AstraZeneca’s Voydeya Scores FDA Approval, Adding to Company’s Multi-Billion Dollar PNH Franchise
XTalks
APRIL 3, 2024
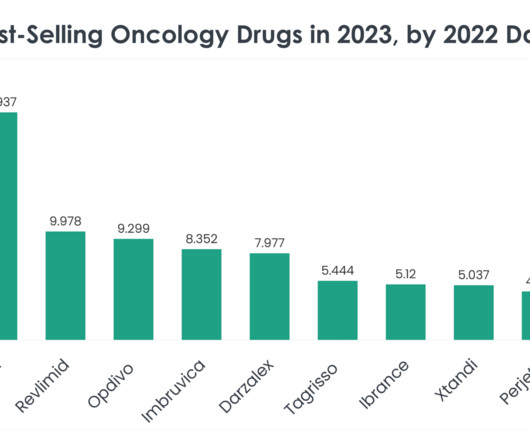
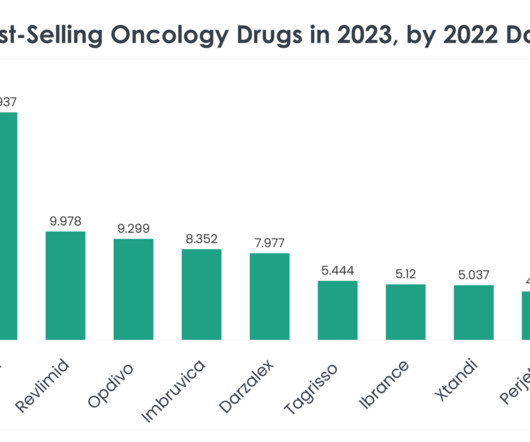
While Soliris had its market launch 17 years ago with $3.2 billion in sales in 2023, Ultomiris was approved in 2018 and generated $3 billion in revenue last year. Competitors in the PNH market include Novartis, which won approval for its factor B inhibitor Fabhalta (iptacopan), the first oral pill for PNH treatment, late last year.









































Let's personalize your content