EQRx resets strategy, abandoning plans to disrupt drug pricing
Bio Pharma Dive
MAY 8, 2023
Launched in 2020 with a "radical" vision, EQRx ran into roadblocks that stymied its efforts to develop new medicines and undercut competitors on price.

Bio Pharma Dive
MAY 8, 2023
Launched in 2020 with a "radical" vision, EQRx ran into roadblocks that stymied its efforts to develop new medicines and undercut competitors on price.
Pharmaceutical Technology
MAY 8, 2023
The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms, and the gravity of unmet needs, as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Immuno-oncology in Pharmaceuticals: Microbio t a rest
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
MAY 8, 2023
The company agreed to buy Cyclica and Valence after winnowing down a list of more than 100 potential takeover targets, its CEO told BioPharma Dive.
Pharmaceutical Technology
MAY 8, 2023
The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Anti-influenza antibody compositions.

Bio Pharma Dive
MAY 8, 2023
Although there is currently no cure, there is great promise in the fight against lupus. Real-world data and artificial intelligence are both critical to the future of clinical research.

Pharmaceutical Technology
MAY 8, 2023
Ei s ai and Bliss Biopharmaceutical have entered into a joint development agreement for antibody-drug conjugate (ADC), BB-1701, to treat cancers. This deal also includes option rights for a strategic collaboration. The collaboration will help in further development of BB-1701 across the world and advance the ADC towards late stage of development. Currently, BB-1701 is being evaluated in Phase I/II trials in China and the US.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.
Pharmaceutical Technology
MAY 8, 2023
The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms, and the gravity of unmet needs, as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Anti-viral antigen-bas

Bio Pharma Dive
MAY 8, 2023
Providing accurate information to protect patients from misinformation is a key goal for pharmaceutical companies, and print media can be a crucial part of the solution.
Pharmaceutical Technology
MAY 8, 2023
The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms, as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Anti-inflammatory anaesthetics.

Bio Pharma Dive
MAY 8, 2023
Six months after being named head of RNA-focused biotech Laronde, Pablo Cagnoni has left the buzzy startup to run Incyte’s R&D work.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations
Pharmaceutical Technology
MAY 8, 2023
The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms, and the gravity of unmet needs, as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Zinc-finger nucleases.

Bio Pharma Dive
MAY 8, 2023
The deal is the latest in a string of private equity-backed deals for contract development and manufacturing businesses in the pharmaceutical sector.
Pharmaceutical Technology
MAY 8, 2023
The pharmaceutical industry continues to be a hotbed of innovation, with activity driven by the evolution of new treatment paradigms, and the gravity of unmet needs, as well as the growing importance of technologies such as pharmacogenomics, digital therapeutics, and artificial intelligence. In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Immuno-oncology in Pharmaceuticals: Monoclonal antibo

Bio Pharma Dive
MAY 8, 2023
Today’s commercial model cannot support pharma’s future. A new future calls for bold transformation.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Pharmaceutical Technology
MAY 8, 2023
Shanghai Junshi Biosciences and Dr. Reddy’s Laboratories have partnered for the development and commercialisation of the anti-PD-1 monoclonal antibody, toripalimab, in 21 countries. Under the terms of the license and commercialisation deal, Dr. Reddy’s will get licence for the development and commercialisation of toripalimab in India, Panama, Peru, Colombia, South Africa, Brazil, Argentina, Chile, Uruguay, and Mexico.
Medical Xpress
MAY 8, 2023
When you need a bit of motivation, it often has to come from within. New research suggests cancer-fighting immune cells have found a way to do just that.

Pharmaceutical Technology
MAY 8, 2023
The China National Medical Products Administration (NMPA) has granted approval for BeiGene’s four applications for Brukinsa (zanubrutinib). The approvals were granted for two Supplemental Applications to covert conditional approval to regular approval, and two Supplemental New Drug Applications to treat Waldenström’s macroglobulinemia (WM), and chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL) in adult patients.

Rethinking Clinical Trials
MAY 8, 2023
The NIH Pragmatic Trials Collaboratory is partnering with AcademyHealth to offer a 1.5-day preconference workshop at the 2023 Annual Research Meeting in Seattle, Washington. The workshop, “ Driving Tomorrow’s Outcomes Through Clinical Research in Real-World Settings ,” will cover the essentials of embedded healthcare systems research, along with strategies for conducting clinical trials that provide real-world evidence necessary to inform both practice and policy.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
Medical Xpress
MAY 8, 2023
Cancer remains one of the leading causes of death worldwide. Researchers are constantly looking for new therapies with improved anticancer activity, fewer side effects, and lower costs. In recent years, interest in bacterial agents as anticancer therapeutics has grown, due to the ability of some bacteria to selectively grow inside hypoxic tumors, i.e., these cells do not receive rich oxygen supply.

FDA Law Blog
MAY 8, 2023
By Lisa M. Baumhardt, Senior Medical Device Regulation Expert & Philip Won & Gail H. Javitt — FDA recently published a long-awaited draft guidance aimed at reducing the need for prior FDA authorization of modifications to artificial intelligence/machine learning (AI/ML)-enabled device software functions (ML-DSFs). The draft guidance, “Marketing Submission Recommendations for a Predetermined Change Control Plan for Artificial Intelligence/Machine Learning (AI/ML)-Enabled Device Software
Medical Xpress
MAY 8, 2023
Animal studies indicate that a new COVID-19 vaccine developed at Rutgers may provide more durable protection against SARS-CoV-2 and its emerging variants than existing vaccines.
XTalks
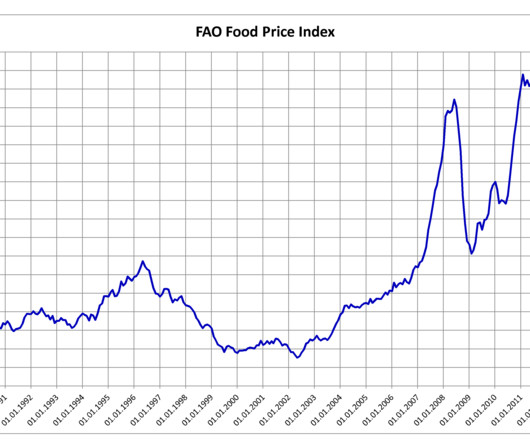
MAY 8, 2023
According to the United Nations’ (UN) Food and Agriculture Organization’s (FAO) food price index , the world price index saw an increase in April after a year-long decline. However, despite this rise, the index remains approximately 20 percent lower than the record high it reached in March 2022, following Russia’s invasion of Ukraine. The FAO food price index monitors the prices of the most traded food commodities worldwide and measures the average monthly change in international pr

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.
Medical Xpress
MAY 8, 2023
If you're a health care provider treating people with type 2 diabetes (T2D), University at Buffalo researcher Mehmet A. Eskan has this suggestion for you: check your patients' teeth.

Fierce Pharma
MAY 8, 2023
As COVID vaccine sales plummet, BioNTech looks for new growth opportunities zbecker Mon, 05/08/2023 - 11:02
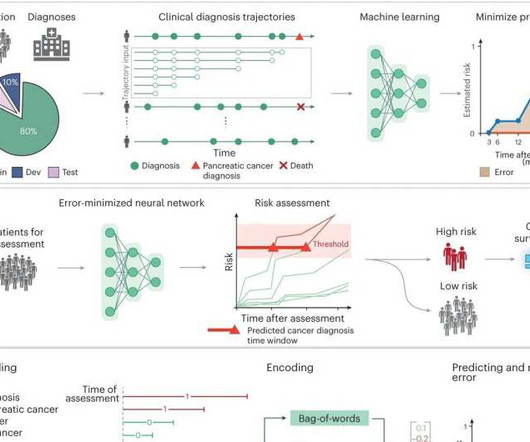
Medical Xpress
MAY 8, 2023
An artificial intelligence tool has successfully identified people at the highest risk for pancreatic cancer up to three years before diagnosis using solely the patients' medical records, according to new research led by investigators at Harvard Medical School and the University of Copenhagen, in collaboration with VA Boston Healthcare System, Dana-Farber Cancer Institute, and the Harvard T.H.

Fierce Pharma
MAY 8, 2023
Pfizer, AstraZeneca run it back in IDEA Pharma's annual innovation rankings kdunleavy Mon, 05/08/2023 - 10:21

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.
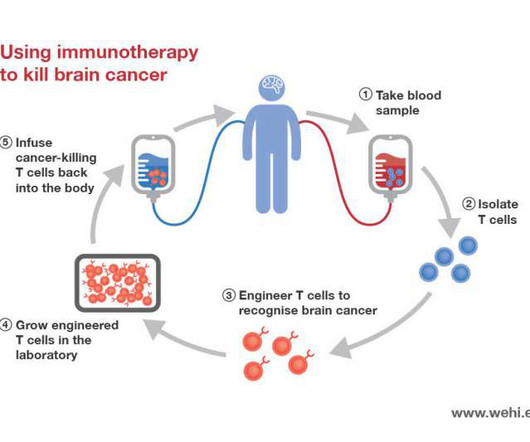
Medical Xpress
MAY 8, 2023
Usually occurring in children aged 5 to 7 years old, Diffuse Intrinsic Pontine Glioma (DIPG) is an aggressive type of brain tumor that affects 20 children in Australia each year.

Fierce Pharma
MAY 8, 2023
After production problems, Catalent delays earnings and plans to slash guidance fkansteiner Mon, 05/08/2023 - 10:47

Medical Xpress
MAY 8, 2023
The Southwest Rural Health Research Center at the Texas A&M University School of Public Health has published a peer-reviewed paper detailing Healthy People 2030 priorities that will be most critical for rural America in the upcoming decade. These priorities were identified by rural health stakeholders across the United States. This publication comes ahead of the center's release of Rural Healthy People 2030—a continuation of a long-standing tradition of the Southwest Rural Health Research Center

BioSpace
MAY 8, 2023
The Swiss pharmaceutical company filed a suit Friday joining a growing group of legal complaints by Big Pharma seeking to block the Inflation Reduction Act’s Drug Price Negotiation Program.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content