5 FDA decisions to watch in the fourth quarter
Bio Pharma Dive
OCTOBER 2, 2023
The regulator is considering approval of what would be the first CRISPR medicine, as well as important clearances for Alnylam, Bristol Myers, Amgen and Pfizer.

Bio Pharma Dive
OCTOBER 2, 2023
The regulator is considering approval of what would be the first CRISPR medicine, as well as important clearances for Alnylam, Bristol Myers, Amgen and Pfizer.

Antidote
OCTOBER 3, 2023
Clinical trials are an integral part of the medical industry, as they are required for any new treatment to be approved. Every medicine used today, whether prescription or over the counter, is a result of the rigorous research process that is a clinical trial — and for these trials to take place, volunteers are vital.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
OCTOBER 3, 2023
Only two of the eight biotech companies that priced offerings since July 1 trade at or above their debut share price, according to data from BioPharma Dive.

pharmaphorum
OCTOBER 4, 2023
Navigating EAPs: Clinical trials and the path to investigational drugs Mike.

ACRP blog
OCTOBER 3, 2023
Lessons learned from a new clinical research internship program held in central Pennsylvania last summer have helped prepare new and upcoming entrants for the research workforce, as well as to inform how the next generation of the program will evolve. As reported by the Penn State College of Medicine , six interns spent 11 weeks taking active roles in existing research projects, immersing themselves in the conduct of research, and learning more about careers in clinical research at the college’s

Cloudbyz
OCTOBER 4, 2023
Electronic Data Capture (EDC) systems are widely used in clinical trials to collect, manage, and analyze data from clinical studies. While EDC systems primarily focus on data capture and management, integrating trial budget management software within the EDC system can offer several advantages: 1. Streamlined Workflow: Combining EDC and budget management functionalities within the same system can create a more streamlined workflow for clinical trial teams.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.
Drug Discovery World
OCTOBER 2, 2023
Sophie Winandy , Executive Director of Medical Imaging at ICON explores the benefits of AI and how it can be integrated throughout medical imaging within clinical trials. Artificial intelligence (AI) is rapidly taking hold in the healthcare sector 1 , including in medical imaging, where AI often targets screening, patient eligibility, detection and disease response (Figure 1).

Pharmaceutical Technology
OCTOBER 2, 2023
The Physiology and Medicine Prize has gone to two researchers whose work laid the foundation for Pfizer and Moderna’s Covid-19 vaccines.

AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 3, 2023
If you believe the headlines, seaweeds can do almost anything from storing tons of carbon and stopping cows from belching methane, to making biofuels and renewable plastics – all while sustaining vibrant coastal ecosystems and feeding communities.

Bio Pharma Dive
OCTOBER 6, 2023
The funding, which is part of ACT for ALS, will support expanded access studies of drugs developed by Prilenia Therapeutics, Clene Nanomedicine and Rapa Therapeutics.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Rethinking Clinical Trials
OCTOBER 2, 2023
The Patient-Centered Outcomes Core has developed a new tool kit to provide resources to support the capture of patient-reported outcome (PRO) measures in diverse study populations. The tool kit is intended for research teams conducting pragmatic clinical trials, including those participating in the NIH Pragmatic Trials Collaboratory’s Demonstration Projects.

Pharmaceutical Technology
OCTOBER 5, 2023
Lumicell co-founder, Moungi Bawendi, has been awarded the accolade alongside two others for his research into quantum dots.
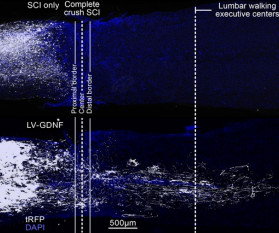
AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 2, 2023
Researchers have reversed ‘irreversible paralysis’ in mice with complete spinal cord injuries using gene therapy. The team found that regrowing neurons is not enough to fully restore walking and that regenerative therapies must target specific neurons and guide them back to where they belong, which was previously unknown.

Bio Pharma Dive
OCTOBER 5, 2023
Kyowa will pay nearly $400 million to acquire Orchard, which sells the gene therapy Libmeldy in Europe but has struggled to find paths to market for other experimental treatments.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.
Rethinking Clinical Trials
OCTOBER 5, 2023
The GRACE trial, an NIH Pragmatic Trials Collaboratory Demonstration Project, was featured in a segment on CBS News Chicago. Co–principal investigator Ardith Doorenbos was interviewed for the segment as part of the news program’s observance of Sickle Cell Awareness Month. “Millions of people around the world with sickle cell disease really suffer from very serious chronic pain, which impacts all their life experiences,” Doorenbos said. “What the GRACE trial is doing is ta
Pharmaceutical Technology
OCTOBER 6, 2023
Ocular Therapeutix has confirmed the launch of a trial of an intravitreal eye implant intended to combat macular degeneration.

AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 5, 2023
Milk teas – a variety of sweet drinks including bubble tea – have become phenomenally popular in China and other parts of Asia in recent years, and a new study has highlighted some worrying links between the beverage and mental health issues.

Bio Pharma Dive
OCTOBER 3, 2023
The longtime partners believe that, by combining their technologies, they can create “in vivo” genetic medicines for nervous system and muscular disorders.

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.

Rethinking Clinical Trials
OCTOBER 4, 2023
In this Friday’s PCT Grand Rounds, David Murray of the NIH Office of Disease Prevention will kick off our special series, Advances in the Design and Analysis of Pragmatic Clinical Trials, with his presentation, “Hybrid Studies Should Not Sacrifice Rigorous Methods.” The session will be held on Friday, October 6, at 1:00 pm eastern and will be moderated by Jonathan Moyer.
Pharmaceutical Technology
OCTOBER 2, 2023
An NDA submission for EryDex is currently intended for Q4 2025, assuming positive Phase III study results.
AuroBlog - Aurous Healthcare Clinical Trials blog
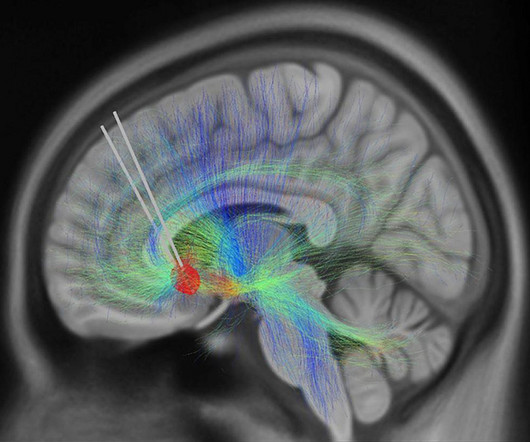
OCTOBER 4, 2023
It could soon be possible to measure changes in depression levels like we can measure blood pressure or heart rate. In a new study, 10 patients with depression that had resisted treatment were enrolled in a six-month course of deep brain stimulation (DBS) therapy.

Bio Pharma Dive
OCTOBER 4, 2023
The company is planning to start a Phase 3 trial of the vaccine this year, and is targeting a regulatory approval in 2025.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Pharmaceutical Commerce
OCTOBER 5, 2023
In an interview at LogiPharma USA 2023 with Pharma Commerce Editor Nicholas Saraceno, Tracy Nasarenko, Sr. Director of Community Engagement for Pharmaceuticals, GS1 US highlights her “DSCSA—Final Checks to Have Before Deadlines.

Pharmaceutical Technology
OCTOBER 3, 2023
Drug manufacturer Viatris has announced the divestment of a number of businesses owned by the company in a $.37bn deal.

AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 5, 2023
Advanced, portable and intuitive diagnostic tools are considerably benefiting the Indian healthcare to tackle diseases such as dengue and malaria. Easily transportable and convenient diagnostics systems are invaluable in remote areas, aiding timely diagnosis and patient care.

Bio Pharma Dive
OCTOBER 5, 2023
The company says Syfovre prescriptions accelerated again in August after a rocky summer of side effect probes.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
BioSpace
OCTOBER 1, 2023
After a groundbreaking year in the Alzheimer’s space, Parkinson’s disease researchers express renewed hope based on a greater biological understanding of neurodegeneration.

Pharmaceutical Technology
OCTOBER 4, 2023
Strides earned market approval in the US as part of a larger government endeavor to address HIV worldwide.

AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 4, 2023
The Union health ministry has issued a draft notification to include antiviral drugs oseltamivir and zanamivir into the Schedule H1 of the Drug Rules, 1945, allowing the retailers to store and sell the drug against prescription by maintaining a separate record for the details of the particular sales.
Bio Pharma Dive
OCTOBER 2, 2023
Silos abound in the pharma world, especially when it comes to customer care and call center support. Using AI can change everything, though. Here’s how.

Advertisement
Clinical research has entered a new era, one that requires real-time analytics and visualization to allow trial leaders to work collaboratively and to develop, at the click of a mouse, deep insights that enable proactive study management. Learn how Revvity Signals helps drug developers deliver clinical trial data insights in real-time using a fast and flexible data and analytics platform to empower data-driven decision-making.
Let's personalize your content