in non-COVID clinical research, which Torbett says was challenging for the industry to deal with. “We’ve We’ve also seen a very mixed picture in the market for pharmaceuticals,” he says. Strength in the second wave.

The Pharma Data
JANUARY 11, 2021
Testing Therapies, Antivirals and Vaccines. By the end of January, Johnson & Johnson is expected to deliver preliminary trial results for its one-dose COVID-19 vaccine candidate. . One of the open questions about the various vaccines against COVID-19 has been how long the protection offered will last. Diagnostics.
Roots Analysis
AUGUST 15, 2023
Overview of Cell Free Systems Cell-free systems are in-vitro platforms which allow occurrence of biochemical reactions in the absence of living cells. These expression systems utilize bio machinery harvested from the lysate of disrupted cells for the manufacturing of a wide array of macromolecular and small molecule products.
Drug Discovery World
FEBRUARY 16, 2024
DDW recently hosted an online event, sponsored by Taconic Biosciences, which outlined areas of advancement and opportunity in the cancer research market. Megan Thomas summarises the key points examined by the participating experts. They make the most of their environment to gain a survival advantage, and there are a lot of elements involved.

The Pharma Data
MARCH 25, 2022
People not adequately protected by a COVID-19 vaccine may particularly benefit from pre-exposure prophylaxis with Evusheld. People not adequately protected by a COVID-19 vaccine may particularly benefit from pre-exposure prophylaxis with Evusheld. 2-4 Evusheld was generally well-tolerated in the trial.(2-4).

XTalks
MARCH 2, 2021
As the US Food and Drug Administration (FDA) handed out an emergency use authorization (EUA) to Janssen’s COVID-19 vaccine over the weekend — making it the third authorized COVID-19 vaccine in the US — many people became laser-focused on the 66 percent average efficacy of the single-dose shot.

Drug Discovery World
MAY 25, 2023
Following the Scottish Research and Drug Development Forum meeting, DDW’s Diana Spencer provides a snapshot of drug discovery in Scotland. With over 150 pharma services/supply companies and 19 universities, Scotland is one of the most advanced life science sectors in the UK and the world. There are various other phenomics projects currently ongoing.
Delveinsight
FEBRUARY 23, 2021
The company develops transformative biologics including vaccines and therapeutics for the world’s most debilitating diseases. The company plans to use the capital to expand its pipeline of protein-based vaccines and biologic cancer therapies utilizing its innovative and proprietary Trimer-Tag technology platform.
pharmaphorum
NOVEMBER 4, 2020
However, in vitro or binding-based efficacy insight needs to be confirmed by reliable clinical evidence, as shown by the outcomes of COVID-19 trials with remdesivir and hydroxychloroquine. million worldwide and continues to increase despite the global public health measures that have been put in place.

The Pharma Data
MARCH 29, 2022
Evusheld has the potential to provide long-lasting protection against COVID-19 for a broad population of individuals, including those who aren’t adequately protected by a COVID-19 vaccine, as well as those at increased risk of exposure.”. 2 subvariant, now the dominant strain in Europe. 1-3 Evusheld was generally well-tolerated in the trial.
XTalks
NOVEMBER 15, 2023
France-based biotech Valneva has won approval from the US Food and Drug Administration (FDA) for its chikungunya vaccine Ixchiq for the prevention of infection from the chikungunya virus. The vaccine is approved for adults 18 years of age and older who are at increased risk of exposure to the virus.

pharmaphorum
AUGUST 4, 2022
If not internally, such specialist training programmes can be outsourced to third-party organisations, whether that involves pharmaceuticals, in vitro diagnostics (IVDs) or QP training. This has resulted in staff and talent shortages, meaning that industries have been forced to respond quickly to stay afloat. Harness technology.

Pfizer
AUGUST 15, 2022
I am grateful to have received four doses of the Pfizer-BioNTech vaccine and I am feeling well while experiencing very mild symptoms. I am incredibly grateful for the tireless efforts of my Pfizer colleagues who worked to make vaccines and treatments available for me and people around the world.

The Pharma Data
OCTOBER 14, 2021
Sanofi’s investigational oral Bruton’s tyrosine kinase (BTK) inhibitor, tolebrutinib, demonstrated favorable one-year tolerability in a Phase 2b long-term extension study (LTS) in patients with relapsing forms of multiple sclerosis (RMS). Therapeutic Area Head, Neurology Development at Sanofi.

The Pharma Data
JUNE 17, 2022
“The broadly neutralizing antibodies we described were more efficient in vitro than many anti–SARS-CoV-2 monoclonals previously approved by the FDA for treatment or prevention. A modified version of the Cv2.1169 antibody was also effective at treating SARS-CoV-2 infection in mice and hamsters.
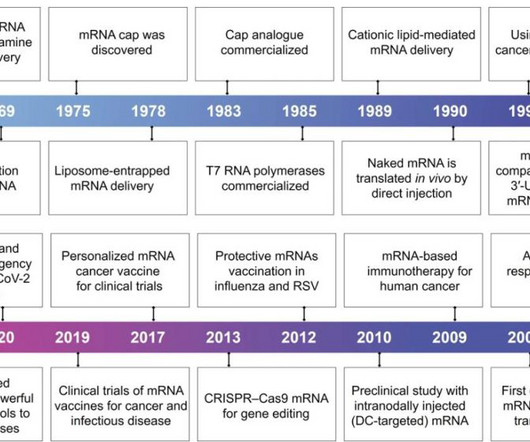
Pharma Mirror
APRIL 21, 2024
With the rapid development of biotechnology and molecular medicine, the introduction of mRNA as a vaccine or therapeutic agent enables the production of almost any desired functional protein/peptide within the human body.

Roots Analysis
JANUARY 14, 2024
With the increased interest and gradual shift of investment from small molecule drugs to biologics and the establishment of several biologics manufacturing companies / biologics CMOs, more than 250 biologic therapies and vaccines have been developed, globally. They are different from small molecules in terms of their size and complexity.
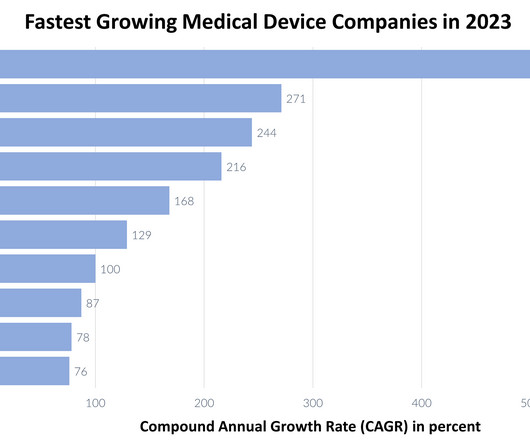
XTalks
DECEMBER 11, 2023
Medical device companies continue to maintain a crucial role in enhancing patient care and diagnostic precision in 2023. Let’s examine the list of the top ten fastest growing medical device companies in 2023, ranked by their compound annual growth rate (CAGR). In 2021, Axonics achieved an impressive revenue of $180.3
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content