Gene editing startup Metagenomi files for an IPO
Bio Pharma Dive
JANUARY 6, 2024
The Emeryville, California- based gene therapy developer has several preclinical programs in development with Moderna, Ionis and Affini-T.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Bio Pharma Dive
JANUARY 6, 2024
The Emeryville, California- based gene therapy developer has several preclinical programs in development with Moderna, Ionis and Affini-T.
Worldwide Clinical Trials
NOVEMBER 27, 2023
Casgevy, the commercial product formerly known as exa-cel, is administered by taking stem cells out of a patient’s bone marrow and editing a gene in the cells in a laboratory, with the modified cells then infused back into the patient after conditioning treatment to prepare the bone marrow.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharmaceutical Technology
MAY 25, 2023
ElevateBio has raised $401m in a Series D financing round for advancing its technology platforms to expedite the design, production and development of cell and gene therapies. New investors included research and development partner Novo Nordisk, Lee Family Office (Asia) and Woodline.

Camargo
JULY 27, 2021
How and When to Incorporate PK Design into Your Gene Therapy Development Plan. Gene therapy, which was in its infancy around 30 years ago, is now becoming a more prominent treatment method in many therapeutic areas, from personalized therapy to mass vaccinations against COVID-19. Gene Therapy Definition.

Pharmaceutical Technology
FEBRUARY 23, 2023
Moderna has entered a strategic research and development partnership with ElevateBio’s Life Edit Therapeutics to discover and develop new in-vivo mRNA gene editing therapies. Moderna will also assume further development, manufacturing, and commercialisation responsibilities on exercising a target option.
Drug Discovery World
MAY 2, 2024
Hailed as a revolution in the treatment of many diseases, cell and gene therapy (CGT) is the fastest growing area of therapeutics. The post Cell and gene therapy: Global Innovation and Opportunity appeared first on Drug Discovery World (DDW). 2024 is potentially shaping up to break this record.
Bio Pharma Dive
FEBRUARY 23, 2021
Guide Therapeutics, the company Beam acquired, is working on ways to more efficiently deliver gene editing therapies into the body.

pharmaphorum
JANUARY 29, 2021
Cutting edge’ is, for once, a truly apt description when it comes to gene editing – both because the field is pushing medicine into areas we might never have dreamed possible, and because these technologies involve literally cutting DNA at a specific point in the genome. The exact mechanism depends on the disease in question.

STAT News
FEBRUARY 22, 2023
Moderna is aiming to build a gene editing franchise powered by some of the same technologies used in its COVID-19 vaccines. The Cambridge biotech company announced Wednesday that it will partner with Life Edit Therapeutics to develop potentially permanent treatments for rare genetic diseases and other conditions.

Drug Discovery World
DECEMBER 6, 2023
Gene editing company Eligo Bioscience has announced a successful $30 million Series B funding round, led by Sanofi Ventures. “We The funding is earmarked for accelerating the development of Eligo’s flagship programme, EB005, which targets moderate to severe acne vulgaris.

XTalks
DECEMBER 13, 2023
The US Food and Drug Administration (FDA) has approved the first gene therapies for the treatment of sickle cell disease, approving two on the same day. The landmark approvals were awarded to bluebird bio’s Lyfgenia (lovo-cel) and Vertex Pharmaceuticals and CRISPR Therapeutics’ jointly developed Casgevy (exa-cel).
Pharmaceutical Technology
NOVEMBER 16, 2022
Under the deal, the LentiPeak lentiviral vector technology platform and cell therapy production expertise of ElevateBio BaseCamp will be used by Affini-T to develop its investigational oncogenic driver programmes in the clinic. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.

Worldwide Clinical Trials
OCTOBER 24, 2023
While this is a small number of people in each rare disease patient community, the significant and continued growth of rare disease drug development is unsurprising given that there are over 10,000 rare diseases , with more being discovered every year. Roughly 80% of rare diseases are due to a known genetic driver.

pharmaphorum
FEBRUARY 23, 2022
As investment in gene therapies continues to grow rapidly, more effectively engaging patients throughout development has become a priority for any company serious about building safer and more meaningful gene therapy programs. The post 2nd Annual Gene Therapy Patient Engagement Summit appeared first on.
pharmaphorum
JUNE 12, 2022
A gene-editing drug developed by CRISPR Therapeutics and Vertex Pharma has continued to show impressive results in clinical trial, with an update at the EHA congress showing long-term effects on the symptoms of both beta thalassaemia and sickle cell disease. billion product if it gets approved for both indications. .”

pharmaphorum
SEPTEMBER 28, 2022
Vertex Pharma and partner CRISPR Therapeutics will start a rolling marketing application in the US for their gene-editing drug for sickle cell disease (SCD) and beta thalassaemia later this year. The time places exa-cel in pole position to become the first drug developed based on CRISPR/Cas9 gene-editing technology to reach the market.

pharmaphorum
OCTOBER 13, 2022
Gene therapies and research into them have grown immensely in recent years, offering more novel tools in regenerative medicine to fight disease, including rare diseases and genetic disorders. billion deal with Precision Biosciences to support its development of one-time treatments for beta-thalassemia and sickle cell.

pharmaphorum
OCTOBER 4, 2022
AstraZeneca’s rare disease firm Alexion is set to expand its genomic medicine portfolio with the acquisition of gene editing specialist LogicBio Therapeutics, in a deal worth approximately $68 million. The post AstraZeneca pays record 660% premium for gene editing company LogicBio appeared first on.

Drug Discovery World
SEPTEMBER 6, 2022
In recent years the advanced therapy industry has been the most significant disruptor in medicine. Cell and gene therapies are not only transforming the way diseases are treated but are also aiming to cure entirely new patient populations. ViaCyte, Beam, and Verve are all due to begin gene editing trials this year.

Drug Discovery World
DECEMBER 11, 2023
The US Food and Drug Administration (FDA) has approved the first cell-based gene therapies for the treatment of sickle cell disease (SCD), Casgevy and Lyfgenia. Casgevy, from Vertex Pharmaceuticals and CRISPR Therapeutics, is also the first FDA-approved treatment to use CRISPR gene editing technology.

XTalks
NOVEMBER 3, 2023
After spending almost an entire day deliberating the safety of Vertex Pharmaceuticals’ and CRISPR Therapeutics’ CRISPR-based gene therapy exa-cel for sickle cell disease, a US Food and Drug Administration (FDA) advisory panel appears to be satisfied with what it saw. CRISPR works as genetic scissors to edit parts of the genome.
pharmaphorum
OCTOBER 21, 2020
million) to develop its gene therapy for a rare ocular disease. In a statement, the company said funds will be used to further develop SparingVision’s SPVNo6, for treating all forms of the rare eye disease retinitis pigmentosa. The UK biotech said in June that it would start an internal review of the two gene therapies.

pharmaphorum
SEPTEMBER 14, 2022
SparingVision has raised €75 million in second-round funding that will be used to fund clinical trials of gene therapies for ocular diseases retinitis pigmentosa (RP) and dry age-related macular degeneration (AMD). The post SparingVision raises €75m for eye disease gene therapies appeared first on.
BioPharma Reporter
FEBRUARY 23, 2023
The messenger RNA (mRNA) specialist Moderna has teamed up with ElevateBio-owned Life Edit Therapeutics to develop gene editing therapies that are delivered into patients in vivo.

Pharmaceutical Technology
MARCH 28, 2023
Vertex Pharmaceuticals has signed a new non-exclusive licensing agreement with CRISPR Therapeutics to expedite the development of its hypoimmune cell therapies to treat type 1 diabetes (T1D). The gene-editing technology allows for precise, directed changes to genomic DNA.
Advarra
NOVEMBER 29, 2022
The field of cell and gene therapies (CGT) is constantly evolving, and there has been significant progress in this area of research. However, despite the promise of these therapies, the regulations governing them lag the science, which in turn hinders the clinical translation of these novel medicines.

The Pharma Data
JUNE 7, 2023
Bayer strengthens gene therapy portfolio with lipid nanoparticle technology from Acuitas Therapeutics Bayer AG is joining forces with Acuitas Therapeutics, Inc., a biotechnology company specializing in the development of lipid nanoparticle (LNP) delivery systems for molecular therapeutics.
Drug Discovery World
MAY 23, 2023
DDW’s Megan Thomas caught up with George White , General Manager, Product Management, Cell & Gene Therapy at Cytiva, to learn more about the company’s insight into the CGT market. Key challenges for cell and gene therapies In order for cell and gene (CGT) therapies to succeed, George White identifies several challenges to overcome.

Drug Discovery World
FEBRUARY 27, 2023
An investigational gene therapy for Sanfilippo syndrome – which leads to a form of childhood dementia – has shown promising early results in a proof-of-concept study. Patients with MPS-IIIA have a mutation in the SGSH gene, causing them to lack an enzyme which normally breaks down large sugar molecules.

pharmaphorum
AUGUST 24, 2021
Vertex Pharma has ramped up its involvement in gene-editing medicines for the third time in a matter of months, agreeing a partnership with CRISPR specialist Arbor Biotechnologies that could be worth up to $1.2 If approved, it could mount a challenge to bluebird bio’s gene therapy Zynteglo, which is already approved in Europe.
Pharmaceutical Technology
APRIL 28, 2023
The US Food and Drug Administration (FDA) has granted an Orphan Drug Designation to Editas Medicine’s gene therapy EDIT-301 in sickle cell disease, based on an April 27 announcement. The US agency previously granted the Orphan Drug Designation to EDIT-301 for its study in beta thalassemia, in May 2022.
Pharmaceutical Technology
OCTOBER 13, 2022
Vita Therapeutics has raised $31m in a Series B financing round for the development of cell therapies for neuromuscular diseases and cancers. VTA-100 has been designed to be an autologous therapy which combines gene correction as well as induced iPSC technology for repairing and replacing muscle cells for LGMD2A patients.

Pharmaceutical Technology
AUGUST 26, 2022
ElevateBio has signed a long-term strategic collaboration with the University of Pittsburgh, US, to establish a biomanufacturing centre for expediting cell and gene therapy development. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.

BioTech 365
AUGUST 13, 2020
ASC), a leading gene editing company focused on the development of enabling tools for cell and gene therapy has been awarded close to $2.0 MILPITAS, Calif.–(BUSINESS –(BUSINESS WIRE)–#AntibodyDiscovery–Applied StemCell, Inc.

The Pharma Data
JANUARY 12, 2021
Marianne De Backer, Head of Business Development & Licensing in Bayer’s Pharmaceuticals Division, pictured above. Also in December, the company partnered with Atara Biotherapeutics to develop off-the-shelf T-cell immunotherapies for high mesothelin-expressing tumors. Photo courtesy of Bayer. We have set the bar high.

Roots Analysis
FEBRUARY 27, 2024
How is the genome editing market landscape evolving: Currently, there is an evident increase in demand for complex biological therapies (including regenerative medicine products), which has created an urgent need for robust genome editing techniques.

Pharmaceutical Technology
APRIL 25, 2023
After missing the previous Q1 deadline, bluebird bio has submitted a Biologics License Application (BLA) to the US Food and Drug Administration (FDA) for its sickle cell disease gene therapy lovo-cel, based on an April 24 company announcement. Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.
Drug Discovery World
JANUARY 17, 2023
Researchers developed nanoparticles able to penetrate the neural retina and deliver mRNA to the photoreceptor cells whose proper function makes vision possible. . Limitations of AAV gene therapy . We’re hoping to use what we’ve learned so far about LNPs to develop an improved gene editor delivery system.” .

The Pharma Data
DECEMBER 5, 2020
5, 2020 — A pair of new gene therapies promise a potentially lasting cure for sickle cell disease by subtly altering the genetic information in patients’ bone marrow cells, researchers report. Both of the new gene therapy studies were published online Dec. SATURDAY, Dec.

Drug Discovery World
DECEMBER 21, 2023
DDW’s top 10 most popular articles for 2023 reflect the key subjects in drug discovery and development. In a record year for gene therapy approvals, it isn’t surprising that cell & gene therapy (CGT) has been a hot topic. Read the article – Expert view: What’s next for cell and gene therapy?

pharmaphorum
JUNE 23, 2022
Novartis has shouldered its way into the in vivo gene editing category via a deal with US biotech Precision BioSciences, focused on a therapy for sickle cell disease (SCD). billion in potential milestones if the project advances through development and onto the market. billion agreement that started in 2020.

BioSpace
NOVEMBER 17, 2022
Editas Medicine is pausing its ocular gene therapy program after demonstrating a favorable safety profile and seeking a potential partner to develop EDIT-101, the company announced Thursday.
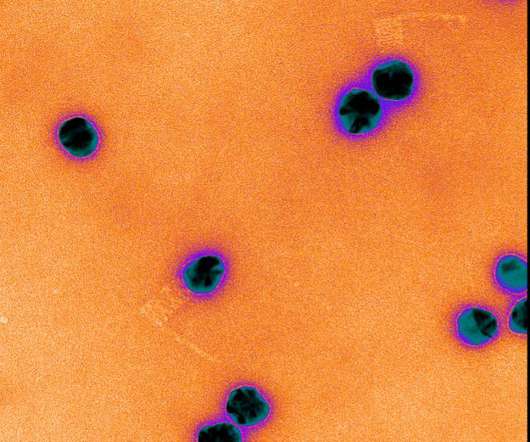
Scienmag
FEBRUARY 23, 2021
CRISPR gene editing has transformed research, but it is not perfect, and can sometimes target unintended genes; to watch CRISPR enzymes respond to different genes, Leipzig University researchers developed a new method using DNA origami and were able to me Credit: Image courtesy of Julene Madariaga Marcos.

XTalks
OCTOBER 20, 2023
Intellia said NTLA-2001 is the first investigational in vivo CRISPR-based gene editing therapy cleared to enter late-stage clinical development. As an in vivo therapy, it can edit genes inside the body rather than in cells extracted from patients.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content