Why small particles matter: Manufacturing and micro-material contamination
Pharmaceutical Technology
MARCH 6, 2024
To protect patient safety and reduce recall risks, drug manufacturers must reduce particle contamination sources wherever possible.

Pharmaceutical Technology
MARCH 6, 2024
To protect patient safety and reduce recall risks, drug manufacturers must reduce particle contamination sources wherever possible.

Fierce Pharma
NOVEMBER 20, 2023
The threat of microbial contamination has triggered yet another drug recall, this time from German pharma bigwig Bayer. after routine stability testing discovered the presence of microbial contamination identified as Penicillium brevicompactum. Bayer is recalling one lot of Vitrakvi oral solution in the U.S.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Fierce Pharma
SEPTEMBER 25, 2023
Drugs tainted with impurities ranging from bacteria and particulates to likely cancer agents have triggered multiple FDA recalls in recent years. Drugs tainted with impurities ranging from bacteria and particulates to likely cancer agents have triggered multiple FDA recalls in recent years.

Drug Discovery World
MAY 17, 2023
A first-in-human clinical trial of an experimental oral drug for removing radioactive contaminants from inside the body has begun. The trial is testing the safety, tolerability and processing in the body of escalating doses of the investigational drug product HOPO 14-1 in healthy adults.

XTalks
JULY 16, 2021
Johnson & Johnson is recalling sunscreens from two of its popular brands over concerns of carcinogenic benzene contamination. In fact, benzene is one of the 20 most widely used chemicals in the US, serving as a starting material for making plastics, dyes, rubbers, drugs and pesticides. Growing Contamination Issues.

Scienmag
DECEMBER 8, 2021
Food and Drug Administration, tested multiple indicators of wastewater contamination to identify potential sources of contamination […]. A recent study by researchers at the Dauphin Island Sea Lab and the University of South Alabama, in collaboration with the U.S.
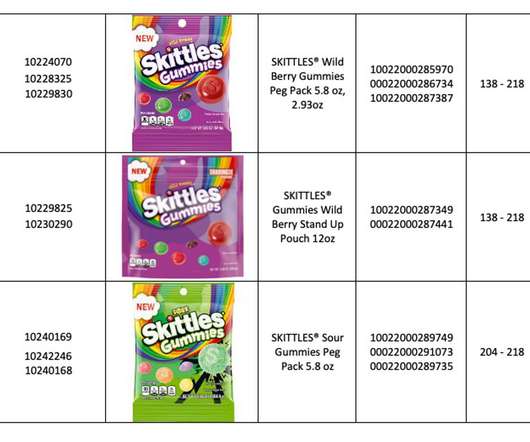
XTalks
MAY 19, 2022
The US Food and Drug Administration (FDA) posted the company’s recall announcement , which includes a complete list of products affected by the recall. . So why is metal contamination such a common reason for launching voluntary recalls? The Mars recall impacted 13 product stock keeping units (SKUs) ranging from 3.5-ounce
Let's personalize your content