FDA $7bn plans for 2024: disclose contract manufacturers, restart Cancer Moonshot
Pharmaceutical Technology
MARCH 30, 2023
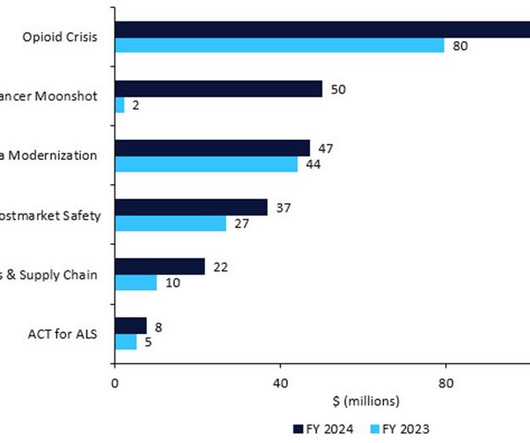
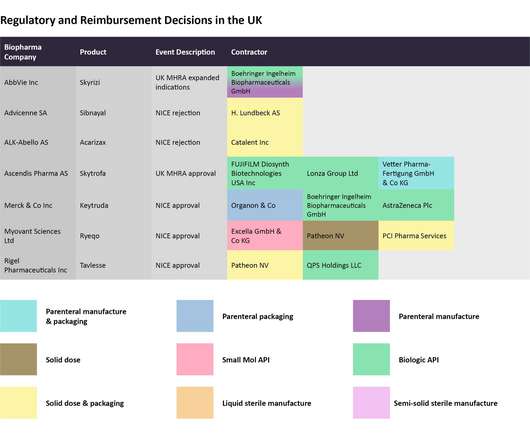
Getting generics to market faster: Patent exclusivity for first-line generics will be revised to encourage them to launch swiftly after approval, and to ease the way for second-line applications. Postmarket Safety Collaborative: +$10.1m for a total of $36.8m to enhance the safety surveillance of adverse events. Source: FDA © GlobalData



































Let's personalize your content