5 trends in biotech dealmaking to watch in 2021
Bio Pharma Dive
JANUARY 13, 2021
Brain drugs, private equity players and blank-check companies are just some of the things deal experts say they'll be keeping an eye on this year.

Bio Pharma Dive
JANUARY 13, 2021
Brain drugs, private equity players and blank-check companies are just some of the things deal experts say they'll be keeping an eye on this year.

BioSpace
JANUARY 13, 2021
The COVID-19 pandemic that has upended life across the globe has also ignited a reawakening to the value of vaccines, and Hu believes the momentum will carry forward into the new year.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
JANUARY 13, 2021
The deal expands an earlier agreement between Regeneron and the U.S., though many of the issues limiting the drug's use haven't been solved.
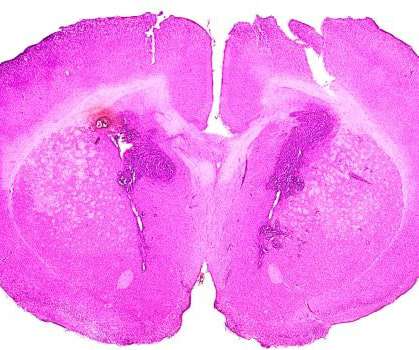
BioSpace
JANUARY 13, 2021
Adastra Pharmaceuticals’ positive results from a Phase Ib trial of its therapy for recurrent high-grade glioblastomas suggest an effective treatment could be available within the next several years.

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Scienmag
JANUARY 13, 2021
International Ukraine Genetic Diversity Project finds a quarter of the genetic variation in Europe, dramatically increasing information on population diversity and medical genetic variation Credit: Oakland University Today, the largest study of genetic diversity in Ukraine was published in the open science journal GigaScience. The project was an international effort, bringing together researchers in Ukraine, […].
BioSpace
JANUARY 13, 2021
In many regards, the incoming Biden Administration is expected to be a redux of the Obama Administration, likely with strong support for scientific research and a reinvigoration of the Affordable Care Act. Yet concerns of pricing controls still loom.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

BioSpace
JANUARY 13, 2021
In an interview with BioSpace, Marianne De Backer, Head of Business Development & Licensing in Bayer’s Pharmaceuticals Division, outlined Bayer’s thought process for opening its purse strings and diving into the deep end of the cell and gene therapy space.

pharmaphorum
JANUARY 13, 2021
Everything, from exercise classes to pub quizzes, went online last year – and medical education was no exception. In their droves, congresses, conferences, and masterclasses went virtual in a bid to ensure healthcare professionals were supported, up to date, and socially distanced. But is this change here to stay? Are the days of queuing for coffee, rushing to symposiums, and the glitzy conference dinner a thing of the past?

BioSpace
JANUARY 13, 2021
Flagship Pioneering launched a new company called Inzen Therapeutics, which will discover and develop a new class of medicines based on a new area of biology that will provide new therapeutic approaches to multiple types of diseases.
pharmaphorum
JANUARY 13, 2021
IO Biotech, an oncology specialist formed and backed by Denmark’s Novo Holdings, has raised €127 million ($155 million) to further develop its cancer vaccine technology that has boosted efficacy of PD-1 immunotherapy in early trials. The Series B fundraiser follows the FDA’s decision to grant Breakthrough Therapy designation last month for a combination of its lead immune-oncology therapies IO102 and IO103, with anti-PD-1 monoclonal antibodies for patients with metastatic melanoma.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Outsourcing Pharma
JANUARY 13, 2021
A leader from the anti-counterfeit solutions firm says that while packaging-level efforts may have an effect, on-dose technology is worth a closer look.

BioSpace
JANUARY 13, 2021
Sana Biotechnology has filed with the U.S. Securities and Exchange Commission to raise up to $150 million in an initial public offering to help support the company’s cell-based therapy discovery and development operations.

Outsourcing Pharma
JANUARY 13, 2021
Fight the Fakes gathers manufacturers and other stakeholders to brainstorm on ways to combat falsified medicines and safeguard the worldâs medicine supply.

BioSpace
JANUARY 13, 2021
Cancer companies Delfi Diagnostics and Elucida Oncology announced Series A raises worth $144 million so far.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Outsourcing Pharma
JANUARY 13, 2021
The pharmaceutical firm reports a counterfeit version of its Symtuza HIV treatment has been found and pulled from shelves at three American pharmacies.

BioSpace
JANUARY 13, 2021
Major players in the biotech industry are moving full steam ahead in their 2021 operations goals despite the ongoing pandemic, with global pharmaceutical giant Takeda being no exception.
Pharma Times
JANUARY 13, 2021
First patient in global trial dosed in the UK
pharmaphorum
JANUARY 13, 2021
Hackers posted stolen confidential information about Pfizer/BioNTech’s COVID-19 vaccine online, the European Medicines Agency has said in an update. The EMA last month revealed that documents relating to the Pfizer/BioNTech vaccine were stolen in a cyber-attack on its records. Since then the agency has been investigating the incident involving the theft of documents that formed part of BioNTech’s filing for the BNT-162b vaccine.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

Scienmag
JANUARY 13, 2021
Credit: KIRSTY CHALLEN, B.SC., MBCHB, MRES, PH.D., LANCASHIRE TEACHING HOSPITALS, UNITED KINGDOM. DES PLAINES, IL — Among children who were not in an independently verified incident, evaluation for child abuse should be done by specialty consultation in children aged less than three-years old presenting with rib fractures and children aged less than 18-months presenting with […].

Outsourcing Pharma
JANUARY 13, 2021
Authorities discovered the fake Pfizer erectile-dysfunction tablets in one of three cargo containers full of fraudulent goods, recently arrived from China.

Scienmag
JANUARY 13, 2021
Dark Energy Survey releases catalog of 700 million objects Elliptical galaxies are generally characterized by their relatively smooth appearance when compared with spiral galaxies (one of which is to the left) which have more flocculent structure interwoven with dust… view more The Dark Energy Survey has released a massive, public collection of astronomical data and […].

BioSpace
JANUARY 13, 2021
Biopharma and life sciences companies from across the globe provide updates on their pipelines and businesses.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.

Scienmag
JANUARY 13, 2021
Researchers at the University of Tsukuba in Japan have discovered a group of neurons in the mouse brainstem that suppress unwanted movement during rapid eye movement sleep Credit: University of Tsukuba Tsukuba, Japan — We laugh when we see Homer Simpson falling asleep while driving, while in church, and while even operating the nuclear reactor. […].

BioSpace
JANUARY 13, 2021
Here’s a look at some of the stories that emerged from the conference on Wednesday.
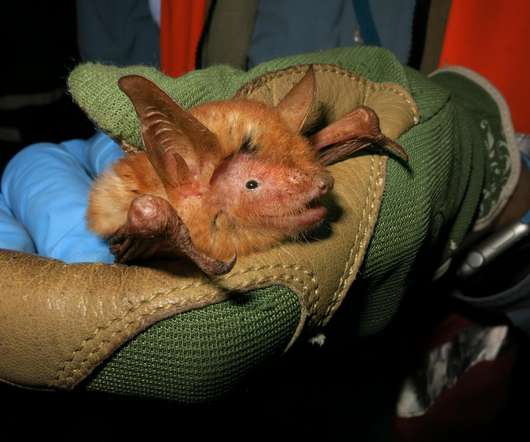
Scienmag
JANUARY 13, 2021
Credit: © Bat Conservation International A group of scientists led by the American Museum of Natural History and Bat Conservation International have discovered a new species of a striking orange and black bat in a mountain range in West Africa. The species, which the researchers expect is likely critically endangered, underscores the importance of sub-Saharan […].

Pharma Times
JANUARY 13, 2021
Investigational antibody targets a modified form of beta amyloid called N3pG

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Scienmag
JANUARY 13, 2021
Nearly 200 researchers pitched in to gather, process, and stitch together images for half of the sky to prepare for the start of the Dark Energy Spectroscopic Instrument’s observations Credit: DESI Legacy Imaging Surveys Before DESI, the Dark Energy Spectroscopic Instrument, can begin its 5-year mission from an Arizona mountaintop to produce the largest 3D […].

Clinical Trial Gurus
JANUARY 13, 2021
Large CROs tend to take larger contracts which makes it more difficult for anyone to open a new CRO and get ahead. One solution to this problem is to focus on opening a site and then work on establishing the new site as a leader in a specific area while networking with other sites. Establishing a strong network of sites can be more profitable in the long run because it allows you to interact with more sponsors and potentially offer services to them.

Scienmag
JANUARY 13, 2021
Credit: (U.S. Navy photo by John F. Williams) ARLINGTON, Va.–The Department of Defense (DoD) recently awarded $50 million in grants to 150 university scientists–54 of whom are sponsored by the Office of Naval Research (ONR)–via the Defense University Research Instrumentation Program (DURIP). Through DURIP, the DoD supports the purchase of state-of-the-art equipment and instrumentation to […].

pharmaphorum
JANUARY 13, 2021
Trial sites have adapted swiftly to the restrictions of COVID-19, and patients have seen many knock-on benefits as a result. The next step is ensuring the industry does not regress to old ways of working once the pandemic is over, say Karen McIntyre and Allyson Small. COVID-19 has changed everything for clinical trials – but in most cases these are changes that were well overdue.

Advertisement
The global landscape of clinical trials is rapidly changing as studies become more complex. An increasing number of sponsors are seeking enhanced flexibility in their supply chains to address a variety of clinical supply challenges, including patient demand and reducing delays. Demand-led supply and direct-to-patient distribution are next-generation solutions that are helping to meet these growing needs, allowing for more streamlined processes and patient-centric studies.
Let's personalize your content