FDA grants approval for Novartis’ hidradenitis suppurativa treatment
Pharmaceutical Technology
NOVEMBER 1, 2023
The US FDA has granted approval for Novartis’ Cosentyx to treat adult patients with moderate to severe hidradenitis suppurativa (HS).

Pharmaceutical Technology
NOVEMBER 1, 2023
The US FDA has granted approval for Novartis’ Cosentyx to treat adult patients with moderate to severe hidradenitis suppurativa (HS).
AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 1, 2023
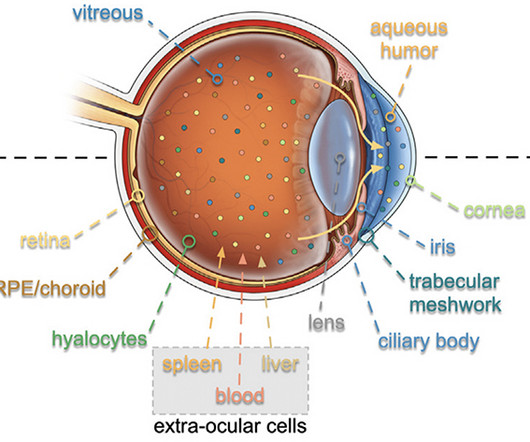
Scientists have developed a new tool for determining the age of eye cells without sampling regenerative tissue, which could make treatments for eye disease more personalized and better targeted. The team, led by researchers from Stanford University, adapted a technique used for analyzing eye fluid.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
NOVEMBER 1, 2023
Pfizer has posted a net loss of $2.38bn in Q3 2023 compared to a net income of $8.61bn during the same period of 2022.

AuroBlog - Aurous Healthcare Clinical Trials blog
NOVEMBER 1, 2023
The Drug Controller General of India (DCGI) has added in-vitro diagnostic (IVD) medical devices including those for diagnosis of Covid-19, ribonucleic acid (RNA) and deoxyribonucleic acid (DNA) extraction kits, among others into the Class C risk category under the Medical Devices Rules (MDR), 2017.

Rethinking Clinical Trials
NOVEMBER 1, 2023
In this Friday’s PCT Grand Rounds, Jonathan Moyer of the NIH Office of Disease Prevention will continue our special series, Advances in the Design and Analysis of Pragmatic Clinical Trials, with his presentation, “The Perils and Pitfalls of Complex Clustering in Pragmatic Trials.” The session will be held on Friday, November 3, at 1:00 pm eastern and will be moderated by Andrea Cook.

Bio Pharma Dive
NOVEMBER 1, 2023
Sales of GSK’s new RSV shot Arexvy were more than double what Pfizer reported for its rival product, suggesting substantial demand this fall.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Bio Pharma Dive
NOVEMBER 1, 2023
The San Francisco-based venture capital firm has already invested in precision medicine as well as cell and gene therapy.
Pharmaceutical Technology
NOVEMBER 1, 2023
ONL Therapeutics plans to raise $75-$100m to support Phase II trials in geographic atrophy and open-angle glaucoma.
XTalks
NOVEMBER 1, 2023
This episode features an interview with Dr. Stephanie Manson Brown, VP & Head of Clinical Development & Scientific Innovation of R&D at Allergan Aesthetics at AbbVie. Allergan Aesthetics is focused on creating products and technologies that drive the advancement of aesthetic medicine. Dr. Manson Brown’s Clinical Development and Scientific Innovation team are responsible for global clinical trial development strategy for the Aesthetic Medicine portfolio, covering pharmaceuticals and d
Pharmaceutical Technology
NOVEMBER 1, 2023
Kite has signed agreement with Epic Bio for the development of new cancer cell therapies leveraging the latter’s gene regulation platform.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

BioSpace
NOVEMBER 1, 2023
The company’s blockbuster cancer drug continues to snap up FDA approvals, this time in treating biliary tract cancer in combination with cisplatin and gemcitabine. It’s the sixth indication in gastrointestinal cancers.

Pharmaceutical Technology
NOVEMBER 1, 2023
Johnathan Milner suspended his vote against campaign today in the wake of "extensive feedback" from Abcam shareholders.

Fierce Pharma
NOVEMBER 1, 2023
Cosentyx may soon share the hidradenitis suppurativa (HS) field with another inflammatory biologic—but, for now, the Novartis drug has a head start. | The FDA approved Cosentyx for the treatment of moderate to severe hidradenitis suppurativa in adults, Novartis said Tuesday. The green light makes Cosentyx the first new biologic to treat the lesser-known skin affliction in nearly a decade.

Pharmaceutical Technology
NOVEMBER 1, 2023
GSK and Arrowhead Pharmaceuticals have signed an agreement for Johnson & Johnson company Janssen Pharmaceuticals’ JNJ-3989.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Pharma Times
NOVEMBER 1, 2023
The AI tools to be deployed will help NHS staff to analyse X-rays and CT scans - News - PharmaTimes

Pharmaceutical Technology
NOVEMBER 1, 2023
Curavit will incorporate HEOR services into trials to elucidate the real-world financial impact of a new product.

Pharma Times
NOVEMBER 1, 2023
All CDCs will be open by March 2024 to speed up lifesaving checks and tests - News - PharmaTimes
Pharmaceutical Technology
NOVEMBER 1, 2023
The Amgen biosimilar Wezlana has been approved for all indications and is forecasted to yield $455m in 2029.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.
Outsourcing Pharma
NOVEMBER 1, 2023
OSP had a quickfire interview with Rebecca Starkie, senior global patient engagement director, at Advanced Clinical where she explains the challenges faced by rare disease patients - and suggests ways to improve.

Pharmaceutical Technology
NOVEMBER 1, 2023
As per WHO, about 1 in 10 medical products in low- and middle-income countries is substandard or falsified.

Outsourcing Pharma
NOVEMBER 1, 2023
A transaction between Citius Pharmaceuticals and TenX Keane which will result in the formation of Citius Oncology, Inc. is due to close in the first half of next year (2024).

Fierce Pharma
NOVEMBER 1, 2023
The FDA has signed off on Amgen’s biosimilar version of Johnson & Johnson’s autoimmune standout Stelara. | The FDA has approved Amgen’s biosimilar version of Johnson & Johnson’s autoimmune standout Stelara. But the new product will not launch until 2025 because of a settlement the companies made earlier this year, which delayed the launch in exchange for J&J dropping a patent infringement lawsuit.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Outsourcing Pharma
NOVEMBER 1, 2023
Element Biosciences, Inc., will share news of its sequencing methodology and host a workshop on higher accuracy sequencing for improved variant detection in Washington DC from today.

BioSpace
NOVEMBER 1, 2023
Riding a wave of skyrocketing Wegovy and Ozempic sales, the company on Thursday raised its sales and operating profit outlook for the rest of the year as part of its third-quarter earnings announcement.

Fierce Pharma
NOVEMBER 1, 2023
After AstraZeneca’s Imfinzi last year claimed new territory the biliary tract cancer space, Merck’s Keytruda has hit the scene with an FDA nod to match its rival. | The approval was based off of Merck's phase 3 KEYNOTE-966 trial and marks Keytruda's sixth in gastrointestinal cancers. Meanwhile, AZ's Imfinzi scored its first approval in the space last year.

BioSpace
NOVEMBER 1, 2023
The Swiss pharma has set up its challenge to AbbVie’s blockbuster immunosuppressive drug Humira with a label expansion for Cosentyx in hidradenitis suppurativa, a painful long-term skin condition.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Fierce Pharma
NOVEMBER 1, 2023
After 18 years at the helm, BioMarin’s CEO Jean-Jacques Bienaimé is hanging up the gloves. | BioMarin lured Genentech's CEO Alexander Hardy to take over upon Jean-Jacques Bienaimé's upcoming retirement after 18 years at the helm.

BioSpace
NOVEMBER 1, 2023
The biopharma industry is moving toward using AI to try to determine how well a given person would perform in a role, with applications that go beyond recruiting.

Fierce Pharma
NOVEMBER 1, 2023
Besides Pfizer’s fast-declining COVID-19 sales, vaccine rival GSK is giving the New York drugmaker another reason to worry. | Aside from Pfizer's declining COVID-19 sales, GSK is giving the New York drugmaker another reason to worry. In the first round of their RSV vaccine showdown, GSK's Arexvy pulled down $860 million, more than doubling the haul from Pfizer's Abrysvo.

BioSpace
NOVEMBER 1, 2023
Sales of type 2 diabetes treatment Mounjaro brought in $1.4 billion in the third quarter. However, the company cut its full-year profit guidance due to charges mainly related to recent acquisitions.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content