Gene therapy biotech Jaguar spins out manufacturing company
Bio Pharma Dive
JANUARY 18, 2024
Called Advanced Medicine Partners, the new company will offer specialized cell and gene therapy manufacturing services to biotech and pharma clients.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Bio Pharma Dive
JANUARY 18, 2024
Called Advanced Medicine Partners, the new company will offer specialized cell and gene therapy manufacturing services to biotech and pharma clients.

Pharmaceutical Technology
APRIL 19, 2024
Paolo Morgese, vice president of European public affairs at the Alliance for Regenerative Medicine, discusses the barriers affecting access to gene therapies.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
FEBRUARY 8, 2024
Despite several ground-breaking developments, pharmaceutical and biotech companies are often reluctant to invest in cell and gene therapy.

Pharmaceutical Technology
MAY 4, 2023
Forge Biologics and global life sciences company Labcorp have announced a strategic adeno-associated virus (AAV) gene therapy development and manufacturing collaboration. This will improve accessibility to services for AAV-mediated gene therapy programmes.
Pharmaceutical Technology
JULY 7, 2022
But scientists have struggled to find effective treatments for many of these diseases since the dawn of modern medicine. Recent years have seen a steadily growing number of approvals for cell and gene therapies, which has spurred on the community to continue innovating in this space. A new frontier in cancer research.
Bio Pharma Dive
JULY 28, 2023
The British pharma is deepening its investment in genetic medicine through a deal for a group of Pfizer’s gene therapy delivery tools, while the New York giant cuts back.
Pharmaceutical Technology
OCTOBER 19, 2022
Eli Lilly and Company has signed a definitive agreement for the acquisition of all outstanding shares of precision genetic medicine firm Akouos for a total deal value of up to nearly $610m or up to $15.50 Akouos focuses on developing adeno-associated viral gene therapies to treat inner ear conditions, including sensorineural hearing loss.

pharmaphorum
MARCH 20, 2024
Orchard Therapeutics has revealed the US price of Lenmeldy, its gene therapy for rare disease MLD, placing a $4.25m price tag on the one-shot treatment

Bio Pharma Dive
APRIL 1, 2022
Taysha Gene Therapies is narrowing its research efforts and cutting around 35% of its workforce, the latest in a series of layoffs announced by makers of genetic medicines.
BioPharma Reporter
APRIL 30, 2024
million gene therapy for hemophilia B just got FDA approval. Hereâs what it means for patients and the future of medicine Pfizer's groundbreaking $3.5

Pharmaceutical Technology
MAY 24, 2023
Forge Biologics has joined the public-private collaboration, the Bespoke Gene Therapy Consortium (BGTC), to expedite the development and manufacture of new AAV [adeno-associated virus] gene therapies to treat patients with rare diseases. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.

Pharmaceutical Technology
MAY 16, 2023
The Foundation for the National Institutes of Health (FNIH) has announced its plans to prioritise eight rare diseases to provide industry standards for manufacturing, preclinical testing and product analytical testing for gene therapy development. This will include pairing up indications with manufacturers amongst the BGTC’s partners.

Pharmaceutical Technology
MARCH 10, 2023
On 10 March, the National Health Service Blood and Transplant (NHSBT) opened a new Clinical Biotechnology Centre (CBC) with the aim of improving the UK’s ability to develop and manufacture cell and gene therapies. Personalised medicines will also be developed at the centre.
Pharmaceutical Technology
AUGUST 25, 2022
The European Commission (EC) has granted conditional marketing authorisation (CMA) for BioMarin Pharmaceutical ’s gene therapy, Roctavian (valoctocogene roxaparvovec), to treat adults with severe haemophilia A (congenital Factor VIII deficiency). Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.

Pharmaceutical Technology
MAY 25, 2023
ElevateBio has raised $401m in a Series D financing round for advancing its technology platforms to expedite the design, production and development of cell and gene therapies. ElevateBio intends to use the funds to advance its genetic medicine current good manufacturing practice (cGMP) and process development business, BaseCamp.
Pharmaceutical Technology
JANUARY 5, 2023
Capsida Biotherapeutics and Eli Lilly and Company ’s wholly owned subsidiary Prevail Therapeutics have announced a partnership for the development of non-invasive gene therapies for central nervous system (CNS) diseases. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.

Pharmaceutical Technology
NOVEMBER 9, 2022
On November 2, the Institute for Clinical and Economic Review (ICER) released its updated evidence aimed at measuring the clinical effectiveness and cost of the two haemophilia gene therapies. Known by the brand name Roctavian, BioMarin’s haemophilia A therapy valoctocogene roxaparvovec could be fairly priced in the range of $1.95–1.96

Pharmaceutical Technology
FEBRUARY 15, 2023
In the last three years alone, there have been over 633,000 patents filed and granted in the pharmaceutical industry, according to GlobalData’s report on Immuno-oncology in Pharmaceuticals: Gene therapy delivery using viral vectors. Sanofi is one of the leading patent filers working with viral vectors.

pharmaphorum
FEBRUARY 27, 2024
Looking to the future of clinical trials: Gene therapy, precision medicine, and the ongoing quest for rare disease solutions Mike.Hammerton Tue, 27/02/2024 - 12:30 Bookmark this

Fierce Pharma
APRIL 29, 2024
After last quarter presenting three factors necessary for a more successful launch of hemophilia A gene therapy Roctavian, BioMarin has now added a divestiture to the list of potenti | After last quarter presenting three factors necessary for a more successful launch of hemophilia A gene therapy Roctavian, BioMarin has now added a divestiture to the (..)
Pharmaceutical Technology
MAY 12, 2023
Editas Medicine will release new efficacy and safety results of its gene therapy EDIT-301 in severe sickle disease as part of an oral presentation at the European Hematology Association’s (EHA) Hybrid Congress. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.
Pharmaceutical Technology
MAY 24, 2023
It can correct several disease elements associated with PKD, a rare, monogenic red blood cell disorder caused by a mutation in the PKLR gene. The therapy has also received orphan drug and fast-track designations. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.

BioPharma Reporter
MARCH 7, 2024
In the realm of modern medicine, few advancements have inspired and challenged the industry quite like gene therapy.
Pharmaceutical Technology
APRIL 25, 2023
Forge Biologics has received a qualified person (QP) declaration to manufacture adeno-associated virus (AAV) gene therapies to support European clinical programmes. The FBX-101 is an AAV serotype rh10 gene therapy, given intravenously following haematopoietic stem cell transplantation infusion.

Bio Pharma Dive
JANUARY 24, 2024
An 11-year-old boy born deaf can hear after receiving Lilly's genetic medicine. Others, including biotechs in China and Regeneron in the U.S., are taking aim at the same drug target.

Fierce Pharma
MARCH 14, 2024
After charting multiple personalized medicine expansions in recent years, contract manufacturer Charles River Laboratories is back with another gene therapy pact—this time centered on an adeno-asso | After charting multiple personalized medicine expansions in recent years, contract manufacturer Charles River Laboratories is back with another gene therapy (..)

Bio Pharma Dive
MARCH 20, 2024
million list price, the highest of any genetic medicine to come to market. Orchard is counting on the long-term data it’s accrued to convince insurers to cover Lenmeldy’s $4.25
Bio Pharma Dive
DECEMBER 8, 2023
Casgevy, the first CRISPR therapy approved by the FDA, will cost $2.2 million, while a competing genetic medicine also cleared Friday is priced at $3.1

Bio Pharma Dive
JUNE 22, 2023
The cost makes Sarepta’s treatment, called Elevidys, among the most expensive medicines in the world. But company executives said they don’t expect significant insurer pushback.

Bio Pharma Dive
JUNE 29, 2023
After a prolonged journey, the medicine, known as Roctavian, is now cleared for certain patients with hemophilia A, the more common form of the rare bleeding disorder.

Pharmaceutical Technology
JUNE 19, 2023
Although only a small number of gene therapies have reached the market thus far, the industry is poised to grow quickly over the next few years. According to GlobalData’s clinical trials database, there are currently 1,231 planned and ongoing trials for gene therapies and gene-modified cell therapies alone.

Bio Pharma Dive
APRIL 24, 2023
Submission of the application comes weeks after rivals Vertex and CRISPR filed their gene editing medicine for the disease with the agency.
Fierce Pharma
DECEMBER 8, 2023
| Vertex Pharmaceuticals and CRISPR Therapeutics have won an historic FDA approval for their sickle cell disease gene therapy Casgevy. The transformational treatment is a potential cure for the debilitating and life-threatening disease which affects more than 100,000 in the United States, most of them Black.

Pharmaceutical Technology
FEBRUARY 27, 2024
Making therapies feasible from biological, regulatory, and commercial standpoints drives scalable genetic medicine.

Fierce Pharma
NOVEMBER 2, 2023
The third quarter marked a momentous one for Sarepta Therapeutics, bringing the long-anticipated launch of its Duchenne muscular dystrophy (DMD) gene therapy Elevidys. Even before a potential label expansion, the gene therapy Elevidys has raked in around $69 million since its launch.

Drug Discovery World
NOVEMBER 16, 2023
The Medicines and Healthcare products Regulatory Agency (MHRA) has authorised the world’s first gene therapy for sickle-cell disease (SCD) and transfusion-dependent β-thalassemia (TDT). Casgevy is designed to work by editing the faulty gene in a patient’s bone marrow stem cells so that the body produces functioning haemoglobin.

Bio Pharma Dive
APRIL 1, 2024
The FDA could soon expand use of two multiple myeloma cell therapies as well as a top-selling medicine from Sanofi and Regeneron, while Pfizer’s years of gene therapy work might finally pay dividends.
pharmaphorum
DECEMBER 16, 2022
CSL’s gene therapy for haemophilia B has been recommended for approval by the EMA’s human medicine committee, setting up a decision by the European Commission early next year. CSL launched the therapy in the US with a list price of $3.5
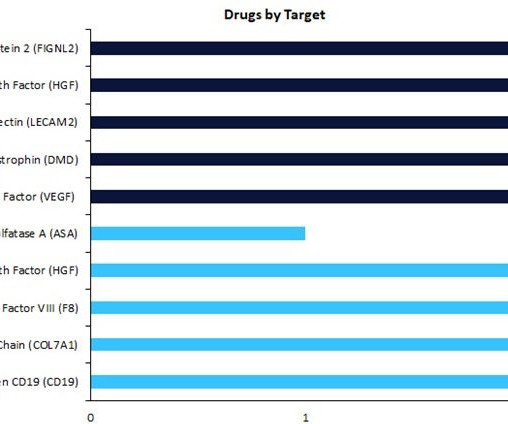
Pharmaceutical Technology
MARCH 27, 2023
Regenerative medicines in early-stage development (preclinical, discovery, or investigational new drug [IND]/ clinical trial application [CTA] filed status) have seen a change in drug targets compared to therapies in late-stage development (Phase II to pre-registration stage).

Camargo
DECEMBER 20, 2021
Many of the advancements included in PDUFA VII are driven in large part by an increase in the development of innovative products such as cell and gene therapies. The increase is designed to strengthen CBER’s capacity and capabilities for regulating cell and gene therapies. 2024: 48 staffers. 2025: 29 staffers.

Drug Discovery World
NOVEMBER 27, 2023
This 24-page eBook sponsored by Sartorius explores the latest innovations in cell and gene therapies, where the challenges lie and the possible solutions to overcoming these obstacles.

Drug Discovery World
JUNE 23, 2023
Sarepta Therapeutics’ Elevidys has become the first gene therapy for Duchenne muscular dystrophy (DMD) to gain marketing authorisation in the US. It is contraindicated in patients with any deletion in exon 8 and/or exon 9 in the DMD gene.
Pharmaceutical Technology
OCTOBER 14, 2022
Pharmaceutical companies Xcell Biosciences (Xcellbio) and aCGT Vector have partnered to expedite the development of cell and gene therapies. The two companies will aim to improve the manufacturing and analytic procedures used to develop personalised cell and gene therapies to treat cancer patients.

Bio Pharma Dive
AUGUST 17, 2022
The regulator cleared the biotech’s medicine Zynteglo for transfusion-dependent beta thalassemia, giving patients a powerful new treatment option. But it will come at a very high cost of $2.8 million in the U.S.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content