BridgeBio secures $1.25bn for genetic therapy development
Pharmaceutical Technology
JANUARY 19, 2024
BridgeBio Pharma has secured strategic financing of $1.25bn from Blue Owl Capital and CPP Investments for genetic therapies.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
JANUARY 19, 2024
BridgeBio Pharma has secured strategic financing of $1.25bn from Blue Owl Capital and CPP Investments for genetic therapies.

Pharmaceutical Technology
JULY 18, 2023
Scribe Therapeutics and Sanofi have expanded partnership to progress the development of in vivo genetic therapies to treat genomic diseases.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
DECEMBER 23, 2022
Eli Lilly and Company has expanded a licencing and partnership agreement with ProQR Therapeutics to discover, develop and market new genetic medicines. The post Lilly and ProQR to expand genetic medicine development agreement appeared first on Pharmaceutical Technology.

Worldwide Clinical Trials
JANUARY 24, 2024
Written By: Derek Ansel, MS, CCRA, Executive Director, Therapeutic Strategy Lead, Rare Disease Given that 80% of rare diseases have a genetic etiology, genetic implications should be addressed at the onset of a clinical program to support trial enrollment. One diagnostic example that I discussed in my presentation is autism.
Pharmaceutical Technology
NOVEMBER 8, 2022
Fulgent Genetics has acquired clinical-stage therapeutics development firm Fulgent Pharma for a total deal price of nearly $100m. According to the deal, the purchase price, contingent on adjustments, has to be paid by Fulgent Genetics as a combination of cash on hand and its shares of common stock.

Pharmaceutical Technology
MARCH 24, 2023
Moderna has entered a strategic partnership with Generation Bio for the development of non-viral genetic medicines. Moderna’s biological and technical expertise will be combined with core technologies of the non-viral genetic medicine platform from Generation Bio.

AuroBlog - Aurous Healthcare Clinical Trials blog
FEBRUARY 18, 2024
Indian healthcare industry is of the view that emergence of genetic diagnostics will be a paradigm shift towards personalized medicine. Through the ever-evolving landscape of healthcare, the role of genetic diagnostics offers personalized insights and proactive solutions for disease management and prevention.

Pharmaceutical Technology
FEBRUARY 13, 2023
SOPHiA GENETICS is expanding its collaboration with AstraZeneca to include multimodal approaches for developing cancer drugs. This collaboration aims to use the global SOPHiA DDM platform, a cloud-native platform of SOPHiA GENETICS, and multimodal algorithmic capabilities for AstraZeneca’s oncology portfolio.
Pharmaceutical Technology
SEPTEMBER 15, 2023
Cure Genetics has entered a partnership and licensing deal with Frametact to develop gene therapy for familial neurological ailments.

Pharmaceutical Technology
MARCH 1, 2024
1910 Genetics has entered an agreement with Microsoft, aiming to revitalise pharmaceutical research and development (R&D) productivity.

Bio Pharma Dive
FEBRUARY 23, 2023
Graphite Bio and Sangamo are stopping work on their respective sickle cell gene therapies, while Intellia revealed partner Novartis ended development of its genetic treatment for the blood disease.

Bio Pharma Dive
SEPTEMBER 14, 2020
Merck's deal for Seattle Genetics' antibody-drug conjugate comes as Gilead agreed to acquire rival developer Immunomedics in the biotech industry's largest buyout of 2020.

Pharmaceutical Technology
OCTOBER 25, 2022
Ast ellas Pharma has announced plans to make a strategic investment to back the development of Taysha Gene Therapies’ adeno-associated virus (AAV) development programmes for Rett syndrome and giant axonal neuropathy (GAN). This would allow the companies to boost the development of new therapy options for Rett syndrome and GAN patients.

Pharma Mirror
APRIL 28, 2021
Catalent will provide process development and CGMP manufacturing of AavantiBio’s adeno-associated viral (AAV) vector-based therapeutic candidate for use in clinical trials in the U.S. and Europe. Catalent will further support process optimization and look to reduce material.

Medical Xpress
MAY 16, 2023
However, how the brain has developed over the course of evolution and which genes are responsible for the high cognitive abilities is still largely unclear. The better our understanding of the role of genes in brain development, the more likely it will be that we will be able to develop treatments for serious brain diseases.

Medical Xpress
MARCH 14, 2023
National Institutes of Health researchers compared a new genetic animal model of Down syndrome to the standard model and found the updated version to be enhanced. The results of this study, published in Biological Psychiatry, may help researchers develop more precise treatments to improve cognition in people with Down syndrome.

Pharmaceutical Technology
DECEMBER 20, 2023
LC-002 is under clinical development by Genetic Immunity and currently in Phase II for Human Immunodeficiency Virus (HIV) Infections (AIDS).
Pharmaceutical Technology
FEBRUARY 15, 2023
In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Genetically modified animal models. Regeneron Pharmaceuticals is one of the leading patent filers in the generation of genetically modified animal models.

Pharmaceutical Technology
MAY 24, 2023
Forge Biologics has joined the public-private collaboration, the Bespoke Gene Therapy Consortium (BGTC), to expedite the development and manufacture of new AAV [adeno-associated virus] gene therapies to treat patients with rare diseases. Shilling was appointed to represent the company on the steering committee of the BGTC.
Pharmaceutical Technology
OCTOBER 19, 2022
Eli Lilly and Company has signed a definitive agreement for the acquisition of all outstanding shares of precision genetic medicine firm Akouos for a total deal value of up to nearly $610m or up to $15.50 Akouos focuses on developing adeno-associated viral gene therapies to treat inner ear conditions, including sensorineural hearing loss.

Pharmaceutical Technology
MAY 17, 2023
Chinook Therapeutics and Ionis Pharmaceuticals have entered a partnership to develop an antisense oligonucleotide (ASO) therapy to treat a rare, severe chronic kidney disease. The collaboration aims to discover, develop and commercialise an ASO therapy.
Pharmaceutical Technology
JUNE 4, 2023
In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Transcription factors for genetically modified cells. However, not all innovations are equal and nor do they follow a constant upward trend.

Worldwide Clinical Trials
FEBRUARY 6, 2023
When using genetic screening to identify clinical trial volunteers, a sponsor’s obligations for further testing and disclosure of results to patients and families are unclear, especially when the results have no impact on medical management. Guidance in this area is much needed. Read the full article!

Outsourcing Pharma
JANUARY 23, 2024
Sano Genetics says it is developing software that will enable âthe precision medicine revolutionâ and has raised $11.4 million in new funding.

Pharmaceutical Technology
MAY 4, 2023
Forge Biologics and global life sciences company Labcorp have announced a strategic adeno-associated virus (AAV) gene therapy development and manufacturing collaboration. This will improve accessibility to services for AAV-mediated gene therapy programmes. This will improve accessibility to services for AAV-mediated gene therapy programmes.

pharmaphorum
JUNE 9, 2023
3rd Rare & Genetic Kidney Disease Drug Development Summit Mike.Hammerton Fri, 09/06/2023 - 07:13 Bookmark this
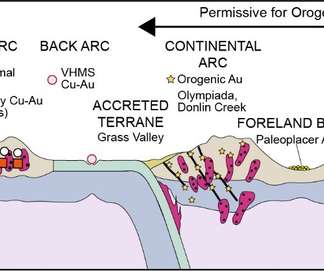
Scienmag
MARCH 30, 2022
Mineral systems: Their advantages in terms of developing holistic genetic models and for target generation in global mineral exploration Credit: GeoGeo Mineral systems: Their advantages in terms of developing holistic genetic models and for target generation in global mineral exploration [link] Announcing a new publication for Geosystems and Geoenvironment (..)

BioPharma Reporter
FEBRUARY 21, 2023
The Danish-US biotech Hemab Therapeutics bags $135m to finance the clinical development of prophylactic treatments for genetic bleeding disorders including Glanzmann thrombasthenia and von Willebrand disease.

Pharmaceutical Technology
FEBRUARY 9, 2023
The collaboration is aimed at discovering and developing new advanced treatments for genitourinary (GU) tumours as well as other related serious diseases using ReviR’s RNA-targeted technologies. Its initial focus is to develop therapeutics in oncology, CNS, and genetically defined disease indications.

Pharmaceutical Technology
APRIL 26, 2023
MiNA Therapeutics has entered into a research collaboration and option licensing agreement with BioMarin Pharmaceutical to speed up the development of therapeutic ribonucleic acid activation (RNAa) candidates to treat rare genetic diseases. BioMarin may then license the assets fully to develop and commercialise them across the globe.
STAT News
DECEMBER 10, 2022
NEW ORLEANS — An emerging class of genetically targeted drugs is inducing remissions in about one-third of patients with advanced leukemia, according to updates Saturday from separate clinical trials. Continue to STAT+ to read the full story…

Bio Pharma Dive
APRIL 1, 2022
Taysha Gene Therapies is narrowing its research efforts and cutting around 35% of its workforce, the latest in a series of layoffs announced by makers of genetic medicines.

pharmaphorum
JANUARY 24, 2023
Drug development has long been an issue for the pharma industry, due to the expense and the high failure rate of potential treatments. Ben Hargreaves finds that the vast amount of genetic data that exists today could help provide a faster, more targeted way of developing new drug candidates.

Roots Analysis
JANUARY 19, 2024
Introduction In a strategic move to accelerate the development and launch of genetic medicines, BridgeBio Pharma has successfully secured up to USD 1.25 Billion to Propel Genetic Medicine Development appeared first on Blog. billion from Blue Owl Capital and Canada Pension Plan Investment Board.

Pharma Mirror
FEBRUARY 23, 2021
Catalent is also announcing the launch of pDNA development and manufacturing services at its Rockville, Maryland facility. The post Catalent to Acquire Delphi Genetics and Launch US Plasmid Manufacturing Site appeared first on Pharma Mirror Magazine. SOMERSET, N.J. SOMERSET, N.J.

Medical Xpress
FEBRUARY 21, 2023
Researchers from Children's Hospital of Philadelphia (CHOP) developed two new zebrafish models for studying a specific genetic form of mitochondrial disease that represents the most common cause of Leigh syndrome. The findings were recently published in the journal Human Molecular Genetics.

FDA Law Blog
DECEMBER 13, 2023
Sasinowski — On December 12, 2023, FDA announced the creation of a new advisory committee specifically for treatments for genetic metabolic diseases, the Genetic Metabolic Diseases Advisory Committee, or “GeMDAC.” There are hundreds of known genetic metabolic diseases, most of which are rare and carry significant morbidity.

Pharmaceutical Technology
JUNE 10, 2023
CIGB-128 is under clinical development by Center for Genetic Engineering and Biotechnology and currently in Phase I for Brain Tumor. GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data. Buy the report here.

Pharmaceutical Technology
JUNE 10, 2023
CIGB-128 is under clinical development by Center for Genetic Engineering and Biotechnology and currently in Phase I for Brain Tumor. GlobalData tracks drug-specific phase transition and likelihood of approval scores, in addition to indication benchmarks based off 18 years of historical drug development data. Buy the report here.

pharmaphorum
MARCH 21, 2022
Neuroscience is arguably the next great frontier in medicine and 2022 dawned with a wave of exhilarating progress for the neurodegenerative drug development community. A startup led by Novartis veteran raises $80 million to develop treatments for people with degenerative brain conditions.
Drug Discovery World
JANUARY 19, 2024
UK scientists have uncovered the interplay between cancer-driving genetic mutations and inherited genetic variants in a rare type of blood cancer. In the future, this knowledge could aid drug development and interventions that reduce the risk of disease.
Outsourcing Pharma
FEBRUARY 24, 2022
The pharmaceutical companyâs Lilly Institute for Genetic Medicine reportedly will focus on developing RNA-based therapeutics in a âstate-of-the-artâ facility.

Bio Pharma Dive
DECEMBER 12, 2023
Based on the work of MIT scientists, the well-funded startup is developing ways to insert large sizes of genetic material anywhere in the genome without damaging or breaking DNA.

XTalks
MARCH 28, 2024
Duvyzat, a histone deacetylase (HDAC) inhibitor that works to reduce inflammation and muscle loss, is the first nonsteroidal drug approved to treat patients with all genetic variants of DMD. Progressive muscle weakness in the disease is caused by genetic mutations in the dystrophin gene that lead to a lack of functional dystrophin protein.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content