Almirall and Microsoft partner for dermatological drug development
Pharmaceutical Technology
FEBRUARY 1, 2024
Almirall has entered a strategic collaboration with Microsoft to steer innovation and digital transformation in dermatology.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
FEBRUARY 1, 2024
Almirall has entered a strategic collaboration with Microsoft to steer innovation and digital transformation in dermatology.

Pharmaceutical Technology
NOVEMBER 15, 2023
Almirall has entered into a collaboration with Absci for the development and commercialisation of new treatments for dermatological ailments.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Drug Discovery World
FEBRUARY 15, 2024
Alys believes that emerging indications such as atopic dermatitis, vitiligo, alopecia areata and chronic spontaneous urticaria, will see the next breakthroughs in dermatological advancements. The post $100M financing to advance immuno-dermatology pipeline appeared first on Drug Discovery World (DDW).

pharmaphorum
OCTOBER 9, 2023
Champion Innovation at the 7th Dermatology Drug Development Summit Mike.Hammerton Mon, 09/10/2023 - 14:39 Bookmark this

pharmaphorum
MAY 9, 2023
4th Dermatology Drug Development Summit Europe Mike.Hammerton Tue, 05/09/2023 - 20:11 Bookmark this
Drug Discovery World
MARCH 23, 2023
Aldena Therapeutics, a biotech company that is pioneering siRNA-based therapies for dermatological indications, has updated its immuno-dermatology pipeline, appointed a Scientific Advisory Board (SAB), and announced $30 million financing by Medicxi. appeared first on Drug Discovery World (DDW).

Outsourcing Pharma
MAY 23, 2022
With Veeva Link for Key People, LEO Pharma is looking to deepen its efforts to engage with knowledgeable medical professionals in the dermatology community.
Drug Discovery World
MARCH 13, 2023
LEO Pharma and ICON have formed a strategic partnership that will enable LEO to offer clinical trials within medical dermatology that are patient-centric and cost effective. The post LEO partners with ICON to offer patient-centric dermatology trials appeared first on Drug Discovery World (DDW).

BioTech 365
JULY 6, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Leo taps X-Chem to discover anti-inflammatory dermatology drugs.Leo taps X-Chem to discover anti-inflammatory dermatology drugs ntaylor Tue, 07/06/2021 – 07:16 from FierceBiotech: Biotech [link].

BioTech 365
DECEMBER 3, 2020
DermTech Presents Updates in Precision Medicine at 2020 Dermatology Drug Development Summit DermTech Presents Updates in Precision Medicine at 2020 Dermatology Drug Development Summit LA JOLLA, Calif.–(BUSINESS –(BUSINESS WIRE)–DermTech, Inc.

BioTech 365
MARCH 15, 2021
Dermatology Drugs Market Research Report 2020 – Global Industry Analysis and Growth Forecast to 2030 – ResearchAndMarkets.com Dermatology Drugs Market Research Report 2020 – Global Industry Analysis and Growth Forecast to 2030 – ResearchAndMarkets.com DUBLIN–(BUSINESS WIRE)–The “Dermatology Drugs Market Research (..)

Pharmaceutical Technology
MAY 30, 2023
According to GlobalData’s Looking Ahead to 2023 – the Future of Pharma report, five drugs set for approval in 2023 are projected to attain blockbuster status or near-blockbuster status by 2028 with US company dominance. Genmab AS’s epcoritamab presents as the top European drug launch and only oncological therapy.

BioTech 365
SEPTEMBER 21, 2021
Journal of Drugs and Dermatology Publishes Clinical Study on Topix Pharmaceuticals, Inc. New Vitamin C E + green Tea Polyphenols Serum Journal of Drugs and Dermatology Publishes Clinical Study on Topix Pharmaceuticals, Inc. New Vitamin C E + green Tea … Continue reading →

Drug Discovery World
FEBRUARY 16, 2024
This week our news highlights focus on skin conditions, with positive updates on hidradenitis suppurativa, systemic sclerosis, wound-healing, and other dermatological conditions like atopic dermatitis and vitiligo. The post This week in drug discovery (12-16 February) appeared first on Drug Discovery World (DDW).

BioTech 365
APRIL 26, 2021
United States Generic Drugs Market Report 2020-2026: Focus on CNS, Cardiovascular, Dermatology, Genitourinary/Hormonal, Respiratory, Anti-infective & Oncology – ResearchAndMarkets.com United States Generic Drugs Market Report 2020-2026: Focus on CNS, Cardiovascular, Dermatology, Genitourinary/Hormonal, Respiratory, Anti-infective & (..)
BioPharma Reporter
NOVEMBER 21, 2023
A partnership combining an integrated drug creation platform with a company specializing in dermatological expertise will deliver life-changing medicines to patients.

Pharmaceutical Technology
SEPTEMBER 29, 2022
The latest takeover is anticipated to boost the presence of Torrent in the dermatology segment. Holding a presence in the cosmetic dermatology sector, Curatio’s portfolio comprises more than over 50 brands that are commercialised in India. Among these facilities, four are approved by the US Food and Drug Administration (FDA).
XTalks
AUGUST 2, 2023
Verrica Pharmaceuticals is a dermatology therapeutics company developing medications for skin diseases that require medical interventions. Tune into this episode to learn more about YCANTH and its approval, as well as the changing landscape of dermatology, from Dr. Eichenfield.

BioTech 365
AUGUST 3, 2021
Eurofins Strengthens Its Global Leadership Position in Cosmetics and Personal Care Products Testing and Clinical Services and Grows Its Position in Dermatology Drug Testing Eurofins Strengthens Its Global Leadership Position in Cosmetics and Personal Care Products Testing and Clinical Services … Continue reading →

Pharmaceutical Technology
JANUARY 19, 2023
Concert’s patent portfolio includes oral Janus kinases JAK1/2 inhibitor, deuruxolitinib, to treat Alopecia Areata, an autoimmune dermatological disease. Sun Pharma stated that it will follow Concert’s plan to seek approval from the US Food and Drug Administration (FDA) for its new drug application in the first half of the year.
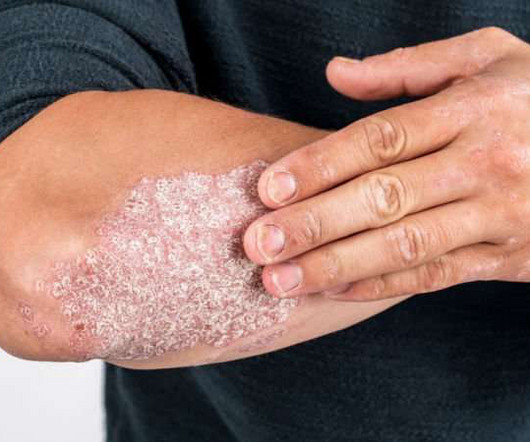
Drug Discovery World
MARCH 18, 2024
IL-23 plays a critical role in pathogenic T-cell activation in moderate-to-severe PsO and underpins the inflammatory response in PsO and other dermatological, rheumatological and gastroenterological IL-23-mediated diseases. JNJ-2113 is the first and only investigational targeted oral peptide designed to block the IL-23 receptor.

BioSpace
APRIL 21, 2021
On the back of the two positive Phase III trials, Ortho said it will submit a new drug application (NDA) to the U.S. Food and Drug Administration (FDA) in the second half of 2022.
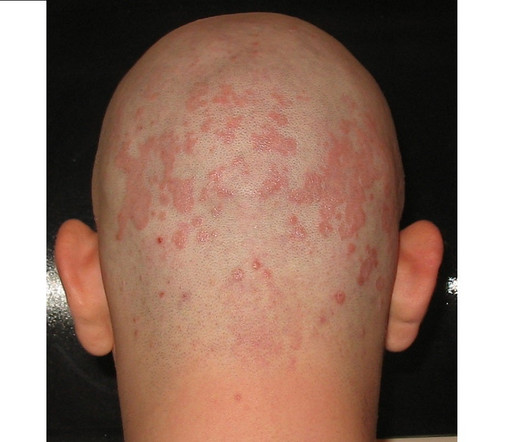
Pharmaceutical Technology
APRIL 19, 2023
The US Food and Drug Administration (FDA) has accepted Arcutis Biotherapeutics ’ new drug application (NDA) for roflumilast foam 0.3% The regulator has set 16 December 2023 as a prescription drug user fee act (PDUFA) target action date for the decision on the application.

Pharmaceutical Technology
JULY 12, 2022
Health Canada has accepted to review Arcutis Biotherapeutics’ New Drug Submission (NDS) for roflumilast cream 0.3% (ARQ-151) to treat plaque psoriasis in adult and adolescent patients. A validated target in dermatology, PDE4 is an enzyme that induces overactive immune responses.

Pharmaceutical Technology
FEBRUARY 28, 2023
According to GlobalData, Phase I drugs for Melanoma have a 76% phase transition success rate (PTSR) indication benchmark for progressing into Phase II. GlobalData’s report assesses how KFA-115’s drug-specific PTSR and Likelihood of Approval (LoA) scores compare to the indication benchmarks. Buy the report here.

Pharmaceutical Technology
APRIL 19, 2023
The technology for the pilot project, which is cleared for use by Health Canada and the US Food and Drug Administration (FDA), is a cloud-based platform that facilitates patient access to a dermatologist. In Canada, access to dermatological care is extremely limited, with only 1.7
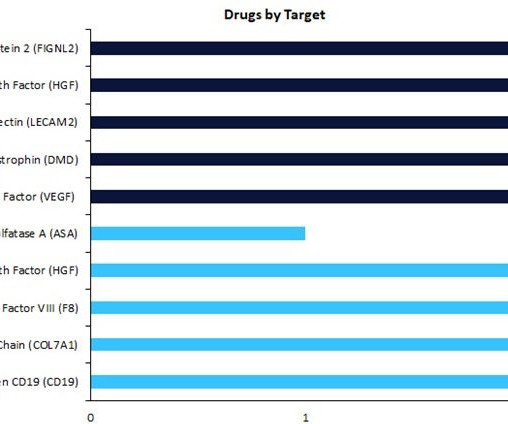
Pharmaceutical Technology
MARCH 27, 2023
Regenerative medicines in early-stage development (preclinical, discovery, or investigational new drug [IND]/ clinical trial application [CTA] filed status) have seen a change in drug targets compared to therapies in late-stage development (Phase II to pre-registration stage).

FDA Law Blog
OCTOBER 10, 2023
has joined the firm as a Principal Drug Regulatory Expert. Dr. Beitz served in leadership positions in FDA’s Center for Drug Evaluation and Research for nearly three decades. (“HP&M”), the largest FDA-dedicated law firm, is pleased to announce that Julie Beitz, M.D.
Drug Discovery World
APRIL 5, 2023
Regenerative medicines in early-stage development have seen a change in drug targets compared to therapies in late-stage development. According to analysts GlobalData, early-stage therapies are now focused on vascular endothelial growth factor (VEGF) as the top drug target, with three drugs currently in development.

Pharmaceutical Technology
MAY 19, 2023
The US Food and Drug Administration (FDA) has approved AbbVie’s Rinvoq (upadacitinib) for patients with Crohn’s disease who do not respond to TNF blockers, a common immune suppressant treatment for the condition. The FDA nod means Rinvoq is now approved for seven indications in gastroenterology, rheumatology and dermatology.

The Pharma Data
OCTOBER 29, 2020
30, 2020 /PRNewswire/ — Eli Lilly and Company (NYSE: LLY) and Incyte (NASDAQ: INCY) announced today new data for baricitinib (marketed as OLUMIANT ® ) will be presented at the annual Fall Clinical Dermatology meeting taking place virtually October 29-November 1, 2020. INDIANAPOLIS , Oct.
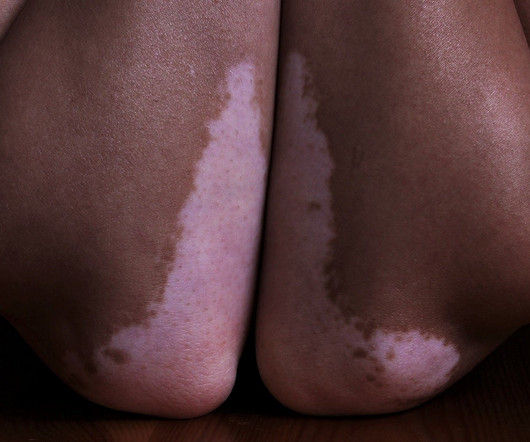
Pharmaceutical Technology
APRIL 17, 2023
QPS is preparing the pre-investigational new drug applications to be submitted to the US Food and Drug Administration (FDA) and the EU’s European Medicines Agency (EMA) for the CannQuit and ReneCann products. French company Eurofins Scientific is currently developing and manufacturing both products.

BioSpace
NOVEMBER 2, 2020
“With limited oral therapeutic options available for psoriasis, there remains a significant need for safe and effective therapies,” said April Armstrong, associate dean and professor of dermatology at the University of Southern California.
STAT News
DECEMBER 2, 2022
And a fragmented clinical trials industry has made it a prime target for private equity, which often consolidates markets by merging companies.

Pharmaceutical Technology
FEBRUARY 28, 2023
According to GlobalData, Phase I drugs for Malignant Mesothelioma have a 77% phase transition success rate (PTSR) indication benchmark for progressing into Phase II. GlobalData’s report assesses how KFA-115’s drug-specific PTSR and Likelihood of Approval (LoA) scores compare to the indication benchmarks.

pharmaphorum
OCTOBER 1, 2021
Already a major player in atopic dermatitis with Dupixent, Sanofi looked to expand its position in the category earlier this year when it bought Kymab and its lead drug KY1005 for the skin disorder for $1.1 Now, it says the drug has delivered “exciting” results in a proof-of-concept trial. billion upfront.

XTalks
APRIL 17, 2023
These results are very promising for patients with atopic dermatitis,” said Jonathan Silverberg, MD, professor of dermatology at George Washington University School of Medicine & Health Sciences and co-investigator of the studies, in the company’s press release. ADvocate2 showed similar highly statistically significant results.

Pharmaceutical Technology
FEBRUARY 28, 2023
According to GlobalData, Phase III drugs for Paroxysmal Nocturnal Hemoglobinuria have a 60% phase transition success rate (PTSR) indication benchmark for progressing into Pre-Registration. Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval.

Pharmaceutical Technology
FEBRUARY 28, 2023
According to GlobalData, Phase III drugs for Glomerulonephritis have a 67% phase transition success rate (PTSR) indication benchmark for progressing into Pre-Registration. GlobalData’s report assesses how Iptacopan hydrochloride’s drug-specific PTSR and Likelihood of Approval (LoA) scores compare to the indication benchmarks.

pharmaphorum
AUGUST 28, 2020
Shares in dermatology specialist Cassiopea, which is listed on the SWX exchange in Switzerland, rose almost 17% after news of the FDA approval emerged. The post FDA hands Cassiopea its first approval, for acne drug Winlevi appeared first on. Retinoids can help unclog pores, while antibiotics can limit the growth of bacteria.
pharmaphorum
SEPTEMBER 16, 2022
Atopic dermatitis (AD) is a highly prevalent chronic dermatological disease, with symptoms which include itching, dry skin, severe pain, and inflammation – all of which create considerable stress in the daily lives of patients and often negatively affect their day-to-day and long-term well-being.

Pharmaceutical Technology
FEBRUARY 24, 2023
According to GlobalData, Phase I drugs for Genital Warts (Condylomata Acuminata) have a 91% phase transition success rate (PTSR) indication benchmark for progressing into Phase II. Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval.

Pharmaceutical Technology
FEBRUARY 24, 2023
According to GlobalData, Phase I drugs for Cervical Intraepithelial Neoplasia (CIN) have a 92% phase transition success rate (PTSR) indication benchmark for progressing into Phase II. Attributes of the drug, company and its clinical trials play a fundamental role in drug-specific PTSR and likelihood of approval.
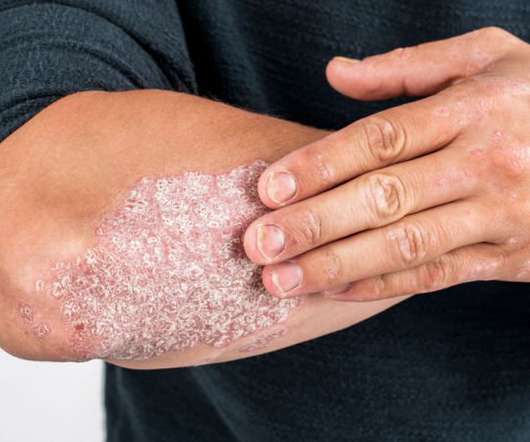
Drug Discovery World
SEPTEMBER 13, 2022
The US Food and Drug Administration (FDA) has approved Bristol Myers Squibb’s allosteric tyrosine kinase 2 inhibitor Sotyktu (deucravacitinib) for the treatment of adults with moderate-to-severe plaque psoriasis. The post FDA approves new first-line treatment for plaque psoriasis appeared first on Drug Discovery World (DDW).
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content