Pfizer brings mRNA flu vaccine into Phase 3 testing
Bio Pharma Dive
SEPTEMBER 14, 2022
The trial, expected to enroll 25,000 adults, is the second late-stage test of an mRNA flu shot to begin this year, following the June start of a Moderna study.

Bio Pharma Dive
SEPTEMBER 14, 2022
The trial, expected to enroll 25,000 adults, is the second late-stage test of an mRNA flu shot to begin this year, following the June start of a Moderna study.

Pharmaceutical Technology
SEPTEMBER 14, 2022
Tumour-infiltrating lymphocyte (TIL) therapy has been investigated as a treatment modality for melanoma for almost 20 years. As an autologous cell therapy, manufacturing the infusion product is a highly specialised process involving tumour harvest and the extraction and large-scale ex vivo expansion of TILs. Before TIL infusion, the patient undergoes lymphodepletion, which reduces the proportion of myeloid immune suppressive cells and creates space for the incoming lymphocytes.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Bio Pharma Dive
SEPTEMBER 14, 2022
The well-funded startup is planning Phase 3 trials for a drug it claims could be superior to top-selling medicines from Novartis and others.
Pharmaceutical Technology
SEPTEMBER 14, 2022
Novavax and Serum Institute of India (SII) have reported that the former’s Covid-19 vaccine, NVX-CoV2373, has received full product registration from the South African Health Products Regulatory Authority (SAHPRA), with conditions. The protein-based vaccine is indicated as a two-dose initial regimen for active immunisation for the prevention of Covid-19 in adults aged 18 years and above.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud
Bio Pharma Dive
SEPTEMBER 14, 2022
The joint venture with Radiopharm Theranostics is pursuing a field of research that’s seen a string of recent deals and investments by large drugmakers.

Pharmaceutical Technology
SEPTEMBER 14, 2022
Regulatory T cells (Tregs) are a subpopulation of T cells that act in an immunosuppressive manner downregulating the activation and proliferation of effector T cells. Generation of an anti-tumour immune response can lead to complete and durable tumour regression across cancer types, with effector T cells being a critical component. Multiple studies have reported high levels of tumour-infiltrating Tregs to correlate with adverse patient outcomes, making a target for Treg depletion an area of inte
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Rethinking Clinical Trials
SEPTEMBER 14, 2022
Dr. Tor Biering-Sørensen. In this Friday’s PCT Grand Rounds, Dr. Tor Biering-Sørensen of the University of Copenhagen will present “Using Nationwide Registries to Conduct Pragmatic Randomized Trials: The DANFLU Program.” The Grand Rounds session will be held on Friday, September 16, 2022, at 1:00 pm eastern. Dr. Biering-Sørensen is professor and head of the Center for Translational Cardiology and Pragmatic Randomized Trials at the University of Copenhagen and head of the Cardio

Pharmaceutical Technology
SEPTEMBER 14, 2022
The ulcerative colitis (UC) therapeutics market has many options available and a relatively strong development pipeline. Despite this, there are some unmet needs that remain and opportunities that could be further explored. GlobalData’s upcoming report, Ulcerative Colitis: Global Drug Forecast and Market Analysis, explores several unmet needs and opportunities in the UC space, some of which include developing treatments for patients with certain comorbidities, and combination products.

AuroBlog - Aurous Healthcare Clinical Trials blog
SEPTEMBER 14, 2022
The medical devices industry in the country has sought the Centre to define online and offline mode of sale, provide more clarity on the regulations that are binding the online sales and suggested inclusion of online medical devices players under the Uniform Code for Medical Devices Marketing Practices (UCMDMP). The Association of Indian Medical Device […].
Pharmaceutical Technology
SEPTEMBER 14, 2022
RayzeBio has raised $160m in a Series D funding round to advance targeted radiopharmaceuticals for cancer. Viking Global Investors, Sofinnova Investments and Wellington Management jointly led the financing round. Existing investors of the company and additional new investors, including Ally Bridge Group, Laurion Capital Management, Soleus Capital, Sands Capital and an undisclosed international investor, also took part in the funding.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

NPR Health - Shots
SEPTEMBER 14, 2022
Thursday, nearly all abortions must cease in Indiana after state lawmakers passed a full ban in August. Since Dobbs , Indiana has been a destination for those in surrounding states seeking abortions.

BioPharma Reporter
SEPTEMBER 14, 2022
Novo Nordisk and Microsoft have entered a new strategic collaboration to combine Microsoftâs computational services, cloud, and artificial intelligence (AI) with Novo Nordiskâs drug discovery, development, and data science capabilities.
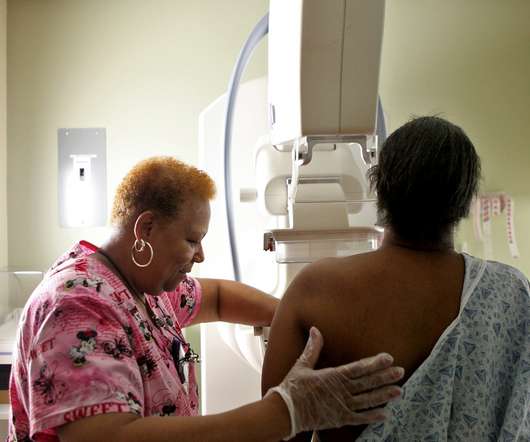
NPR Health - Shots
SEPTEMBER 14, 2022
The ACA has required health insurers to provide many medical screenings and other preventive services with no out-of-pocket cost to health plan members. But a recent court decision could upend that.

Pharmaceutical Technology
SEPTEMBER 14, 2022
Pharmaceutical drug research and development (R&D) activities are capital-intensive, which makes the outsourcing of clinical dose manufacturing and marketing popular. The growing clinical trial market is increasing the need for efficient clinical dose contract manufacturing and marketing capabilities to achieve technical and commercial goals. The clinical trials market is expected to rise further in the upcoming years due to factors such as trial globalisation, rapid evolution of technologie

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

NPR Health - Shots
SEPTEMBER 14, 2022
Child poverty is at a historic low, according to Census bureau data, and the rate of those without health insurance dropped in 2021. But the good news may be short-lived, as policy measures expire.

Pharmaceutical Technology
SEPTEMBER 14, 2022
API peptides and proteins-based drugs have gained much attention in the past decade. They are promising therapeutics for the treatment of a variety of metabolic and oncological disorders. More than 100 approved peptide-based therapeutics are currently available in the market. Victoza, Copaxone, Lupron, Zoladex, Sandostatin, and Somatuline are some of the popularly marketed peptide API therapeutic drugs while more than 600 peptide-based pharmacological leads are being investigated worldwide, acro

NPR Health - Shots
SEPTEMBER 14, 2022
With abortion illegal in a growing number of states, both groups that help patients access the procedure and those who oppose it are reporting more calls for help.

STAT News
SEPTEMBER 14, 2022
The rollout of the Covid-19 vaccination program has reached new heights of complexity with the start of the bivalent booster program, leading to concerns about the potential for more errors in the administration of vaccines. Even before the addition of the new booster shots, more than 5,300 errors in vaccine dose delivery in children alone were reported, according to data from the Centers for Disease Control and Prevention.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

pharmaphorum
SEPTEMBER 14, 2022
A $14 million research project is getting underway in the US to see whether cancer and other diseases can be diagnosed by picking up subtle changes in a person’s voice patterns. The National Institutes of Health (NIH)-funded project is being run by researchers at the University of South Florida in collaboration with Weill Cornell Medicine in New York City, 10 other institutions in the US and Canada, and with the help of French/US artificial intelligence specialist Owkin.
STAT News
SEPTEMBER 14, 2022
While pharma companies struggle to show that cutting-edge therapies can treat, prevent, or slow Alzheimer’s disease and other forms of dementia, a new study suggests that a far simpler and cheaper tool could improve cognition in seniors: daily multivitamins. Researchers found that adults 65 and older who took the common multivitamin Centrum Silver over the course of a three-year study showed more improvement on scores of overall cognition and memory than participants who took a placebo.
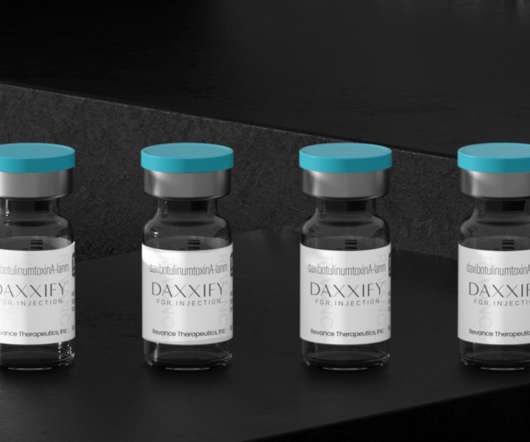
XTalks
SEPTEMBER 14, 2022
Daxxify was FDA-approved for similar cosmetic purposes as Botox and other neuromodulators like Dysport and Xeomin. With age, faces can undergo a variety of changes from the various expressions made over the years and daily use of facial muscles. Many people choose to undergo non-invasive cosmetic procedures to prevent or reduce the appearance of signs of aging.

STAT News
SEPTEMBER 14, 2022
A familiar refrain echoes through drug ads in the United States. It’s heard at the end of TV spots and plastered across magazine pages: Ask your doctor if this drug is right for you. But as medicine moves increasingly online, direct-to-consumer advertising is adopting a more assertive catchphrase: Talk to a doctor now. Dozens of drug sites now have built-in buttons to “talk to a doctor now” about everything from novel migraine medications to a treatment for sickle cell disea

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

Drug Discovery World
SEPTEMBER 14, 2022
Nicola Richmond, VP of AI, BenevolentAI, will be speaking at the Tech Theatre, ELRIG Drug Discovery 2022, sponsored by DDW. Nicola Richmond is a mathematician with over 20 years’ experience of developing digital solutions to problems in drug discovery and development. She will be presenting a talk on ‘The lab of the future is now: Using AI or automation for the drug discovery process’ The Tech Theatre will feature a series of seminars focused on automation and technology for dr

STAT News
SEPTEMBER 14, 2022
Christopher Hartnick never expected his work as a doctor to intersect with political discussions about abortion and the right of pregnant people to make choices about their own bodies. Yet as a pediatric ear, nose, and throat physician who specializes in treating babies and children who have difficulty breathing, he’s had up-close looks at how prospective parents make life-or-death decisions over the course of a pregnancy.

Pharmaceutical Commerce
SEPTEMBER 14, 2022
Professor Laura Empson pulls back the curtain on insecure overachieving professionals and explains how they can come to accept damaging patterns of overwork as inevitable and even desirable.
STAT News
SEPTEMBER 14, 2022
Trade-offs in medicine can be harsh, even when clearly necessary. Chemotherapy can save people from cancer, but the side effects can wreak havoc on their bodies for months or years. These trade-offs aren’t limited to the clinical world. They are also part of research. For half a decade, I oversaw a clinical trial testing a new vaccine against the hepatitis C virus, a slowly progressing and usually silent infectious disease that damages the liver over decades, and can cause cancer and even

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

NPR Health - Shots
SEPTEMBER 14, 2022
The decision to grant a 14-day restraining order against the state law means abortions through 20 weeks' gestation can continue for now. (Image credit: Barbara J.

STAT News
SEPTEMBER 14, 2022
Drug ads have long tried to influence patients’ medical choices, directing them to “ask your doctor” if a marketed medication is right for them. But now, pharma companies have an even more powerful tool to start those conversations and turn them into prescriptions: telemedicine. When a patient sees an ad or visits a manufacturer’s website today, many will find buttons to “talk to a doctor now” about their symptoms.

pharmaphorum
SEPTEMBER 14, 2022
With countless media stories regarding COVID-19 published over a period of several years, it was obvious that people around the globe were tuned in to every step of the fight against the pandemic, eagerly learning about everything from symptoms to vaccine development. It was the most engaged a global audience has ever been with a public health issue, media playing a key role.
STAT News
SEPTEMBER 14, 2022
Good morning, everyone, and welcome to the middle of the week. Congratulations on making it this far, and remember there are only a few more days until the weekend arrives. So keep plugging away. After all, what are the alternatives? While you ponder the possibilities, we invite you to join us for a delightful cup of stimulation. Our choice today is, once again, maple cinnamon French toast.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content