Seattle institute lands $18m funding to develop RNA vaccine for chikungunya
Pharmaceutical Technology
AUGUST 3, 2023
AAHI is developing a novel RNA vaccine that overcomes manufacturing and storage limitations to increase accessibility.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
AUGUST 3, 2023
AAHI is developing a novel RNA vaccine that overcomes manufacturing and storage limitations to increase accessibility.
Pharmaceutical Technology
MAY 11, 2023
Results published in Nature for a personalised pancreatic cancer vaccine that uses neoantigens from patients’ tumours have lent further support to early positive signals. The vaccine, developed by BioNTech, led to half of the patients with pancreatic cancer in the Phase I trial remaining cancer-free 18 months later.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
APRIL 16, 2024
Sanofi has signed an agreement with IDT Australia for preclinical formulation development of its messenger RNA (mRNA) vaccines.

Bio Pharma Dive
FEBRUARY 10, 2023
The pharma and partner BioNTech have kicked off the first human trial of a messenger RNA shot for shingles, believing there’s room to clear the high bar set by GSK’s rival vaccine.

Pharmaceutical Technology
MARCH 28, 2023
Following on from its Covid-19 vaccine programmes, BioNTech has set its sights on a range of infectious diseases for vaccine development. The company saw major successes with its Covid-19 vaccine, developed in collaboration with Pfizer. In response to the lower vaccine sales forecasts, BioNTech shares opened at 3.9%

Pharmaceutical Technology
AUGUST 17, 2022
Merck (MSD outside North America) has entered a partnership agreement with Orna Therapeutics for discovering, developing and marketing various programmes based on next-generation RNA technology. These programmes will include therapies and vaccines in infectious disease and oncology areas.
Bio Pharma Dive
AUGUST 26, 2022
The biotech claims its rivals’ vaccine Comirnaty, one of the world’s top-selling pharmaceutical products, infringes on two patents covering its messenger RNA technology.

Pharmaceutical Technology
DECEMBER 19, 2023
BioNTech has inaugurated a new messenger RNA (mRNA) vaccine manufacturing facility in Kigali, Rwanda, which will have BioNTainers.

Pharmaceutical Technology
AUGUST 1, 2022
GreenLight Biosciences has entered a partnership with the US National Institutes of Health (NIH) for the development of Covid-19 vaccines, which offer broader protection against new variants and with durable effects. They intend to develop vaccines that provide lasting immune responses compared to existing vaccines.
Pharmaceutical Technology
FEBRUARY 3, 2023
Only a few weeks into the new year, the prospect of getting a successful advanced HIV vaccine shrank after the discontinuation of yet another late-stage trial. On January 18, Janssen, a Johnson & Johnson (J&J) subsidiary, stated that its vaccine was not effective in preventing HIV infections.
Pharmaceutical Technology
AUGUST 1, 2022
Moderna has entered a new supply contract with the US Government to deliver 66 million doses of its Covid-19 vaccine booster candidate, mRNA-1273.222. The contract comprises a $1.74bn award to produce and supply these vaccine doses and options to further procure up to 234 million additional doses of the company’s booster candidates.
Pharmaceutical Technology
MARCH 1, 2023
The Covid-19 pandemic led to massive developments and scientific advances within the field of messenger ribonucleic acid (mRNA) vaccines. Pfizer /BioNTech and Moderna’s mRNA-based vaccines were the first to receive emergency use authorisation (EUA) out of all other Covid-19 vaccines under development in late 2020.
Pharmaceutical Technology
OCTOBER 14, 2022
German pharmaceutical firm Merck has extended its partnership with Moderna to jointly develop and sell mRNA-4157/V940, an investigational personalised cancer vaccine (PCV). In 2016, the companies entered a strategic partnership to develop novel messenger RNA (mRNA) based PCVs.
Pharmaceutical Technology
JUNE 1, 2023
A new intracellular drug delivery centre will be established in the UK to support potential ribonucleic acid (RNA) vaccines and therapeutics , as well as the development of innovative drug delivery technologies. It will focus on studying and developing new lipid nanoparticle (LNP) formulations for the delivery of RNA medicine.

Pharmaceutical Technology
MAY 23, 2023
ReNAgade Therapeutics has burst onto the RNA technology scene by announcing a $300m financing round. With its delivery system, Cambridge, Massachusetts-based biotech says it aims to “address major limitations in RNA therapeutics by enabling the delivery of RNA medicines to previously inaccessible tissues and cells in the body.”

Pharmaceutical Technology
AUGUST 2, 2022
Samsung Biologics and GreenLight Biosciences have completed the initial commercial-scale engineering run for their messenger ribonucleic acid (mRNA) Covid-19 vaccine under their manufacturing collaboration. Following the demonstration at Samsung, the clinical trial of GreenLight’s Covid-19 booster vaccine is anticipated to commence this year.
Pharmaceutical Technology
JULY 15, 2022
Health Canada has granted approval for the usage of Moderna’s messenger RNA (mRNA) Covid-19 vaccine, Spikevax, in a 25µg two-dose regimen for active immunisation to prevent Covid-19 in children aged six months to five years. So far, children aged below five years were not eligible to receive the Covid-19 vaccine in Canada.
Bio Pharma Dive
SEPTEMBER 17, 2020
Both companies believe the messenger RNA technology they're using in their coronavirus vaccines could be applied to create a better flu shot.

Pharmaceutical Technology
NOVEMBER 2, 2023
While things may have slowed down a little since the rapid approvals of the COVID-19 vaccines, the excitement around RNA-LNP is still going strong.

AuroBlog - Aurous Healthcare Clinical Trials blog
OCTOBER 16, 2022
The husband-and-wife team who co-founded BioNTech, the biotechnology company that partnered with Pfizer to develop an effective messenger-RNA (mRNA) shot against COVID-19, has predicted that a cancer vaccine could be widely available within the next decade.

pharmaphorum
NOVEMBER 29, 2023
Japan’s medicines regulator has approved the first vaccine based on self-assembling RNA, a spin on the mRNA technology used in current shots, which is designed to deliver greater efficacy with a lower dose.
Bio Pharma Dive
AUGUST 22, 2022
The decision will make a third vaccine available for children 12 to 17 years old in the U.S., giving them an option that doesn’t use the messenger RNA technology of shots by Pfizer and Moderna.

Roots Analysis
NOVEMBER 6, 2023
RNA-based therapeutics have completely revolutionized the healthcare segment, greatly influencing the study and treatment of human diseases by conferring precise targeting ability to therapeutic modalities. The below figure presents the distribution of next generation RNA-based therapeutics and RNA-based vaccines, based on type of molecule.
Pharmaceutical Technology
JUNE 30, 2022
HDT Bio has received Emergency Use Approval from Indian regulators for its Covid-19 vaccine, Gemcovac. The vaccine leverages self-amplifying RNA (saRNA), which can replicate itself after administration and could be effective at very low doses. HDT Bio CEO Steve Reed said: “Our saRNA vaccine is a game-changer.
Pharmaceutical Technology
FEBRUARY 15, 2023
In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Innovation in Pharmaceuticals: Flavivirus vaccine components. Sanofi is one of the leading patent filers for Flavivirus vaccine components.
STAT News
NOVEMBER 21, 2022
This story has been adapted from the STAT Report “The future of messenger RNA: Covid-19 vaccines are just the beginning.” Read the rest…
Pharmaceutical Technology
JUNE 8, 2023
Gritstone bio has reported interim results from an ongoing Phase 1 study evaluating the company’s self-amplifying mRNA (samRNA) vaccine as a boost against Covid-19 (CORAL-BOOST). The Phase I CORAL-BOOST dose-escalation trial (NCT05148962) evaluated Gritstone’s samRNA candidate (GRT-R910) in previously vaccinated healthy older adults.
Pharmaceutical Technology
MARCH 23, 2023
China’s National Medical Products Administration (NMPA) has granted emergency use authorisation (EUA) for CSPC Pharmaceutical Group’s messenger RNA (mRNA) vaccine, SYS6006, to treat Covid-19. The independently developed SYS6006 vaccine has been designed to target some major Omicron variants. efficacy after 14 to 28 days.
STAT News
NOVEMBER 24, 2022
An experimental influenza vaccine developed using messenger RNA technology appears capable of inducing what should be a protective immune response against all known subtypes of flu, at least in animals. If the work is translated into humans it could turn out to be a version of a long-sought universal vaccine.
Drug Discovery World
FEBRUARY 19, 2024
Replicate Bioscience has shared positive results from the Phase I trial of RBI-4000, its srRNA-based rabies vaccine. In this clinical trial, evaluating safety and immunogenicity of RBI-4000 , participants received one or two doses of srRNA vaccine at low doses (0.1mcg, 1mcg or 10mcg).
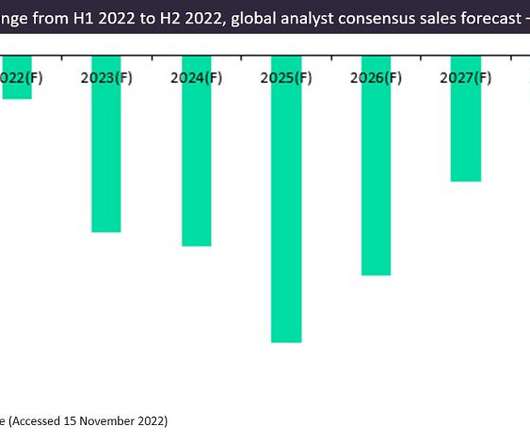
Pharmaceutical Technology
JANUARY 30, 2023
According to GlobalData’s Drugs database, there were 22 prophylactic vaccines in development for COVID-19 with sales forecasts available in H2 2022. Of these 22 vaccines, mRNA-derived vaccines dominate, with Comirnaty and Moderna’s Spikvax accounting for 88% of 2022 sales, with 58% and 30%, respectively.
Pharmaceutical Technology
APRIL 27, 2023
Orbital Therapeutics has raised $270m in a Series A round led by ARCH Venture Partners to advance a portfolio of programmable RNA therapeutics. Orbital will use the new funding to increase the application of RNA-based medicines for use in the fields of new vaccines, immunomodulation and protein replacement.

JAMA Internal Medicine
OCTOBER 3, 2021
Several recent case series have described acute myocarditis after COVID-19 messenger RNA (mRNA) vaccination. While the cardiac complications of vaccines are important, discussion has been limited by small sample sizes that lack gender and racial and ethnic diversity. 414 fully vaccinated individuals). women and 31.2%
Pharmaceutical Technology
NOVEMBER 30, 2022
in a project agreement from the US government for developing self-amplifying RNA (saRNA) vaccine technology against advanced and emergent viral threats. Development of vaccines to Phase I trials under the five-year $59m prototype project comprises additional $28.4m in milestone payments.
Bio Pharma Dive
AUGUST 16, 2022
The pharmaceutical company has formed an alliance with Orna and invested $100 million in its Series B round, hoping the young biotech’s approach could lead to multiple new vaccines and drugs.

Drug Discovery World
OCTOBER 2, 2023
This eBook sponsored by DNA Script explores the advances and breakthroughs taking place within vaccines and virology.

Bio Pharma Dive
MAY 19, 2023
The biotech, best known for its vaccine research, said its clinical trial is the first to report results of a messenger RNA therapeutic designed for intracellular protein replacement.
Pharmaceutical Technology
AUGUST 24, 2022
Mode rna has submitted an application to the US Food and Drug Administration (FDA) to obtain emergency use authorization (EUA) for mRNA-1273.222, its BA.4/BA.5 5 Omicron-targeting bivalent booster vaccine for Covid-19. The submission is made for a 50µg booster dose of the vaccine for usage in adults aged 18 years and above.

pharmaphorum
AUGUST 16, 2022
Merck & Co has ramped up its involvement in the RNA category, partnering with US biotech Orna Therapeutics in a deal valued at up to $3.5 billion, consummating a relationship that first started in 2018 and spanned a range of mRNA-based vaccines for infectious diseases. billion, including $150 million upfront.
STAT News
JULY 19, 2022
An expert panel that advises the Centers for Disease Control and Prevention on vaccine policy voted Tuesday to recommend use of Novavax’s Covid-19 vaccine — an achievement a long time in the making for the Gaithersburg, Md.-based based company.

BioPharma Reporter
DECEMBER 6, 2021
Australiaâs Noxopharm has in-licensed novel RNA tech developed by Hudson Institute of Medical Research: focusing on RNA drug discovery and mRNA vaccine manufacture via Noxopharmâs subsidiary Pharmorage.

Medical Xpress
DECEMBER 30, 2022
Messenger RNA vaccine technology, once an arcane area of research, became household terminology because of the COVID-19 pandemic, and now scientists are working on an mRNA flu vaccine strategy that, at least in this study, involves a first dose administered as a shot but a booster administered as an mRNA nasal spray.

XTalks
MARCH 21, 2022
Credited as a pioneering RNA interference (RNAi) therapeutics company, Alnylam Pharmaceuticals is taking Pfizer and Moderna to court, claiming that the companies’ use of the lipid nanoparticle (LNP)-based RNA delivery technology in their mRNA COVID-19 vaccines infringes on a patented technology.

STAT News
JANUARY 19, 2023
And do normal people care about messenger RNA? We also discuss the latest news in the life sciences, including new vaccines for a vexing infection and the future of mRNA. Where do new drugs come from? Does science need billionaires? We cover all that and more this week on “The Readout LOUD,” STAT’s biotech podcast.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content