Cipla and Glenmark initiate recall of certain products in the US
Pharmaceutical Technology
MAY 6, 2024
Cipla and Glenmark Pharmaceuticals are recalling specific products from the US market due to distinct manufacturing issues.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Pharmaceutical Technology
MAY 6, 2024
Cipla and Glenmark Pharmaceuticals are recalling specific products from the US market due to distinct manufacturing issues.

Pharmaceutical Technology
MARCH 13, 2024
As the demand for medical devices continues to rise, manufacturers are increasingly turning to automation during the assembly process to get their products to market faster. But how can the risks involved be mitigated?
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
JULY 15, 2022
Insulin prices made the headlines again as California governor Gavin Newsom announced plans on 7 July for the state to manufacture low-cost insulin. The state plans to work directly with a contract manufacturing organization (CMO) to manufacture low-cost insulin. In-house manufacturing the norm.
Pharmaceutical Technology
APRIL 13, 2023
The expanded licence will include the development of vaccines and therapeutic proteins beyond Covid-19 for human and animal health markets in Africa. The initial agreement between Dyadic and Rubic involved discovery, development, manufacturing, and distribution of Covid-19 vaccines by transferring and licensing of C1 platform technology.

Pharmaceutical Technology
MARCH 10, 2023
On 10 March, the National Health Service Blood and Transplant (NHSBT) opened a new Clinical Biotechnology Centre (CBC) with the aim of improving the UK’s ability to develop and manufacture cell and gene therapies. This is important due to the UK’s currently limited short-scale manufacturing capacity.

Bio Pharma Dive
MAY 22, 2023
When pharma manufacturers are racing to bring a new product to market, catastrophic printing errors and mislabeled materials can have a significant impact on timelines and budgets.

Pharmaceutical Technology
JUNE 12, 2023
Lotus Pharmaceuticals and Teraju Pharma have entered a strategic partnership to market Lotus’s products in a range of therapeutic areas in Malaysia. Teraju Pharma will be responsible for marketing and promoting the products using its relationships and infrastructure.

Pharmaceutical Technology
SEPTEMBER 8, 2022
Pharmaceutical solid dose manufacturing companies in contract marketing segment have gained a vital position in pharmaceutical formulations. Find the leading pharmaceutical solid dose manufacturing companies in contract marketing. Growth of oral solid dosage contract manufacturing in the pharmaceutical industry.

Bio Pharma Dive
DECEMBER 6, 2023
The funds are meant to boost Fujifilm’s capacity to manufacture cell therapies, a market it expects to grow substantially in the coming years.

Roots Analysis
APRIL 15, 2024
Further, the manufacturing of biologics fill finish is a highly complex and cost-intensive process. Notable examples of fill finish manufacturing companies equipped with SA25 aseptic filling workstation include ( in alphabetical order ) Emergent Bioservices, PCI Pharma Services, Singota Solutions and WuXi Biologics.

AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 2, 2022
Care-Keralam, the Ayush cluster in Kerala, will shortly fine-tune its strategies to build ‘Kerala Brand of Ayurveda Products’ for international marketing and to increase the quantity of exports of traditional and herbal drugs from Kerala to global markets.
Pharmaceutical Technology
NOVEMBER 22, 2022
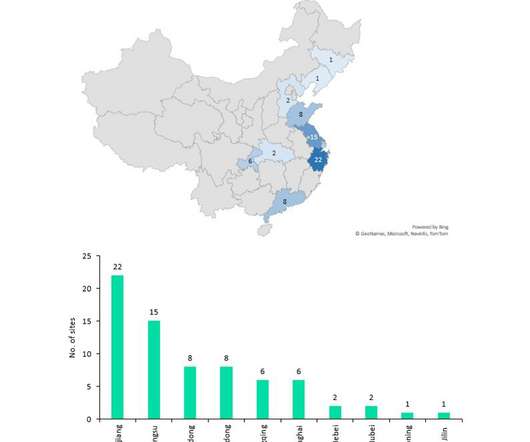
Despite China producing a significant proportion of the world’s API supply (mostly small molecule), it manufactures relatively few biosimilar and innovator drugs and no cell and gene therapies for the western markets of Europe and the US despite investments and an increasing number of startups to improve innovative manufacture.

Pharmaceutical Technology
NOVEMBER 1, 2022
FEATURED COMPANIES COMMENDED : • Innovation • Product Launch • Safety VIEW PROFILE COMMENDED : • Business Expansion • Innovation • Investments VIEW PROFILE COMMENDED: • Social VIEW PROFILE. What are Product Launches? The Product Launches category recognises companies that have launched notable new products or services into the market.
Roots Analysis
AUGUST 16, 2023
The success of COVID-19 vaccines paved the path for mRNA-based drug products. Safety, efficacy and rapid production of mRNA drives the interest in this domain. Concluding Remarks Currently, dozens of preclinical and clinical reports demonstrating the efficacy of these platforms have been published in the last two years alone.

Pharmaceutical Technology
MAY 23, 2023
Croda Pharma has entered a strategic collaboration deal with Botanical Solution Inc (BSI) to expedite the production of sustainable pharmaceutical-grade vaccine adjuvant QS-21. Croda Pharma and BSI will share their knowledge to bring QS-21 to market from quillaja Saponaria plant cell culture, which is made from the soap bark tree.

Bio Pharma Dive
JULY 11, 2022
Gavin Newsom announced a $100 million budget to create a production facility and to develop affordable insulin products, saying the medicines’ high cost “epitomizes market failures.”
Pharmaceutical Technology
APRIL 4, 2023
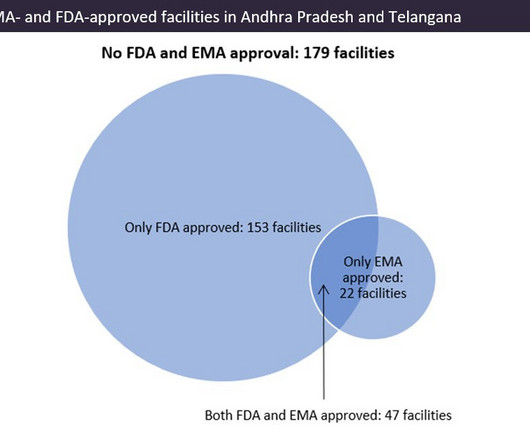
Indian pharma manufacturing continues to be the backbone of drug supplies worldwide, and GlobalData analysis suggests US overreliance on the country for generic drug supply. Pharma manufacturing facilities in Andhra Pradesh and Telangana accounted for 22.5% © GlobalData. © GlobalData. ©GlobalData.

XTalks
SEPTEMBER 1, 2023
GlaxoSmithKline (GSK) has been pumping large sums of cash into expanding its vaccine manufacturing operations and today, it announced an investment of €250 ($272 million) into building a new unit for freeze-drying vaccines at its Wavre campus in Belgium. Both vaccines will be manufactured at the new unit in Belgium. billion) in 2022.
Pharmaceutical Technology
JUNE 23, 2022
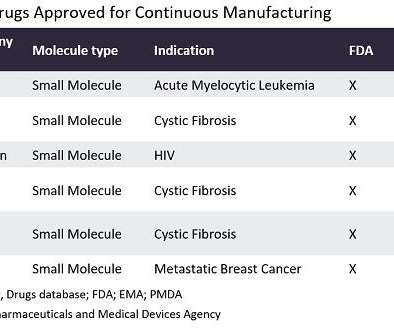
In the last year, well-known contract manufacturing organisations (CMOs) such as Agilent Technologies (Santa Clara, California) and Abzena (Cambridge, UK) have expanded their continuous manufacturing capabilities. US Congress is also making substantial investments to improve the continuous manufacturing of pharmaceuticals.
Roots Analysis
MARCH 26, 2024
However, in order to deal with this situation, the medical agencies came up with the option of advanced technologies, such as continuous manufacturing technology. In fact, drug manufacturers are moving faster than ever to cope up with the drug shortages. Specifically, in the pharmaceutical industry, the standstill days are over.

pharmaphorum
AUGUST 12, 2022
The COVID-19 pandemic led to skyrocketing demand for some products while simultaneously constraining supplies due to logistical breakdowns and protectionist practices. The logic of outsourcing production to other countries, primarily China, suddenly looked dubious as Western countries struggled to import the drugs they needed.
Drug Discovery World
MAY 7, 2024
Where the bottlenecks of achieving higher cell density and higher titers were once thought to be issues of the past, hurdles have now shifted to achieving speed and scale in mAb production without sacrificing quality. DC: Antibodies have and remain a large product class within the biopharmaceutical market.
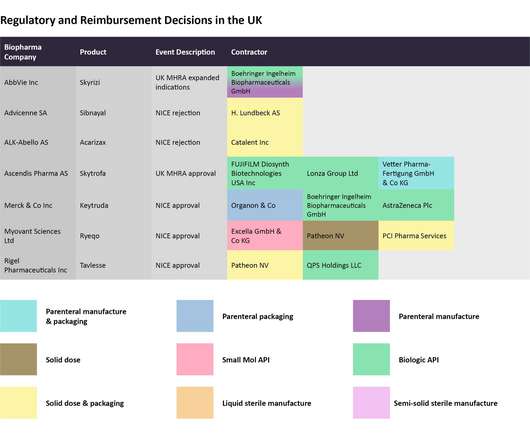
Pharmaceutical Technology
DECEMBER 12, 2022
In this last 2022 edition of the series, which started in June , Pharmaceutical Technology is tracking major trial announcements and decisions by regulators and reimbursement agencies that have occurred since mid-October, as well as their potential impact on manufacturing plans. Iomab-B met the durable complete remission endpoint.

XTalks
JUNE 12, 2023
Bristol Myers Squibb (BMS), a multinational pharmaceutical company based in New York City, has announced that its new state-of-the-art cell therapy manufacturing facility in Devens, Massachusetts, has received approval for commercial production from the US Food and Drug Administration (FDA).

Pharmaceutical Technology
DECEMBER 14, 2022
Harrow has signed a binding agreement to acquire exclusive US commercial rights to five ophthalmic products of Novartis. These products, namely, Ilevro, Vigamox, Maxidex, Nevanac and Triesence, have received approval from the Food and Drug Administration (FDA). Outside of the US region, Novartis will have sole rights to the products.
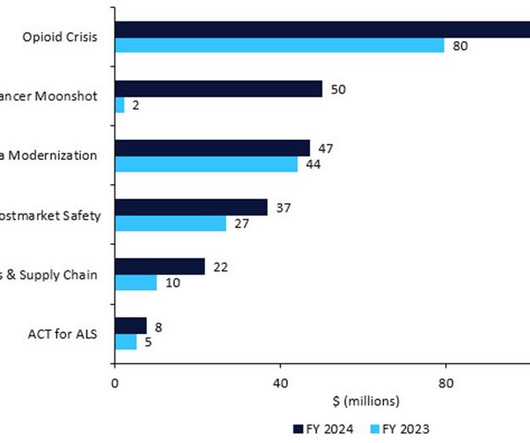
Pharmaceutical Technology
MARCH 30, 2023
The US FDA has revealed its detailed budget proposal for FY2024, which would require pharma companies to name their active pharmaceutical ingredient (API) suppliers, restart President Biden’s Cancer Moonshot, inject cash into amyotrophic lateral sclerosis (ALS) research, and enforce stricter rules around manufacturing, recalls, and shortages.
BioTech 365
JUNE 17, 2022
The global antibodies contract manufacturing market is expected to reach from USD 13.4 Rise in demand for the production of therapeutic antibodies … Continue reading → billion in 2022 to USD 35.9 billion by 2030, registering a CAGR of 13.1% from 2022 to 2030.
Pharmaceutical Technology
NOVEMBER 21, 2022
RVAC Medicines has signed a master research partnership agreement with the Agency for Science, Technology and Research (A*STAR) for analysing and developing solutions to build messenger ribonucleic acid (mRNA) manufacturing and analytics expertise in Singapore. Topic sponsors are not involved in the creation of editorial content.

AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 7, 2022
The Ayush cluster in Kerala, Care-Keralam Ayurvedic Resource Centre at Koratty in Thrissur, is planning to adopt a multi-pronged strategy to support the small and medium ayurvedic drug manufacturing enterprises (SMEs) by focusing on their production growth and marketing of products.

Pharmaceutical Technology
SEPTEMBER 15, 2022
Contract marketing companies offer various marketing services that connect suitable strategies with customers at different points in the product lifecycle, helping to increase the customer base and thus revenue. Finding the top API chemical companies in contract marketing.

STAT News
NOVEMBER 16, 2022
WASHINGTON – The Food and Drug Administration is formally warning multiple companies they must stop selling vapes and other products marketed to young people, including two that STAT highlighted in recent reporting on the industry. were being sold just a few miles from the FDA’s campus.

XTalks
JUNE 14, 2023
The online marketplace PartnerSlate has made waves in the food and beverage contract manufacturing industry, recently securing a $4 million investment to further its mission of connecting food brands and contract manufacturers. This is where San Francisco-based PartnerSlate comes in.

Roots Analysis
MARCH 9, 2022
In fact, over 8,000 biological pharmaceutical products are currently under clinical investigation worldwide. . Biopharmaceutical Excipient Manufacturing Market. Biopharmaceutical Excipient Manufacturing Market. Diverse Landscape of Biopharmaceutical Excipient Manufacturers. Company Competitiveness Analysis.

FDA Law Blog
MARCH 4, 2024
Livornese — On February 6, 2024, FDA issued a draft guidance titled Notifying FDA of a Discontinuance or Interruption in Manufacturing of Finished Products or Active Pharmaceutical Ingredients Under Section 506C of the FD&C Act.

Pharmaceutical Technology
JUNE 8, 2023
The European Commission (EC) has granted marketing authorisation for GSK’s respiratory syncytial virus (RSV) vaccine, Arexvy, for adults aged 60 years and above. The EC approval follows a positive opinion from the European Medicines Agency’s committee on medicinal products for human use (CHMP) in April 2023.

BioPharma Reporter
SEPTEMBER 19, 2023
The contract development and manufacturing organization (CDMO) Fujifilm Diosynth Biotechnologies has completed a UK manufacturing facility for the production of advanced therapies including viral gene therapies, oncolytic viruses and viral vaccines for use in clinical trials.

Pharmaceutical Technology
JANUARY 20, 2023
Bancel explained that this deal will “shrink the time to get from designing the sequence to having products ready by two more weeks”. Increasing manufacturing capacity for pandemic prevention “The other piece we are working on is … how do we build manufacturing capacity around the world?”, said Bancel.
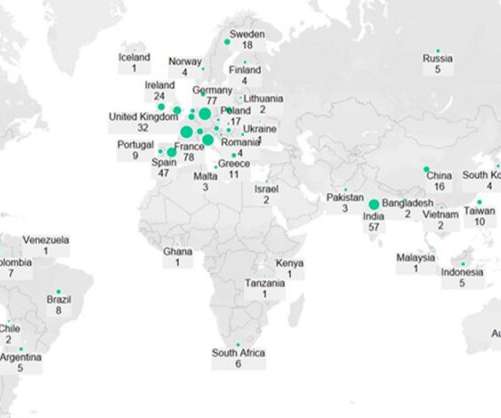
Pharmaceutical Technology
JULY 29, 2022
Pfizer (New York, New York) and Moderna’s (Cambridge, Massachusetts) Omicron-specific Covid-19 vaccines are in late-stage development, have demonstrated good efficacy against the variant and will likely be available to the public later this year to provide an additional booster and increase demand for injectable manufacturing.

Pharmaceutical Technology
NOVEMBER 30, 2022
However, efficiently manufacturing the drug represents another barrier to cross before realizing the full revenue potential then successfully. Each month, Pharmaceutical Technology takes a look at recent decisions taken by regulatory and reimbursement agencies and identifies the key manufacturing players that can be impacted by them.

XTalks
MAY 3, 2024
To combat food poisoning, US agriculture officials introduced a final rule last Friday, mandating significant reductions in Salmonella bacteria in specific chicken products. Starting in 2025, high levels of Salmonella in frozen breaded and stuffed chicken products will classify them as adulterated. How Common Is Salmonella in Chicken?

Pharmaceutical Technology
SEPTEMBER 20, 2022
The production of commercial dose non-sterile products such as tablets, capsules, ointments, creams, and powders is rising due to their growing global demand, attributed to increasing demand for anti-ageing products, hereditary factors, genetic mutations, exposure to harmful radiation, and rising geriatric population.

Roots Analysis
AUGUST 9, 2023
It is worth mentioning that in recent years, several healthcare stakeholders have actively undertaken initiatives to incorporate a manufacturing execution system (MES) into their production lines, with an aim to connect, monitor and control the complex manufacturing process.

XTalks
SEPTEMBER 19, 2023
The US Food and Drug Administration (FDA) has issued warning letters to eight companies manufacturing or marketing unapproved eye products that violate federal law. The FDA said the warning letters are part of the agency’s ongoing effort to protect Americans from potentially harmful ophthalmic products.
Pharmaceutical Technology
MAY 26, 2023
The immuno-oncology market continues to evolve as treatments establish their presence across different types of cancer, having seen approvals in multiple indications in the past decade. There are currently over 700 immuno-oncology products in Phase I-III of development in the eight major markets, said Chalk.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content