Tourmaline shifts focus to developing anti-IL6 antibody following merger
Pharmaceutical Technology
OCTOBER 20, 2023
The company plans to initiate the Phase II trial of anti-IL-6 antibody in atherosclerotic cardiovascular disease in 2024.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
OCTOBER 20, 2023
The company plans to initiate the Phase II trial of anti-IL-6 antibody in atherosclerotic cardiovascular disease in 2024.

Pharmaceutical Technology
JANUARY 9, 2023
Eisai and Biogen have received approval for their antibody Leqembi (lecanemab-irmb) , 100mg/mL injection for intravenous use, from the US Food and Drug Administration (FDA) under the Accelerated Approval Pathway to treat Alzheimer’s disease (AD). The regulatory approval is based on the data obtained from the Phase II trial.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Drug Discovery World
MAY 1, 2024
DDW’s Megan Thomas speaks to industry experts and thought leaders about the potential for therapeutic antibodies in 2024. Many of the best-selling drugs in 2023 were monoclonal antibody (mAB) therapies, including Keytruda (pembrolizumab), Humira (adalimumab), and Dupixent (dupilumab) 1.

Bio Pharma Dive
MARCH 25, 2024
The agency wants Regeneron to make more progress with a confirmatory trial before clearing odronextamab, a “bispecific” antibody being developed for multiple blood cancers.

Pharmaceutical Technology
MAY 8, 2023
Ei s ai and Bliss Biopharmaceutical have entered into a joint development agreement for antibody-drug conjugate (ADC), BB-1701, to treat cancers. The collaboration will help in further development of BB-1701 across the world and advance the ADC towards late stage of development.

Pharmaceutical Technology
JULY 21, 2022
AstraZeneca has signed a deal with the Federal Office of Public Health (FOPH) of Switzerland to deliver over 1,200 doses of antibody therapy, tixagevimab and cilgavimab combination (AZD7442), for Covid-19 prevention and treatment. In May, the company supplied the first doses of the antibody therapy to Swiss hospitals for Covid-19 prevention.
Pharmaceutical Technology
APRIL 14, 2023
Clinical-stage biopharmaceutical company TORL BioTherapeutics has raised $158m in a Series B financing round for advancing the development of new biologics for cancer treatment. TORL BioTherapeutics is a newly formed, US-based company focused on the development of novel antibody-based therapeutics for cancer patients.

Drug Discovery World
MARCH 8, 2024
Biopharmaceutical company Innovent Biologics has launched the first-in-human (FIH) Phase I clinical trial of IBI3002, a novel bispecific antibody targeting Interleukin 4 receptor α (IL-4Rα) and thymic stromal lymphopoietin (TSLP). In vitro assays have shown superiority over the marketed monoclonal antibodies to respective target.
Drug Discovery World
NOVEMBER 14, 2023
DDW’s Megan Thomas looks at how different diseases will benefit from the success of therapeutic antibodies. Cancer Monoclonal antibodies (mAbs) are a type of targeted drug therapy, and as such are often used to treat cancer. This is not only made clear by the fact that the global cancer mAbs market size was valued at $55.6
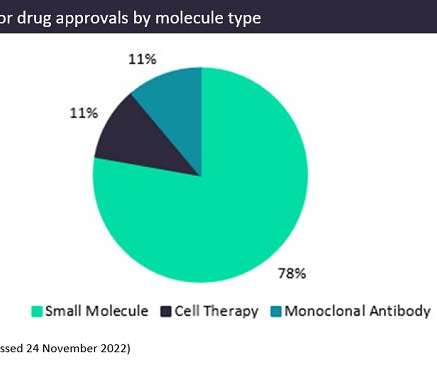
Pharmaceutical Technology
DECEMBER 6, 2022
The three leading Alzheimer’s disease (AD) drugs currently in development are all monoclonal antibodies—donanemab by Eli Lilly , lecanemab by Eisai and Biogen , and gantenerumab by Roche. Monoclonal antibodies make up half of these, with an 11% share of all AD approvals.

BioPharma Reporter
AUGUST 1, 2023
Epsilogen, a leader in the development of immunoglobulin E (IgE) antibodies to treat cancer, has published phase 1 data from the first ever clinical trial of an IgE antibody therapeutic in Nature Communications.
BioPharma Reporter
NOVEMBER 9, 2023
The proof-of-concept trial, dubbed IRIS-RA, is the first to investigate a treatment for rheumatoid arthritis that blocks a protein called neonatal Fc receptor (FcRn).
Drug Discovery World
OCTOBER 12, 2023
The US Food and Drug Administration (FDA) has granted clearance to Transcenta to proceed with its TranStar 301 global Phase III pivotal trial of Osemitamab (TST001) for gastric cancer. The trial will investigate the drug in combination with Nivolumab and chemotherapy as a first-line treatment in patients with HER2-negative, CLDN18.2
Drug Discovery World
JANUARY 3, 2023
A monoclonal antibody to prevent Covid-19 in vulnerable patients, AZD3152, has entered clinical trials less than 12 months after discovery. . The commencement of the trial triggers a $5 million milestone payment to RQ Bio and will enable the company to execute its four scientific programmes focused on viral diseases. .

Drug Discovery World
MARCH 12, 2024
Here, experts weigh in on the future of monoclonal antibodies (mAbs). Amanda Halford, President Bioprocess, Cytiva “In 2024, the emphasis will remain on novel mAbs, specifically antibody drug conjugates. Innovations include next-generation antibodies with improved efficacy and reduced side effects.
Drug Discovery World
MARCH 10, 2023
Cancer Research UK and UCB have agreed a clinical development collaboration to advance two of UCB’s investigational oncology antibody candidates through clinical trials. If successful in clinical trials the investigational candidates may have the potential to offer cancer patients access to new targeted treatment options.

Pharmaceutical Technology
FEBRUARY 14, 2023
Precision oncology firm Corbus Pharmaceuticals and CSPC Megalith Biopharmaceutical have entered an exclusive licensing agreement for the latter’s new clinical stage antibody drug conjugate (ADC), CRB-701 (SYS6002). We look forward to working with CSPC to advance clinical development of this asset and realise its full potential.”
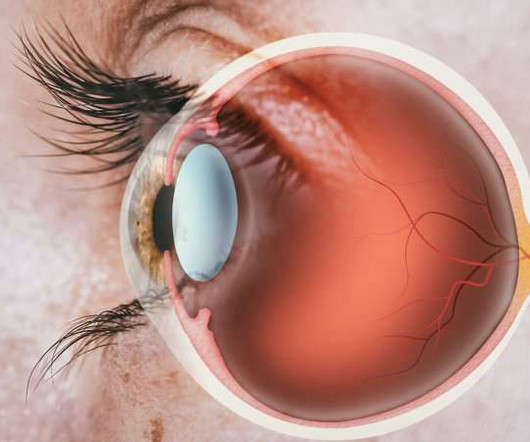
Drug Discovery World
OCTOBER 11, 2023
Collaborators Boehringer Ingelheim and CDR-Life have commenced a Phase I evaluation of BI 771716, their antibody fragment-based treatment developed to preserve the vision of people with geographic atrophy (GA). The company’s first therapeutic candidate, CDR404, targets MAGE-A4 and is expected to enter the clinic in 2024.
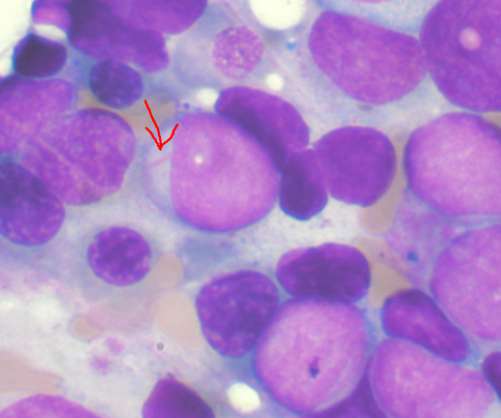
Pharmaceutical Technology
NOVEMBER 22, 2022
Umoja Biopharma has signed a research agreement with IASO Biotherapeutics (IASO Bio) to develop off-the-shelf therapies for haematological malignancies. The post Umoja and IASO partner to develop therapies for haematological malignancies appeared first on Pharmaceutical Technology.
Drug Discovery World
APRIL 14, 2023
Oliver Schon, PhD, is VP Research & Development at BiVictriX Therapeutics, a UK-based emerging drug discovery and development company generating a pioneering first-in-class next-generation ADC anti-cancer therapeutics which exhibit superior selectivity towards cancer.

Pharmaceutical Technology
OCTOBER 25, 2022
Ast ellas Pharma has announced plans to make a strategic investment to back the development of Taysha Gene Therapies’ adeno-associated virus (AAV) development programmes for Rett syndrome and giant axonal neuropathy (GAN). This would allow the companies to boost the development of new therapy options for Rett syndrome and GAN patients.
Pharmaceutical Technology
APRIL 20, 2023
The funds will support Maxion’s KnotBody platform to develop antibodies to treat autoimmune diseases. Antibody-based therapies provide enhanced efficacy and safety whilst reducing the need for frequent administration for autoimmune disease treatment.

BioPharma Reporter
DECEMBER 13, 2021
Memo Therapeutics AG will receive CHF 10.5m (US $11.4m) from the Swiss government to clinically develop a SARS-CoV-2 antibody against COVID-19, allowing it to start Phase 1 studies in Q1, 2022.
Drug Discovery World
JULY 6, 2023
The first patient has been dosed in the clinical Phase I evaluation of the Sema3A monoclonal antibody, part of the Evotec-Bayer multi-target research collaboration in kidney diseases. Sema3A is upregulated in injured human kidneys and implicated in the development and progression of acute and chronic kidney diseases.

Pharmaceutical Technology
SEPTEMBER 16, 2022
The US Food and Drug Administration (FDA) has granted fast track designation to Jasper Therapeutics’ antibody, JSP191, for the treatment of severe combined immunodeficiency (SCID) patients who undergo allogeneic hematopoietic stem cell transplant. . The antibody was also analysed in a total of more than 110 healthy subjects and patients. .

Drug Discovery World
JUNE 1, 2023
Antibody-drug conjugates (ADCs) are biopharmaceutical products in which a monoclonal antibody (mAB) is linked to a small molecule drug with a stable linker 1. According to NCBI, most of the ADCs developed so far are for use in the treatment of cancer, but there is plenty of potential for using ADCs to treat other diseases 1.
Pharmaceutical Technology
APRIL 13, 2023
Alentis Therapeutics intends to use the funding for the Phase II and Phase I programmes of its lead investigational products ALE.F02 and ALE.C04, first-in-class anti-Claudin-1 (CLDN1) antibodies, respectively. With a silenced effector function, ALE.F02 has been designed for targeting the stiff ECM of fibrotic tissues and organs.

Drug Discovery World
APRIL 27, 2023
Biotechnology company Tiziana Life Sciences is initiating a programme to develop intranasal foralumab for the treatment or prevention of type 1 diabetes. We believed that having a fully human CD3 antibody may offer benefits to patients across many different chronic disease indications.”

XTalks
DECEMBER 28, 2023
BillionToOne, a company specializing in molecular diagnostics, has partnered with Janssen Research & Development, LLC, a Johnson & Johnson division, to conduct the AZALEA Phase III clinical trial globally. The trial is set to occur across the US, the European Union (EU) and selected global locations.

pharmaphorum
MARCH 26, 2021
Regeneron has posted findings from a large trial of its COVID-19 antibody cocktail showing the therapy reduced risk of hospitalisation or death by 70%. Data from the phase 3 trial will also be used to convert an Emergence Use Authorisation into a permanent licence, Regeneron said.
Pharmaceutical Technology
APRIL 28, 2023
Biotechnology company Therini Bio has raised $36m in a Series A financing round to develop fibrin-targeted therapies to treat neurodegenerative and retinal diseases. Therini Bio will use part of the funds to advance its potential first-in-class investigational antibody, THN391, into clinical trials.
Drug Discovery World
NOVEMBER 15, 2022
DynamiCure has dosed the first patients in its Phase I clinical trial evaluating the first of the company’s two anti-CD93 monoclonal antibody candidates, DCBY02, in adults with a wide range of advanced cancers. . The post Phase I trial of lead antibody candidate launched appeared first on Drug Discovery World (DDW).
Pharmaceutical Technology
APRIL 26, 2023
Biotechnology company Adcentrx Therapeutics has raised $38m in Series A+ financing to advance its pipeline of new antibody-drug conjugate (ADC) therapeutics into clinical development. Adcentrx Therapeutics is focused on developing protein conjugate therapeutics to treat cancer and other life-threatening diseases.

pharmaphorum
SEPTEMBER 16, 2020
Regeneron’s COVID-19 antibody cocktail has been added to the UK’s RECOVERY trial, one of the largest of its kind in the world that is testing several potential coronavirus therapies at the same time. REGN-COV2 is a combination of two monoclonal antibodies, designed to block infections of SARS-CoV-2, the virus that causes COVID-19.

Roots Analysis
OCTOBER 25, 2023
On 23 October 2023, Merck announced a development agreement for Daiichi Sankyo’s clinical-phase antibody-drug conjugate, namely patritumab deruxtecan (HER3-DXd), ifinatamab deruxtecan (I-DXd), and raludotatug deruxtecan (R-DXd), targeting cancer. Merck has invested $5.5
Drug Discovery World
JULY 26, 2023
A Cancer Research UK-funded clinical trial has shown, for the first time, that a new class of antibody could benefit cancer patients whose existing treatments have stopped working. The drug, MOv18 IgE, was developed by researchers at King’s College London.
Drug Discovery World
DECEMBER 8, 2023
ONK Therapeutics and NAYA Biosciences have agreed to develop a combination therapy of ONK’s optimally-engineered natural killer (NK) cell therapies and NAYA’s FLEX-NK bispecific antibodies. The post Partners explore NK cell therapy and bispecific antibodies combination appeared first on Drug Discovery World (DDW).
pharmaphorum
JANUARY 15, 2021
South Korea’s Celltrion has announced encouraging top-line results for its potential antibody therapy for COVID-19, with the first part of a phase 2/3 trial showing it could cut recovery times and chances of progression from moderate to more severe disease. days shorter than placebo, the company said. reported with placebo.

Pharmaceutical Technology
JUNE 9, 2023
Upstream Bio will use the funds to advance its UPB-101 into Phase II trials to treat asthma and chronic rhinosinusitis with nasal polyps (CRSwNP). It is also preparing for a swift transition to Phase III development. It targets the human thymic stromal lymphopoietin receptor to inhibit signalling.

XTalks
JANUARY 17, 2024
Estimates based on publicly available information suggest more than 40 percent of all new therapies in development are cancer treatments. Given this hotbed of activity, innovation in the space to drive faster decisions and more efficient trials is intense.
Pharmaceutical Technology
NOVEMBER 29, 2022
Researchers at City of Hope in the US have developed a new potential therapy to treat glioblastoma multiforme (GBM), a kind of brain cancer. This chemokine is connected to the epidermal growth factor receptor (EGFR) antibody causing attachment to tumour cells. These viruses are engineered genetically for killing cancer.
BioPharma Reporter
AUGUST 11, 2020
Regeneron Pharmaceuticals aims to post initial virology and biomarker data from clinical trials of its COVID-19 antibody cocktail next month.
Pharmaceutical Technology
AUGUST 16, 2022
Q32 Bio has signed a partnership and option agreement with Horizon Therapeutics for developing ADX-914 to treat autoimmune ailments. According to the deal, Horizon will provide the funding to develop the therapy until the conclusion of its two Phase II clinical trials. Q32 will also receive tiered royalty payments on net sales.

pharmaphorum
JUNE 24, 2021
Armed with a positive mid-stage trial readout in lung cancer, Arcus Biosciences will expand a phase 3 programme for domvanalimab, its anti-TIGIT antibody. That came a few weeks after Bristol-Myers Squibb paid $200 million upfront for rights to AGEN1777, a TIGIT drug developed by Agenus in a deal that could be worth up to $1.56
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content