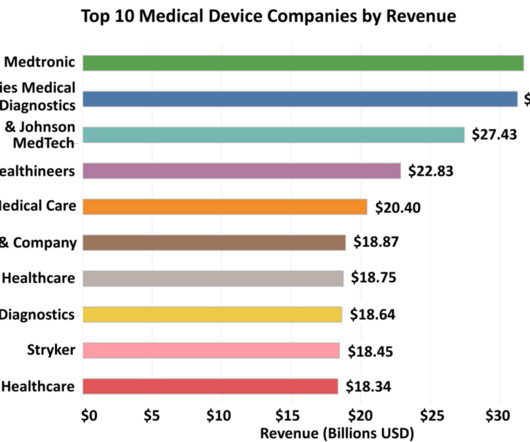
Evotec extends collaboration with Bristol Myers Squibb
Pharmaceutical Technology
MARCH 29, 2023
In September 2021, Bristol Myers Squibb in-licensed the first programme, EVT8683, after the successful filing of an investigative new drug (IND) application with the Food and Drug Administration (FDA). This programme has now proceeded into a Phase I clinical trial.























Let's personalize your content