CBD Research: A Dive into the Regulations of Cannabis Research
Advarra
JULY 26, 2022
However, it’s not legal federally and is considered a Schedule I drug by the U.S. Drug Enforcement Administration (DEA), meaning it has no accepted medical use and a high potential for abuse. Even though CBD doesn’t meet criteria for IND exemption or the FDA’s OTC drug review, it still can be studied in humans.








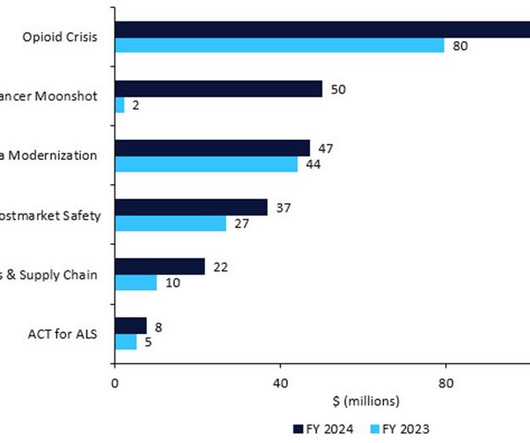






































Let's personalize your content