Verve partners with Lilly on gene editing therapy for heart disease
Bio Pharma Dive
JUNE 15, 2023
Lilly will pay the genetic medicine specialist $60 million upfront to work together on a treatment targeting a risk factor known as Lp(a).
Bio Pharma Dive
JUNE 15, 2023
Lilly will pay the genetic medicine specialist $60 million upfront to work together on a treatment targeting a risk factor known as Lp(a).

Pharmaceutical Technology
JUNE 15, 2023
Eloxx has revealed its lead candidate ELX-02 improved predicted forced expiratory volume (ppFEV1) in patients with Class 1 cystic fibrosis (CF) in a new analysis of a Phase II trial that missed its efficacy endpoints. Following underwhelming topline results from the Phase II trial (NCT04135495) announced in late 2022, Eloxx recalculated the results using the change in ppFEV1 (a secondary outcome) from day 1 instead of from baseline.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Bio Pharma Dive
JUNE 15, 2023
The biosimilar developer’s plans to set steep discounts on its copycat version of AbbVie’s Humira set off a legal back-and-forth between the companies.

AuroBlog - Aurous Healthcare Clinical Trials blog
JUNE 15, 2023
The Tamil Nadu Pharma Traders Association (TN PTA), a registered body of 30,000 pharmaceutical wholesalers and retailers working in the 30 districts of the state, has approached the director-in-charge of the drugs control administration (DCA) with a request to the DTAB and the DCC under the union health ministry to allow three months time for […]

Bio Pharma Dive
JUNE 15, 2023
Lobbying group PhRMA does not expect any near-term changes to the IRA’s drug pricing provisions, but is attempting to lay groundwork for future changes.

Pharma Mirror
JUNE 15, 2023
In recent years, the field of clinical trials has witnessed a significant transformation due to advancements in technology. The integration of technology into clinical trials has revolutionized the way researchers conduct studies, collect data, and analyze results. This article explores the current trends in clinical trial technology and their impact on the research landscape.
Clinical Research Informer brings together the best content for clinical researchers from the widest variety of industry thought leaders.

Pharma Mirror
JUNE 15, 2023
Sarin Akter, a dynamic healthcare sales agent from Moulavibazar, Bangladesh, has been recently honored with iDE’s esteemed “40 Under 40” award. This recognition is a testament to her transformative influence in facilitating access to vital health products in rural Bangladesh. Navigating the complex terrain of rural healthcare sales, Sarin overcame challenges posed by entrenched misconceptions among locals, such as beliefs that modern medicine could lead to infertility in women.
Pharmaceutical Technology
JUNE 15, 2023
Alentis Therapeutics has received US Food and Drug Administration (FDA) clearance for its investigational new drug (IND) application for ALE.C04 to treat Claudin-1 positive (CLDN1+) tumours. ALE.C04 is an investigational antibody designed to target exposed CLDN1 in cancer cells. This approval has been granted for ALE.C04 both as a monotherapy and in combination with pembrolizumab in a first-in-human clinical study in recurrent or metastatic head and neck squamous cell carcinoma (HNSCC).

AuroBlog - Aurous Healthcare Clinical Trials blog
JUNE 15, 2023
Scientists have discovered not only that animals age more quickly when they don’t have enough of the amino acid taurine in the body, but that oral taurine supplements can delay aging and increase a healthy lifespan.
Pharmaceutical Technology
JUNE 15, 2023
Chugai Pharmaceutical has submitted a new drug application (NDA) to Japan’s Ministry of Health, Labour and Welfare (MHLW) for crovalimab to treat paroxysmal nocturnal haemoglobinuria (PNH), a rare blood disorder in which parts of the immune system attack and damage red blood cells and platelets. Crovalimab is an anti-C5 recycling antibody. C5 is an important component of the complement system [a system of plasma proteins] and the drug is expected to control its activity.

Advertisement
White paper that delves into the complex topic of Decentralized Clinical Trials and how to master them within the confines of FDA Regulations

Rethinking Clinical Trials
JUNE 15, 2023
This year’s Annual Steering Committee Meeting for the NIH Pragmatic Trials Collaboratory featured two special guests from the U.S. Food and Drug Administration’s Digital Health Center of Excellence—Matthew Diamond, MD, PhD, Chief Medical Officer, and Sonja Fulmer, PhD, Acting Deputy Director. They joined to share information on the FDA’s approach to clinical decision support (CDS) software, including the agency’s recently released guidance , which helped stimulate a discussion on the associated
Pharmaceutical Technology
JUNE 15, 2023
The US Food and Drug Administration (FDA) has approved Amneal Pharmaceuticals’ 505(b)(2) new drug application (NDA) for a ready-to-use oncology injectable, PEMRYDI RTU. PEMRYDI RTU is indicated as an initial treatment, along with pembrolizumab and platinum chemotherapy, for metastatic non-squamous non-small cell lung cancer patients with no epidermal growth factor receptor or anaplastic lymphoma kinase genomic tumour aberration.

BioPharma Reporter
JUNE 15, 2023
Eli Lillyâs blockbuster type 2 diabetes (T2B) drug Mounjaro is likely to become the dominant therapy in obesity and T2D, due to significant weight loss induced by it, says GlobalData.
Pharmaceutical Technology
JUNE 15, 2023
The US Food and Drug Administration (FDA) has accepted Ipsen’s supplemental new drug application (sNDA) for the Onivyde regimen as a first-line treatment for metastatic pancreatic ductal adenocarcinoma (mPDAC) patients. The Onivyde regimen comprises Onivyde (irinotecan liposome injection) along with 5-fluorouracil/leucovorin and oxaliplatin (NALIRIFOX regimen).

Advertisement
Planning on running clinical trials in Japan? How can you reliably supply these studies? Discover Catalent’s clinical supply packaging facility in Shiga, Japan. Strategically located between Tokyo and Osaka, and one of largest in Japan, this 6,000 square meter facility offers comprehensive services including primary and secondary clinical packaging and labelling, comparator sourcing, cold chain storage, local and global distribution, local language support and white glove service to support stud

Outsourcing Pharma
JUNE 15, 2023
Coordinating with Migraine and Headache Awareness Month (MHAM), this June, Pfizer has announced it is partnering with multi-award winning, singer and actress, Lady Gaga, who was diagnosed with migraine when she was 14.

Pharmaceutical Technology
JUNE 15, 2023
Developing a new drug is a monumental achievement — the reward of years of research and innovation. Successful innovation requires enormous effort from countless project teams that build on the medical, scientific and technological research and development of generations before them. AI in pharma R&D is one of the latest innovations helping teams apply existing knowledge to new problems.

Outsourcing Pharma
JUNE 15, 2023
Multinational pharmaceutical company, Gedeon Richter UK Ltd, announced this week that it has acquired full marketing and distribution rights to a contraceptive range.

Pharmaceutical Technology
JUNE 15, 2023
The need for new medical treatments and drugs has never been greater. But before pharmaceutical companies can go to market with a breakthrough drug, they need to ensure safety and efficacy through clinical trials. While this process is essential, it’s also slow, expensive and unpredictable. Pharma R&D teams are solving this problem by leveraging the power of artificial intelligence (AI) in clinical trials to save time and money.

Advertisement
This new white paper defines and details the impact of Decentralized Clinical Trials on the Pharmaceutical industry and how the impact can be measured along with steps companies can take to ensure adoption.

Pharma Times
JUNE 15, 2023
Researchers show silver retains antimicrobial activity for longer when it is infused into bioactive glass - News - PharmaTimes

Pharmaceutical Technology
JUNE 15, 2023
Two of the biggest challenges facing healthcare R&D are the sheer volume of data and the unique characteristics of every person’s health. Artificial intelligence (AI) and other technologies offer a way forward. AI for drug development is rapidly streamlining R&D processes, speeding up time to market and providing a better experience for patients and providers.

Fierce Pharma
JUNE 15, 2023
While demand for COVID-19 vaccines has plummeted throughout 2023, healthcare officials remain focused on ensuring that the most effective shots are available for those who need them. | While demand for COVID-19 vaccines has plummeted throughout 2023, healthcare officials remain focused on ensuring that the most effective shots are available for those who need them.
BioSpace
JUNE 15, 2023
The data for Reproxalap, Aldeyra Therapeutics’ drug for treating allergic conjunctivitis and dry-eye disease, could potentially pave the way for an alternative to over-the-counter eye drops.

Advertisement
When selecting a clinical supply provider, consideration often focuses upon the manufacturing, packaging, storage and distribution capabilities available that will, at face-value, be sufficient to meet the needs of the sponsor and their trial. However, there are human-based and knowledge-driven factors that are often overlooked that go beyond these basic physical capabilities and are integral to the development and delivery of high performing clinical supply chains.

Fierce Pharma
JUNE 15, 2023
As biosimilars gradually gain steam in the U.S. market, one of the biggest manufacturers of the copycat biologics has unveiled a plan to boost uptake across the globe. | Sandoz' Act4Biosimilars action plan includes a detailed report of challenges preventing wider biosimilar access in each region, starting with the Americas. The initiative's steering group will work with stakeholders across the region to implement steps to boost uptake.

BioSpace
JUNE 15, 2023
In the latest HEALEY trial readout, Clene’s CNM-Au8 significantly reduced plasma neurofilament light chain levels in ALS patients, which could be a strong and reliable signal of its clinical benefit.

Pharma Times
JUNE 15, 2023
EO2463 is an experimental treatment for indolent non-Hodgkin B cell lymphoma - News - PharmaTimes
BioSpace
JUNE 15, 2023
With Roche’s FDA approval on Thursday, both companies' bispecific antibodies will hit the market this summer for relapsed or refractory diffuse large B-cell lymphoma.

Advertisement
Clinical trial data management is increasingly challenging as studies grow in complexity. Quickly accessing and analyzing study data is vital for assessing trial progress and patient safety. In this paper, we explore real-time data access and analysis for proactive study management. We investigate using adverse event (AE) data to monitor safety and discuss a clinical analytics platform that supports collaboration and data review workflows.

Fierce Pharma
JUNE 15, 2023
An announcement early this month by Mark Cuban’s deep discount drug company that it would sell Coherus Biosciences’ biosimilar version of AbbVie’s Humira has the Illinois drugmaker calling foul.
BioSpace
JUNE 15, 2023
In a unanimous 21-0 vote, an FDA advisory committee recommended an updated monovalent formulation of the COVID-19 vaccine targeting the now-dominant XBB.1.5 subvariant.

Fierce Pharma
JUNE 15, 2023
Astellas' new CEO Naoki Okamura talked to Fierce Pharma about the company's business plans. | Astellas' new CEO Naoki Okamura discussed the company's business plans. Eisai and Biogen's Leqembi won backing from the FDA's external advisers. AbbVie has brought a BTK patent infringement lawsuit against BeiGene. And more.
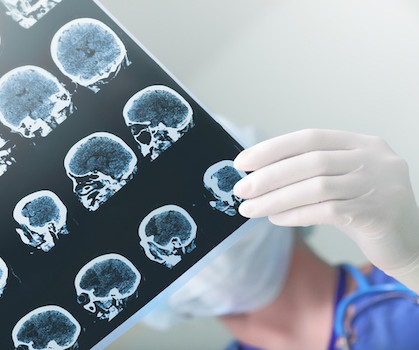
BioSpace
JUNE 15, 2023
After an FDA advisory committee unanimously recommended Leqembi’s full approval, questions linger around amyloid-related imaging abnormalities and a potentially cumbersome patient registry.

Advertisement
As the development pipeline for new drugs continues to grow, biopharmaceutical companies are re-evaluating how to best manage and balance resources across an increasing number of development projects and complex clinical trials. There are two approaches that can be used to speed a drug from development to clinic faster: timeline compression and parallel processing, but only one that considers the benefits of integrating clinical supply into the overall drug development process.
Let's personalize your content