Changing cell & gene therapy landscape and implications on clinical trials
Bio Pharma Dive
APRIL 3, 2023
Cell & gene therapy clinical research may help treat diseases and change how we approach healthcare.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Bio Pharma Dive
APRIL 3, 2023
Cell & gene therapy clinical research may help treat diseases and change how we approach healthcare.

XTalks
MAY 17, 2023
The Foundation for the National Institutes of Health (FNIH) announced this week that the Accelerating Medicines Partnership Bespoke Gene Therapy Consortium (AMP BGTC) has selected eight rare diseases for its clinical trial portfolio.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

BioPharma Reporter
DECEMBER 8, 2022
Emmes has announced the creation of a dedicated center for cell and gene therapy research: which will focus on supporting clinical trials for the Clinical Research Organizationâs (CRO) clients.

XTalks
JANUARY 17, 2024
Changes in what clinical performance data is used to inform the trial design and site selection, new approaches to patient recruitment and the rise of advanced treatments such as cell and gene therapy are rewriting the script. Vidal shared their insights from the 2023 ESMO conference which occurred in Madrid.

XTalks
JUNE 8, 2023
Over the past few years, there has been a significant expansion in the cell and gene therapy landscape, with an increasing number of therapies entering clinical trials and receiving regulatory approvals. Another integral element is timeline management.

XTalks
APRIL 24, 2023
Awareness of rare diseases is growing, and with a better understanding of the pathophysiology of many rare diseases, innovative treatment options are emerging, like gene therapies that can treat the root cause of rare genetic diseases and potentially provide long-term symptom relief, or even a definitive cure.

Worldwide Clinical Trials
JANUARY 24, 2024
Much of what we discuss I covered in my presentation, “Leveraging Genetics to Support Rare Disease Clinical Trials,” at last year’s World Orphan Drug Congress (WODC) EU. In my capstone, I focused on how, if, and when genetic counselors discuss clinical trials with their patients in the context of a counseling session.

Drug Discovery World
DECEMBER 13, 2022
Clinical research organisation (CRO) Emmes has unveiled a dedicated centre to expand its cell and gene therapy research. . The new centre will offer research services in clinical trial operations, study leadership, statistical services, data management, and pharmacovigilance. .

Drug Discovery World
DECEMBER 8, 2023
Andrew Busey , Co-Founder, Form Bio discusses why vertical AI is set to reshape the cell and gene therapy sector. The cell and gene therapy industry is no exception to this digital transformation. Here’s what this means for pharmaceutical leaders and patients.

Cloudbyz
MARCH 22, 2024
In the realm of clinical research, the efficient management of data is paramount. However, traditional methods of data entry, particularly from diverse and unstructured documents, have long posed challenges for researchers, leading to inefficiencies, errors, and compliance risks. Scalability: Cloudbyz’s ClinExtract.AI
Cloudbyz
FEBRUARY 29, 2024
AI-enabled data extraction holds immense promise in expediting case processing within clinical trials, offering a transformative approach to streamline workflows and enhance efficiency. In this blog, we delve into the potential of AI in pharmacovigilance and its implications for clinical trial safety monitoring.

Velocity Clinical Research
OCTOBER 4, 2023
Velocity Clinical Research, the leading multi-specialty clinical sites business, announced it is opening greenfield clinical research sites in Bristol, Leicester, and Romford, demonstrating the company’s commitment to the U.K. The sites launch at a time when clinical trial activity in the U.K.

Camargo
JULY 30, 2021
Barry Mangum talks the importance of pediatric research, its evolution over the years, and the obstacles and opportunities that the industry faces as it moves forward. How long have you been in pediatric clinical research, and how did you enter the field? So I’ve been working in that space about 43 years.
Advarra
NOVEMBER 29, 2022
The field of cell and gene therapies (CGT) is constantly evolving, and there has been significant progress in this area of research. However, despite the promise of these therapies, the regulations governing them lag the science, which in turn hinders the clinical translation of these novel medicines.
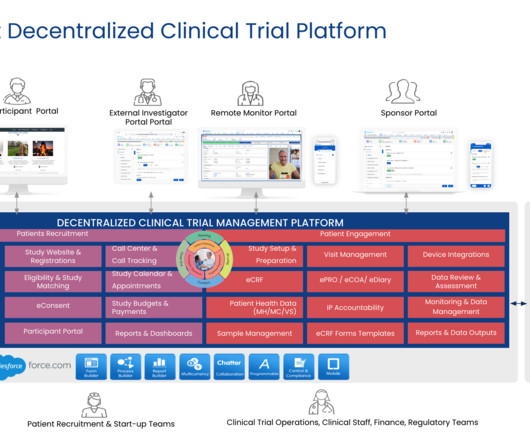
Cloudbyz
JUNE 11, 2023
This transformation has permeated the domain of clinical trials, which is now embracing the concept of decentralized clinical trials (DCTs). This paper delves deep into the universe of DCTs, their distinctiveness from conventional trials, their inherent benefits, existing hurdles, and measures to bolster their adoption.

ACRP blog
SEPTEMBER 27, 2023
Food and Drug Administration’s (FDA’s) Rare Disease Endpoint Advancement (RDEA) Pilot Program , the clinical research enterprise gains new support for efforts aimed at efficacy endpoint (or clinical outcome) development and the timely approval of drugs and biological products that treat rare diseases, including rare diseases in children.
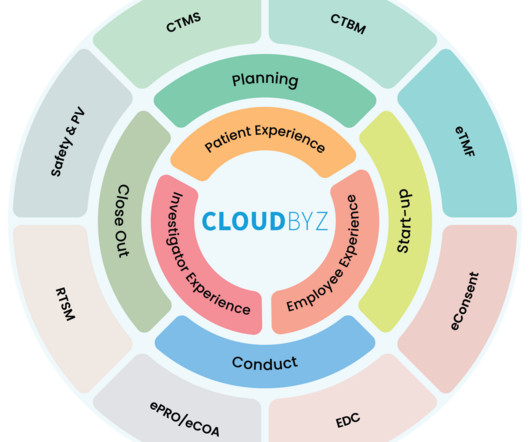
Cloudbyz
AUGUST 9, 2023
The clinical research landscape is rapidly evolving. As it becomes more complex with growing volumes of data, evolving regulations, and the pressure for faster drug development, traditional methods of clinical research management are no longer sufficient. This is where the platform approach comes into play.
Cloudbyz
MAY 19, 2021
Introduction Cell and gene therapy is an upcoming wave of therapeutic innovation in the healthcare and life sciences industry and is being pragmatically accepted worldwide. The gene therapy market reported its first market approvals back in 2017 and the evolution has been extensive ever since.
Worldwide Clinical Trials
NOVEMBER 27, 2023
In recognition of the unmet need and medical urgency for innovative therapies in the sickle cell space, the FDA granted exa-cel Priority Review, with a formal decision expected by December 8, 2023. Why is Casgevy a Breakthrough Gene Therapy for Patients with Sickle Cell Disease?
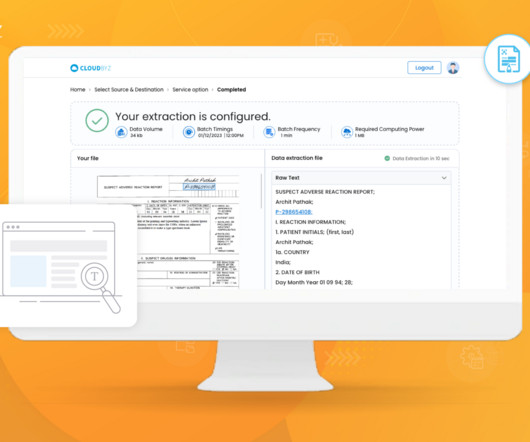
Cloudbyz
JUNE 26, 2023
Generative artificial intelligence (AI) techniques are increasingly being applied in clinical research to enhance various aspects of medical data analysis, diagnosis, treatment planning, and drug discovery. Clinical Trial Design and Optimization: Generative AI can aid in the design and optimization of clinical trials.

XTalks
OCTOBER 18, 2022
A clinical research organization (CRO) is a company that delivers outsourced services to plan, manage and execute clinical trials for biotechnology, pharmaceutical, and medical device companies. Read on to learn why life science and clinical research professionals are choosing a career with Medpace.
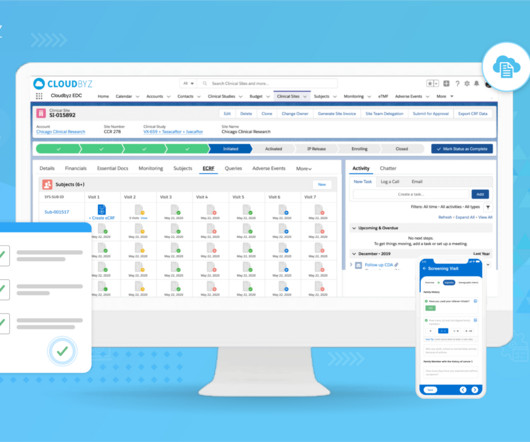
Cloudbyz
JUNE 10, 2023
21 CFR Part 11 is a crucial component of regulatory compliance in clinical trials and, more broadly, any industry subject to the regulations of the U.S. This blog post will serve as a comprehensive guide to understanding and implementing 21 CFR Part 11 compliance in your clinical trials. Let’s explore.

XTalks
MAY 20, 2021
Today is Clinical Trials Day 2021 — a day for recognizing the importance of clinical research in protecting and improving global health. Amid the alarmist headlines posted by other media outlets, Xtalks has remained balanced in its reporting of COVID-19 clinical trial results and safety data.

WCG Clinical
JULY 11, 2023
As countries across the globe reeled from the impact of COVID-19, the clinical research industry found itself in a new era working to understand and navigate the impact and to quickly find a treatment. We can apply this level of rigor and collaboration to your current or upcoming trial.

Worldwide Clinical Trials
MAY 20, 2022
The past few years we’ve had exciting opportunities to work on more cell and gene therapy programs for our sponsors. The industry is just at the tip of the iceberg for exploring the potential of these therapies in revolutionizing how we treat disease, and as the approvals start to trickle in, the pipeline only grows.

Worldwide Clinical Trials
OCTOBER 24, 2023
Seeking relief at the genetic cause of disease Engaged discussion with the audience followed my joint presentation on this topic with Matthew Fuller, PhD, PMP, Executive Director of Gene Therapy Research at Ultragenyx. The post 3 Key Takeaways from Global Genes Summit 2023 appeared first on Worldwide Clinical Trials.

Advarra
MARCH 31, 2023
The use of engineered genetic materials in clinical trials is rapidly expanding, with potential applications for genetic vaccines, gene-modified cellular therapies, and gene therapies. The post Risk Assessment for use of Engineered Genetic Materials in Clinical Research appeared first on Advarra.

Drug Discovery World
MARCH 10, 2023
Perhaps most interestingly this week, after previous caution over in vivo gene therapy trials, the FDA has given the go ahead to Intellia Therapeutics to study NTLA-2002 in hereditary angioedema (HAE). News round-up for 6-10 March by DDW Digital Content Editor Diana Spencer.

Drug Discovery World
FEBRUARY 10, 2023
There were two significant UK regulatory approvals based on key Phase III trial data, while an unusual ‘non-interventional’ study into geographic atrophy enrolled its first patient. The company’s global analysis of all clinical trials conducted in 2022 showed that Covid-19 came second.

pharmaphorum
JANUARY 27, 2023
Clinical trials design and patient input The definition of patient centricity, in fact, and its benefits are now – finally – being defined by patients themselves. “We We talk a lot in clinical trials and drug development about benefit,” Dr Mullen said. But who is benefitting?

Drug Discovery World
FEBRUARY 12, 2024
We’re already seeing biotechs making breakthroughs with AI in this area, with more than 150 small molecule drugs discovered and 15 already in clinical trials. Matt Todd, Head of Architecture, Ori Biotech “We expect AI and data analytics to support pivotal advances in cell and gene therapy (CGT) manufacturing.

XTalks
JULY 6, 2023
Related: Bespoke Gene Therapy Consortium Selects 8 Rare Diseases for Clinical Trial Portfolio “The approval of Ngenla will be significant for children with growth hormone deficiency in the US. The featured speakers will discuss clinical trial site burden, and CROs and the transformative potential of technology.

XTalks
JANUARY 8, 2024
Whether it’s for a treatment for a chronic ambulatory condition, precision medicine or cell and gene therapy, there is a massive uptick in clinical trial complexity. The key considerations she shared include: Project management: The expertise and approach of the team managing the trial.

ACRP blog
APRIL 5, 2023
AI is increasingly important in drug discovery and development as well as clinical trials, operations, pharmacovigilance, and many other areas.” This has led to the emergence of a new ‘pharmaceutical intelligence’ that has allowed the industry to move away from traditional, slow-moving, and costly processes,” Lyons writes. “AI

Advarra
MARCH 10, 2023
Considered a rare disease, research in finding better therapies – and possibly even a cure – for SCD may be competing for funding with conditions impacting broader populations. SCD’s significant impact on African Americans also means it predominantly impacts a minority community historically underserved by clinical research.

Drug Discovery World
DECEMBER 20, 2022
There will be greater emphasis on diversity in clinical trials. A focus on the representation of diverse patient populations in clinical trials will be a very good thing for the industry, but could be challenging. DDW’s Diana Spencer summarises the key trends likely to impact the market in 2023. .
XTalks
MAY 17, 2024
Philipson’s transition to the biotech sector began with a position at UK-based biotech Trizell, where his efforts culminated in a gene therapy FDA submission for bladder cancer. The company also has a Phase II trial looking at setanaxib in patients with head and neck cancer. Can they do it?
XTalks
MAY 9, 2024
XTALKS CLINICAL EDGE: Issue 2 — Genmab’s Interview Xtalks Clinical Edge is a magazine for clinical research professionals and all who want to be informed about the latest trends and happenings in clinical trials. Celebrating 25 years of innovation is a significant milestone.

Drug Discovery World
JANUARY 26, 2024
In the first case of its kind, gene therapy has restored hearing in a patient in the United States. “The AK-OTOF-101 Clinical Trial and AK-OTOF-NHS-002 Natural History Study demonstrate the power of international collaboration in the development of new medicines for rare genetic conditions.”

XTalks
MARCH 13, 2023
The US Food and Drug Administration’s (FDA) Office of Tissues and Advanced Therapies (OTAT) held a recent town hall where three experts from the regulator provided guidance on how to design and conduct gene therapy clinical trials for rare diseases. It’s a very exciting time in gene therapy.
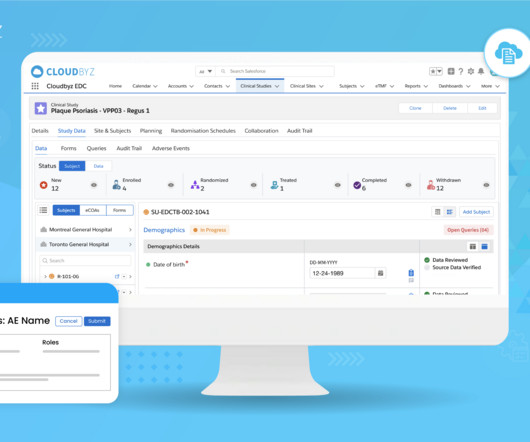
Cloudbyz
JUNE 21, 2023
In the world of clinical research, Electronic Data Capture (EDC) systems play a pivotal role in efficiently collecting, managing, and analyzing clinical trial data. The Study Data Tabulation Model (SDTM) is a widely accepted format that ensures consistency and interoperability of clinical trial data.

WCG Clinical
SEPTEMBER 14, 2023
Patient selection can be a nuanced process in clinical trials, particularly those involving therapeutic areas with more complex or subjective inclusion/exclusion criteria. Don’t navigate complex patient selections alone.

Pharmaceutical Technology
OCTOBER 26, 2022
The biotherapeutics market is rapidly growing, with 2021 seeing the highest-ever cell and gene therapy approval number. Gene therapy uses DNA to manipulate cells and correct defective genes, whereas cell therapy is the infusion or transplantation of cells into a patient.

Advarra
JANUARY 11, 2024
The applications of mRNA-based therapies in cancer research represent one of the next groundbreaking steps toward improved cancer treatments. While mRNA usage has played several roles in clinical research , oncology researchers in particular are eager to explore the possibilities of mRNA-based cancer vaccines.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content