Accelerating recombinant protein vaccine discovery
Drug Discovery World
FEBRUARY 14, 2024
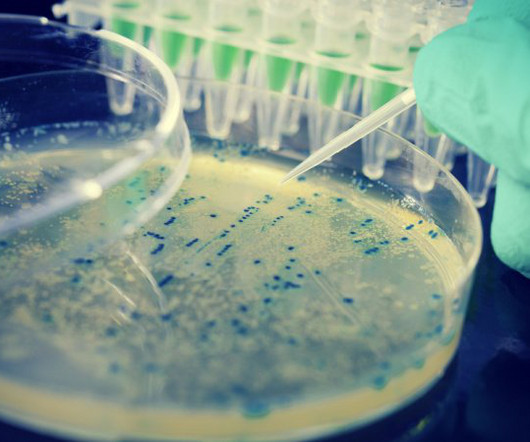
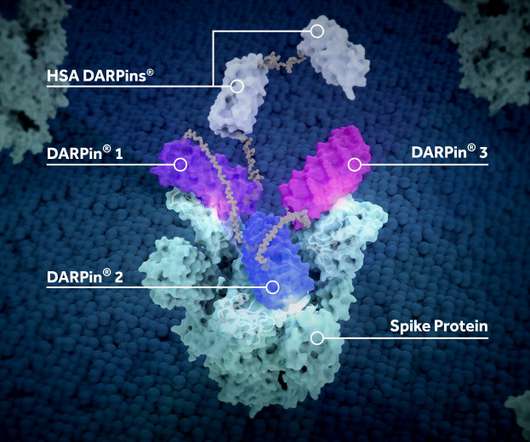
Vaccination responses need to be rapid, efficacious and cost-effective if they are to outpace new viral threats emerging across the globe, says Jian He (Jason) , Chemistry, Manufacturing and Controls (CMC) Head, WuXi Vaccines. This would give more people much faster access to more effective vaccines.























Let's personalize your content