Pfizer seeks approval from US FDA for Covid-19 treatment
Pharmaceutical Technology
JULY 1, 2022
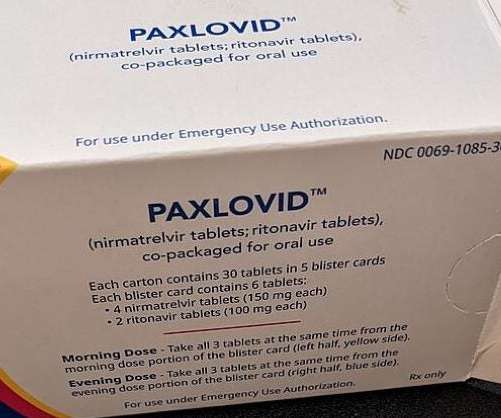
The latest filing is based on non-clinical and clinical data for Paxlovid and also comprised data from the Phase II/III EPIC-HR clinical trial. Furthermore, findings from the final trial report showed a relative risk decline of 86%.










































Let's personalize your content