US FDA approves Pharming’s Joenja to treat APDS
Pharmaceutical Technology
MARCH 27, 2023
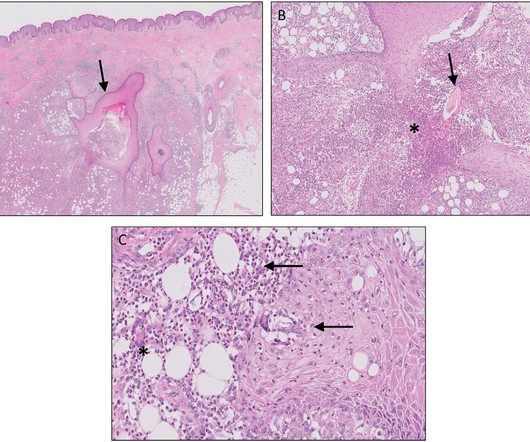
It is claimed to be the first and only therapy to receive approval in the US for the treatment of APDS, a rare and progressive primary immunodeficiency. After assessing the New Drug Application (NDA) under priority review, the FDA granted the approval based on the data obtained from a Phase II/III clinical trial.



































Let's personalize your content