The Use of Antipsychotic Drugs in Nursing Homes
NY Times
SEPTEMBER 16, 2021
A professor, a psychiatrist and a nurse react to a Times investigation. Also: A modern Civil War; a more confident child.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
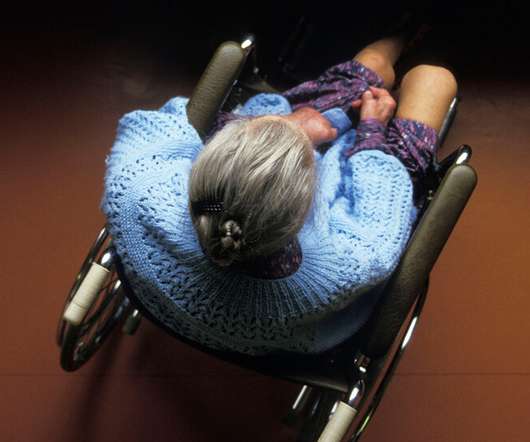
NY Times
SEPTEMBER 16, 2021
A professor, a psychiatrist and a nurse react to a Times investigation. Also: A modern Civil War; a more confident child.
The Pharma Data
DECEMBER 1, 2020
1, 2020 — Health care workers and people in nursing homes should be at the front of the line for upcoming COVID-19 vaccines, a U.S. In 2021, five to 10 million doses of vaccine are anticipated to ship each week. Food and Drug Administration hasn’t yet authorized the two leading vaccines for emergency use.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

NY Times
DECEMBER 18, 2020
The Food and Drug Administration authorized a second coronavirus vaccine for emergency use, clearing the way for millions more Americans to be immunized next week.

NY Times
JANUARY 21, 2021
A monoclonal antibody protected residents and staff members in facilities where the virus had appeared.

NY Times
JANUARY 21, 2021
A monoclonal antibody protected residents and staff members in facilities where the virus had appeared.

World of DTC Marketing
NOVEMBER 27, 2020
SUMMARY: Business by press release has made pharma executives a lot of money but we still haven’t seen any over reviewed data on COVID 19 vaccines. The American Nurses Association, a national professional organization, said one-third of its members do not intend to take the vaccine, and an additional third are undecided.
The Pharma Data
DECEMBER 2, 2020
Britain Approves Emergency Use of Pfizer COVID-19 Vaccine. 2, 2020 — Britain became the first Western country to allow emergency use of a COVID-19 vaccine on Wednesday after approving the Pfizer vaccine candidate in the race to inoculate millions of people around the globe. WEDNESDAY, Dec. The New York Times Article.

NY Times
MARCH 26, 2021
‘This job makes me feel like I’m part of history.’.

World of DTC Marketing
AUGUST 9, 2021
Doximity was flagged for having anti-vaccine information. The best approach is to use social media to connect HCPs to clincial information and drug trials. million other healthcare professionals worldwide, including medical students, residents, nurses, and more,45 Medscape is the largest news site for HCPs around the world, .
The Pharma Data
DECEMBER 12, 2020
Food and Drug Administration approved Pfizer’s coronavirus vaccine for emergency use on Friday, clearing the way for the launch of a national campaign to inoculate enough Americans to stem the spread of COVID-19. ” Who is first in line to be vaccinated? The nursing home industry applauded the approval.
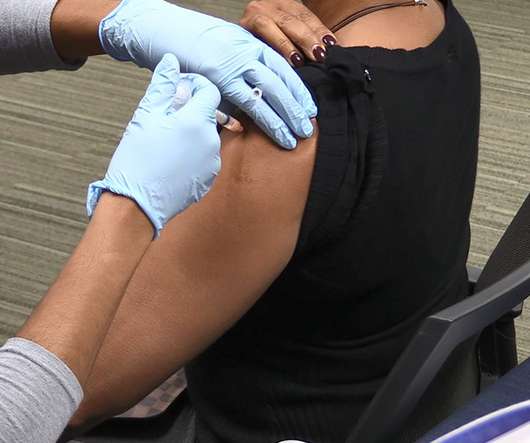
XTalks
JULY 30, 2020
The research provides an even more compelling reason to get vaccinated for those on the fence about the shot’s effectiveness at preventing the seasonal flu. These groups should have the highest vaccination rates because they are the most at risk; however, our findings show the opposite – flu vaccinations are under-utilized,” said Roshni A.

XTalks
FEBRUARY 3, 2021
COVID-19 vaccines have been a topic of conversation since the outbreak began, and last year’s US Food and Drug Administration (FDA) approval of the Pfizer/BioNTech and Moderna vaccines have given the world hope to defeat the COVID-19 pandemic. One country that has already vaccinated 1.8 Xtalks spoke with Dr. Derar H.
The Pharma Data
DECEMBER 14, 2020
14, 2020 — The Pfizer-BioNTech COVID-19 vaccine has received the first emergency use authorization (EUA) for a COVID-19 vaccine in the United States. The vaccine is administered in two doses three weeks apart. Researchers found the vaccine was 95 percent effective in preventing COVID-19. MONDAY, Dec. Redfield, M.D.,
The Pharma Data
SEPTEMBER 7, 2021
The first patients have been enrolled in a phase 1 randomized placebo-controlled clinical trial to study a therapeutic vaccine for opioid use disorder developed by researchers at the University of Minnesota Medical School. The vaccine currently being tested stimulates the body’s immune system to produce antibodies to oxycodone.
The Pharma Data
DECEMBER 11, 2020
Food and Drug Administration is expected to approve emergency use of Pfizer’s coronavirus vaccine as early as Saturday after its advisory panel cleared the way for the start of a national campaign to inoculate Americans and stem the spread of COVID-19. FRIDAY, Dec. 11, 2020 – The U.S. Centers for Disease Control and Prevention.
The Pharma Data
JANUARY 11, 2021
health officials raced to ramp up the delivery of COVID-19 vaccines across the country as both coronavirus case counts and death tallies continued to hit record highs. “We really need to get this vaccine out more quickly, because this is really our only tool,” Dr. Scott Gottlieb, former commissioner of the U.S. MONDAY, Jan.

NY Times
DECEMBER 3, 2020
If approved, the first batch of vaccine could arrive in the state on Dec. 15, the governor said, and contain enough doses for 170,000 people.

XTalks
SEPTEMBER 27, 2021
Although the US Food and Drug Administration (FDA) doesn’t vet pharma ads, it has a “Bad Ad” program that now has an interactive quiz to help in the reporting of misleading information in pharmaceutical promotions. Moreover, the pharmaceutical industry spent more than $24 billion on drug promotion in 2017, of which $18.5

The Pharma Data
DECEMBER 14, 2020
ICU nurse Sandra Lindsay, with Long Island Jewish Medical Center in Queens, New York, received the Pfizer-BioNTech COVID-19 vaccine. Food and Drug Administration (FDA) ’ s Vaccines and Related Biological Products Advisory Committee on Thursday, December 17. billion worth of vaccines. They have since recovered.

Rethinking Clinical Trials
SEPTEMBER 22, 2022
The COlchicine Hypertension Trial (COHERENT) is a pragmatic Phase II trial that looked at only hypertension by using registry data to identify patients that receive at least two hypertension drugs. The aim is to test communication strategies for optimizing the update of the flu vaccine. During the vaccine season they have pop-ups.

The Pharma Data
DECEMBER 2, 2020
2, 2020 – Britain became the first Western country to allow emergency use of a coronavirus vaccine on Wednesday, after approving Pfizer’s candidate in the race to inoculate millions of people around the globe. But there are daunting obstacles to the vaccine’s delivery to the masses. WEDNESDAY, Dec.

pharmaphorum
AUGUST 7, 2020
The US government has selected a potential vaccine from Sanofi and GlaxoSmithKline for its Operation Warp Speed COVID-19 vaccine development initiative, committing up to $2.1 billion for an initial 100 million doses.
NY Times
DECEMBER 12, 2020
Distributing supplies is a daunting logistical challenge, involving sophisticated tracking equipment, military contingencies and tight security.
pharmaphorum
MAY 3, 2022
. “From the beginning of the pandemic, we noted inequities in the way the US population was experiencing disease incidence – the cases of COVID and the access to testing, level of clinical services available, and, when the vaccine became available, differential levels of access to the vaccine,” Labrique says.

Druggist
AUGUST 8, 2020
It is expected to be that demand for flu vaccination service will be high this year. Additionally, eligibility criteria will be extended, for example, phased-out eligibility for free flu vaccination of those aged 50-64. This post is a summary of essential information about flu season 2020 and flu vaccination 2020/2021.

Pharmaceutical Technology
APRIL 20, 2023
Rather than bringing patients to sites, study teams are now exploring ways they can bring the trial to patients using a varying combination of direct-to-patient drug shipments, home nursing, video conferencing, and electronic data collection. Drug development had to change. The face of clinical research is evolving.

Druggist
AUGUST 8, 2020
It is expected to be that demand for flu vaccination service will be high this year. Additionally, eligibility criteria will be extended, for example, phased-out eligibility for free flu vaccination of those aged 50-64. This post is a summary of essential information about flu season 2020 and flu vaccination 2020/2021.

pharmaphorum
NOVEMBER 2, 2020
With a collective sense of uncertainty, many are pinning their hopes on a vaccine and treatment, and sophisticated technology could help speed up the process of finding an effective medicine. This knowledge is critical in understanding how effective certain drugs might be in treating the virus. One example is Novartis.

Pfizer
AUGUST 15, 2022
I am grateful to have received four doses of the Pfizer-BioNTech vaccine and I am feeling well while experiencing very mild symptoms. I am incredibly grateful for the tireless efforts of my Pfizer colleagues who worked to make vaccines and treatments available for me and people around the world. NEW YORK, AUGUST 15, 2022 – Pfizer Inc.

XTalks
OCTOBER 18, 2022
Prospective employees should know that if they join Medpace, they will be part of an organization that has an overall goal to support the development of innovative drugs and devices. The industry awards are based on feedback from sponsors of drugs, biologics, and medical devices as well as clinical research sites. Quality assurance.

Pharmaceutical Technology
JUNE 23, 2022
During the peak of the Covid-19 pandemic this challenge reached new heights, as pharmaceutical firms around the world mobilised the full might of their resources to try and identify, develop, and trial a wide range of vaccines and therapeutics to alleviate the lethal threat posed by the virus. Decentralised trials continue to be in-demand.

pharmaphorum
OCTOBER 25, 2021
The company is best known for its saliva-based health and ancestry DNA testing targeted at consumers, but also uses its genetic database to look for new targets for drug development. The transaction is expected to close before the end of the year. The post 23andMe makes $400m telehealth play, gulping down Lemonaid appeared first on.
pharmaphorum
NOVEMBER 10, 2022
This is why the Maltese Government has set up ‘nurse navigators’, an innovative cancer care service that “brings a personalised, integrated approach to care both during and after treatment”, according to the World Health Organization (WHO). The Maltese situation and nurse navigators. However, we live in a digital age, he said.
pharmaphorum
DECEMBER 23, 2022
There’s been huge progress in treatments for blood cancer in recent years – but drawbacks of expensive CAR-T cell and injected antibody therapies have led drug developers to look at novel oral therapies as patient-friendly alternatives. Side effects and cost implications.

pharmaphorum
APRIL 27, 2022
The reputation of COVID-19 vaccine makers understandably soared early on, as clinical trials were started and completed in record time, giving hopes to millions of people in lockdown. Among individuals not vaccinated by choice, internet searches ranked as the number one source. Working on a diabetes drug?

The Pharma Data
JANUARY 15, 2021
.” Called the “American Rescue Plan,” the legislative proposal would meet Biden’s goal of administering 100 million vaccines by the 100th day of his administration, while reopening most schools by the spring, the Associated Press reported. ” US vaccine rollout nears 1 million doses a day.

The Pharma Data
NOVEMBER 26, 2020
Although Covid-19 deaths and new confirmed cases continue to rise across nursing homes in the US, many of those infected do recover. This has led to growing interest in the relationship between antibodies and their protective value whether generated by previous Covid-19 exposure or by a vaccine, once available. About DNA Link, Inc.

XTalks
FEBRUARY 9, 2021
Nursing homes and long-term care facilities have borne the brunt of the COVID-19 pandemic, with residents being a particularly vulnerable population due to age-related factors and the prevalence of underlying health conditions. The emergence of the COVID-19 pandemic prompted Dr. COVID-19 Vaccine Uptake in Long-Term Care Facilities.

The Pharma Data
MAY 13, 2021
Description: Statements on this website have not been evaluated by the Food and Drug Administration. If you are pregnant, nursing, taking medication, or have a medical condition, consult your physician before using our products. Statements on this website have not been evaluated by the Food and Drug Administration.

The Pharma Data
NOVEMBER 30, 2020
In a bit of good news, the first wave of vaccinations are expected to begin for targeted groups in a matter of weeks, the Post reported. Centers for Disease Control and Prevention advisory committee on immunization practices will meet Tuesday to vote on coronavirus vaccine priority rankings. 21, The New York Times reported.

Drug Discovery World
SEPTEMBER 1, 2022
It also reduces the effectiveness of oral vaccines. . Zyambo also visited Professor Christine Edwards at the University of Glasgow’s School of Medicine, Dentistry and Nursing to learn about a new way to assess the function of gut bacteria. . She will take the spectrometer back to Zambia with her to continue the research.

The Pharma Data
NOVEMBER 4, 2020
You must receive meningococcal vaccines at least 2 weeks before your first dose of ULTOMIRIS if you are not vaccinated. If your doctor decided that urgent treatment with ULTOMIRIS is needed, you should receive meningococcal vaccination as soon as possible. Your doctor will decide if you need additional vaccination.

XTalks
JANUARY 5, 2021
This year will see the rolling out of much-awaited pandemic-ending COVID-19 vaccines. Both drug discovery and drug repurposing to manage and treat the disease will also continue as experts learn more about the virology and epidemiology of the novel SARS-CoV-2 coronavirus that wreaked havoc in 2020.

Pfizer
JANUARY 27, 2023
In the ongoing development of the Pfizer-BioNTech COVID-19 vaccine, Pfizer has not conducted gain of function or directed evolution research. This research provides a way for us to rapidly assess the ability of an existing vaccine to induce antibodies that neutralize a newly identified variant of concern.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content