Beam, capitalizing on stock surge, buys gene therapy tools developer for $120M
Bio Pharma Dive
FEBRUARY 23, 2021
Guide Therapeutics, the company Beam acquired, is working on ways to more efficiently deliver gene editing therapies into the body.
Bio Pharma Dive
FEBRUARY 23, 2021
Guide Therapeutics, the company Beam acquired, is working on ways to more efficiently deliver gene editing therapies into the body.

Pharma Mirror
FEBRUARY 23, 2021
SOMERSET, N.J. and GOSSELIES, BELGIUM — Catalent, the leading global provider of advanced delivery technologies, development, and manufacturing solutions for drugs, biologics, cell and gene therapies, and consumer health products, and Delphi Genetics, a plasmid DNA (pDNA) cell and gene therapy contract development and manufacturing organization (CDMO) based in Gosselies, Belgium, today announced that they have entered into a final and definitive agreement whereby Catalent will acquire 100% of th
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

BioSpace
FEBRUARY 23, 2021
Pfizer has been accused of bullying governments in Latin America over coronavirus disease 2019 (COVID-19) vaccine negotiations, a recent investigative article revealed.

BioPharma Reporter
FEBRUARY 23, 2021
Real-world data from England shows that the Pfizer/BioNTech vaccine offers 'clear protection from first dose' and may help interrupt transmission; while Scottish data shows the first doses of vaccines led to a substantial fall in hospital admissions.

Scienmag
FEBRUARY 23, 2021
Western forests may be less able to regenerate following large-scale diebacks Credit: USGS DURHAM, N.C. — Younger, smaller trees that comprise much of North America’s eastern forests have increased their seed production under climate change, but older, larger trees that dominate forests in much of the West have been less responsive, a new Duke University-led […].

pharmaphorum
FEBRUARY 23, 2021
Sanofi’s cancer immunotherapy Libtayo (cemiplimab) has a third US indication after the FDA approved it for certain untreated lung cancer patients. The approval in first-line lung cancer, in non-small cell lung cancer patients whose tumours have high PD-L1 expression brings the drug into competition with Merck & Co’s Keytruda (pembrolizumab), which has become the go-to therapy in this disease.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Clinical Trial Podcast
FEBRUARY 23, 2021
Almost everyone I’ve met in clinical research has accidentally discovered this hidden profession. Your interest in science and medicine somehow got you involved in clinical research and clinical trial management. But didn’t you wish you had a crystal clear understanding of “Who’s Who In Clinical Research?”. That’s when this post comes handy. Clinical Research is sometimes also referred to as Medical Affairs or Clinical Affairs.

BioSpace
FEBRUARY 23, 2021
Public Health England (PHE) published real-world analyses of people in the UK who had received the first shot of the Pfizer-BioNTech COVID-19 vaccine.

BioPharma Reporter
FEBRUARY 23, 2021
Sanofiâs vaccine manufacturing plant in Marcy lâEtoile, France, will formulate and fill vials of Janssenâs COVID-19 vaccine: producing around 12 million doses a month to help address global supply demands.
BioSpace
FEBRUARY 23, 2021
Biopharma companies may receive NIH funding, but generally, not to the extent that academia does. However, the Genetown Hotbed has several biopharma companies that received significant funds from the NIH in 2020. Here’s a look at the top 10.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

XTalks
FEBRUARY 23, 2021
Reese’s Peanut Butter Cups, one of America’s favorite snacks, is getting an organic makeover. Organic Reese’s Peanut Butter Cups , the Hershey Company’s latest offering, gives chocolate and peanut butter lovers an alternative that is better for the environment. Available in milk and dark chocolate options, the new candy is certified USDA organic and verified by the Non-GMO Project and the Rainforest Alliance.

BioSpace
FEBRUARY 23, 2021
BMS is looking to hire "several hundred employees over the next few years," all working to ensure the production of both their clinical and commercial cell therapies.
BioPharma Reporter
FEBRUARY 23, 2021
Sanofi and GSK have initiated a new Phase 2 study for their COVID-19 vaccine candidate; with a Phase 3 study set to follow in Q2 and potential rollout of the vaccine in Q4.

pharmaphorum
FEBRUARY 23, 2021
The market access landscape in Germany is particularly interesting in 2021, as a key component – AMNOG – is ten years old, and that milestone prompts reflections on what has worked well and what needs reform. In the latest of a suite of features looking at the biggest markets in Europe, Leela Barham takes stock of what 2021 could bring for market access in Germany. .

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

BioPharma Reporter
FEBRUARY 23, 2021
The US Food and Drug Administration (FDA) has issued guidance for companies developing vaccines, diagnostics and therapeutics against new variants of SARS-CoV-2.

Pharma Times
FEBRUARY 23, 2021
Cell and Gene Therapy Catapult report also found a surge in the number of commercially sponsored trials
pharmaphorum
FEBRUARY 23, 2021
There is limited access to biomarker testing for cancer in Europe, despite the huge potential of the technology to improve outcomes, according to a new report. The report from European Federation of Pharmaceutical Industries and Associations (EFPIA ) shows the results of research conducted in 2020 to assess the availability, quality and reimbursement of biomarker tests in the EU27 and the UK.

JAMA Internal Medicine
FEBRUARY 23, 2021
This cohort study evaluates evidence of SARS-CoV-2 infection based on diagnostic nucleic acid amplification test among patients who tested positive versus negative for antibodies.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

BioSpace
FEBRUARY 23, 2021
Your career shouldn’t have to stay in a holding pattern just because you’re not at the office.

JAMA Internal Medicine
FEBRUARY 23, 2021
As a physician working in New York, New York, where coronavirus disease 2019 (COVID-19) hit hard in March and April of 2020, people often ask me how to interpret their severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) antibody results. Many people have positive test results for the antibody, some of them received a diagnosis of COVID-19, some of them had symptoms that were consistent with COVID-19 but were never tested because of a limited availability of testing, and some were never
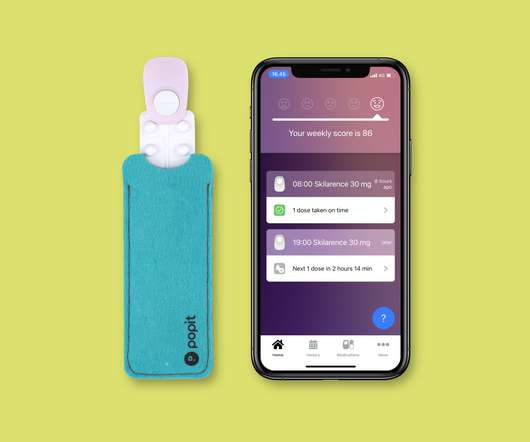
pharmaphorum
FEBRUARY 23, 2021
Ashfield Engage has agreed a deal to incorporate Popit’s digital tools in its patient support programmes, with the aim of improving adherence to treatments. . Finnish startup Popit has developed a device called Popit Sense that clips to a blister pill sheet and uses technology to monitor whether a dose has been taken, sending reminders to patients via a smartphone app.
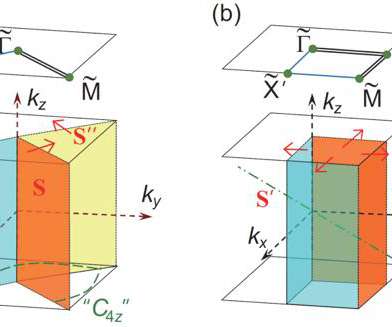
Scienmag
FEBRUARY 23, 2021
Credit: @Science China Press Using the symmetries of the systems, people can define various topological invariants to describe different topological states. The topological materials can be accurately discovered by calculating the topological invariants. Recently, researchers found that irreducible representations and compatibility relationships can be used to determine whether a material is topological nontrivial/trivial insulator (satisfying […].

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

pharmaphorum
FEBRUARY 23, 2021
The FDA is to begin a fast review of Incyte’s Jakafi (ruxolitinib) for patients with chronic graft-versus-host disease (GVHD), which cannot be treated with steroids. The US pharma, which has partnered with Novartis to develop this first-in-class JAK1/JAK2 inhibitor, said the FDA will review data from the phase 3 REACH 3 study comparing Jakafi to best available therapy in patients aged 12 years and older with steroid refractory GVHD.
Scienmag
FEBRUARY 23, 2021
Credit: Joppe Oldenburg Peer review/Experimental study/Animals Propranolol, a drug that is efficacious against infantile haemangiomas (“strawberry naevi”, resembling birthmarks), can also be used to treat cerebral cavernous malformations, a condition characterised by misshapen blood vessels in the brain and elsewhere. This has been shown by researchers at Uppsala University in a new study published in […].

Pharma Times
FEBRUARY 23, 2021
Company has submitted a supplemental new drug application for mild-to-moderate plaque psoriasis
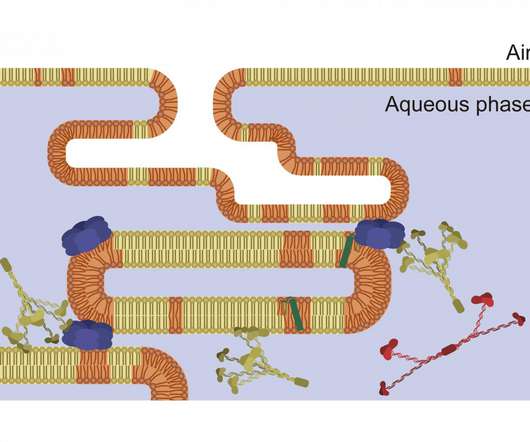
Scienmag
FEBRUARY 23, 2021
A new study reveals how lower temperatures improve the activity of surfactant, a molecular mixture that is essential for breathing. This indicates that therapeutic hypothermia is a potential treatment for acute respiratory distress syndrome, which affects Credit: Image courtesy of Chiara Autilio and created by Alejandro Alonso. ROCKVILLE, MD – A tenth of all intensive […].

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

BioSpace
FEBRUARY 23, 2021
Chinese fusion protein company Clover Biopharmaceuticals raised a $230 million series C round co-led by GL Ventures and Temasek, which will push its COVID-19 vaccine into Phase II/III testing and production planning.
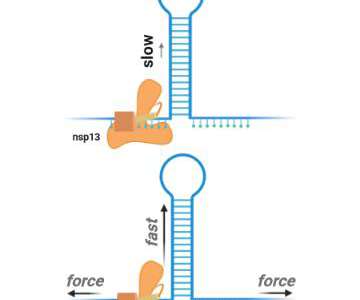
Scienmag
FEBRUARY 23, 2021
Viruses make copies of themselves during an infection, and new research sheds light on one of the coronavirus molecules that is important for this process, providing a potential drug target that could work for Covid-19 and other coronavirus outbreaks too. Credit: Image courtesy of Keith Mickolajczyk. ROCKVILLE, MD – Coronaviruses exploit our cells so they […].

BioPharma Reporter
FEBRUARY 23, 2021
Human B cells continue to work against SARS-CoV-2 months after infection, but derived antibodies do not recognize mutant variants from Brazil and South Africa, finds new research published in Science Immunology.
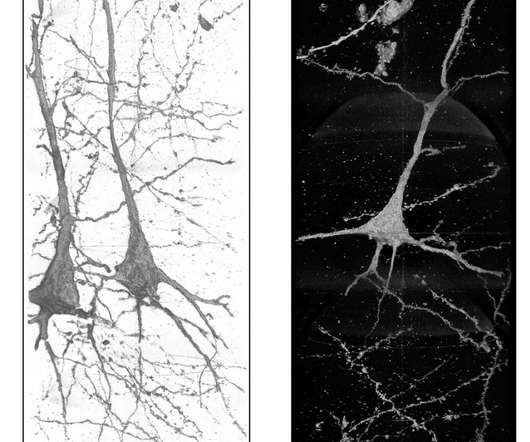
Scienmag
FEBRUARY 23, 2021
Credit: Ryuta Mizutani Schizophrenia, a chronic, neurological brain disorder, affects millions of people around the world. It causes a fracture between a person’s thoughts, feelings and behavior. Symptoms include delusions, hallucinations, difficulty processing thoughts and an overall lack of motivation. Schizophrenia patients have a higher suicide rate and more health problems than the general population, […].

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content