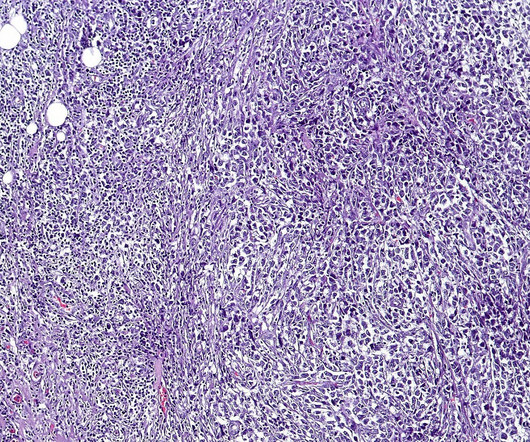
Multiple Myeloma Drug Tecvayli Becomes First FDA-Approved Bispecific T Cell Antibody
XTalks
OCTOBER 27, 2022
The Janssen Pharmaceutical Companies of Johnson & Johnson announced that the US Food and Drug Administration (FDA) approved Tecvayli (teclistamab-cqyv) for the treatment of relapsed or refractory multiple myeloma. Tecvayli is a first-in-class, bispecific T-cell engager antibody administered as a subcutaneous treatment.











































Let's personalize your content