Pfizer’s Ngenla finally secures FDA approval following a rocky road to market
Pharmaceutical Technology
JUNE 28, 2023
Pfizer has announced that the FDA has approved Ngenla (somatrogon-ghla), a paediatric growth hormone deficiency treatment.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Pharmaceutical Technology
JUNE 28, 2023
Pfizer has announced that the FDA has approved Ngenla (somatrogon-ghla), a paediatric growth hormone deficiency treatment.

Pharmaceutical Technology
AUGUST 8, 2022
According to the trial data, Enhertu lowered disease progression or mortality risk by 50% compared with chemotherapy according to the physician’s choice in HER2-low metastatic breast cancer patients with hormone receptor (HR)-positive disease or HR-negative disease. months compared with 5.1 months for the chemotherapy arm.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

XTalks
JULY 6, 2023
Pfizer has been going strong on the approval front, having secured its fifth US Food and Drug Administration (FDA) approval in the past five weeks, with the most recent being for the company’s once-weekly human growth hormone analog Ngenla (somatrogon-ghla).

The Pharma Data
JUNE 28, 2023
FDA Approves Pfizer’s NGENLA™, a Long-Acting Once-Weekly Treatment for Pediatric Growth Hormone Deficiency NEW YORK & MIAMI–(BUSINESS WIRE)– Pfizer Inc. The approval of NGENLA will be significant for children with growth hormone deficiency in the U.S. NYSE: PFE) and OPKO Health Inc.
pharmaphorum
MAY 16, 2022
GLP-1 and GIP are hormones involved in blood sugar control and Mounjaro, a first-in-class medicine that activates both GLP-1 and GIP receptors, demonstrated improved blood sugar control. The post Lilly’s new drug Mounjaro (tirzepatide ) wins US FDA approval appeared first on.

Drug Discovery World
JUNE 1, 2023
A Nature publication confirmed that there are currently 12 FDA-approved ADCs on the market, and nine of these secured FDA approval in the past six years 2. DDW’s Megan Thomas explores three trends in the market.

pharmaphorum
OCTOBER 4, 2022
AstraZeneca and Merck & Co’s Lynparza has been the undisputed market leader in the PARP inhibitor category for some years, but would-be competitors continue to chip away at its market share. The post PARP rivals close in on AZ and Merck’s market leading Lynparza appeared first on.

pharmaphorum
APRIL 1, 2022
market is just beginning to heat up. And already in the first quarter of 2022, the FDA approved a third filgrastim biosimilar, Releuko. With 34 approved biosimilars and dozens more in the pipeline, what does this year have in store? Innovator products working to maintain market share.

The Pharma Data
NOVEMBER 21, 2021
The FDA approved Voxzogo under the accelerated approval pathway, which allows for earlier approval of drugs that treat serious conditions and fill an unmet medical need, based on a surrogate or intermediate clinical endpoint. 3] Achondroplasia is caused by mutations in the FGFR3 gene. Inheritance is autosomal dominant.[1][2]

pharmaphorum
SEPTEMBER 15, 2021
If approved, linzagolix will be the only drug in the class with a dosing regimen intended for women with uterine fibroids who cannot or do not want to take hormone therapy, as well as options for those women happy to do so, according to ObsEva.

Pharmaceutical Technology
OCTOBER 13, 2022
The drug won its original FDA approval in May last year to treat heavy menstrual bleeding that accompanies uterine fibroids in premenopausal women. Myfembree is a combination therapy that includes relugolix, estradiol and norethindrone acetate.

pharmaphorum
FEBRUARY 1, 2021
Novartis’ multiple sclerosis drug has been given the green light by the European Medicines Agency’s CHMP scientific committee, paving the way for a likely approval in the coming weeks.

XTalks
FEBRUARY 8, 2024
Understanding the market dynamics of diabetes treatments becomes crucial for professionals across these industries. Ozempic (Semaglutide) Ozempic sales in 2022: $8.713 billion Company/Developer: Novo Nordisk Date of first FDA approval: December 5, 2017 Indications Ozempic is FDA-approved for: Type 2 diabetes Price of Ozempic: $1,029 for 1.5

Drug Discovery World
APRIL 4, 2023
Orserdu (elacestrant), Stemline Therapeutics The first and only treatment specifically indicated for patients with ESR1 mutations in ER+, HER2- advanced or metastatic breast cancer was approved in the US in February. This made Jesduvroq is the first oral hypoxia-inducible factor prolyl hydroxylase inhibitor (HIF-PHI) to be approved in the US.

pharmaphorum
AUGUST 8, 2022
Bayer has won a key FDA approval to extend the use of its prostate cancer therapy Nubeqa, as it tries to claim market share from rival drugs and fulfil its aim of building the drug into a €3 billion ($3.1 billion) blockbuster. Most men with mHSPC will eventually progress to CRPC, which has poor survival prospects.

XTalks
NOVEMBER 3, 2022
Demand for Lilly’s GIP/GLP-1 receptor agonist Mounjaro is also rising because of high patient demand since the drug’s May 13 FDA approval and expanding insurance coverage. In its first quarter on the market since its approval, sales totalled $97 million between July and September in the US.
XTalks
AUGUST 15, 2023
The Janssen Pharmaceutical Companies of Johnson & Johnson announced yesterday that the US Food and Drug Administration (FDA) has granted marketing authorization for its combination treatment Akeega for metastatic castration-resistant prostate cancer (mCRPC) with BRCA1/2 mutations.

The Pharma Data
OCTOBER 14, 2020
WAKIX is the first and only treatment approved by the FDA for people with excessive daytime sleepiness or cataplexy associated with narcolepsy that is not scheduled as a controlled substance by the U.S. WAKIX received FDA approval for the treatment of excessive daytime sleepiness in adult patients with narcolepsy in August 2019.

pharmaphorum
AUGUST 28, 2020
Shares in dermatology specialist Cassiopea, which is listed on the SWX exchange in Switzerland, rose almost 17% after news of the FDA approval emerged. The biotech isn’t revealing how much it intends to charge for the new medicine, saying it will do so closer to launch later this year.

Pharmaceutical Technology
JUNE 1, 2023
Although this vote has narrowed OCA’s chances to get FDA approval by the target PDUFA date of June 2023 it has left the door open for OCA to potentially present more robust data in the near future.

XTalks
FEBRUARY 21, 2024
Recently, Eli Lilly revealed promising results from a mid-stage trial, indicating that its popular drug, tirzepatide (marketed as Zepbound and Mounjaro for weight loss and diabetes, respectively), may be an effective treatment for the fatty liver disease metabolic dysfunction-associated steatohepatitis (MASH).
XTalks
NOVEMBER 9, 2023
Eli Lilly is now fully armed on the GLP-1 front as it’s finally received US Food and Drug Administration (FDA) approval for Zepbound (tirzepatide), its obesity version of Mounjaro. The FDA approved Zepbound for chronic weight management in adults with obesity.

pharmaphorum
OCTOBER 11, 2020
When added to standard treatment with hormone-based drugs, Ibrance (palbociclib) was unable to achieve better iDFS than hormone therapy alone in women with hormone receptor-positive (HR+), human epidermal growth factor-negative (HER2-) early-stage breast cancer.

Delveinsight
AUGUST 31, 2021
FDA’s Green Flag to Ascendis’s Once-Weekly SKYTROFA for Treatment of Pediatric Growth Hormone Deficiency. Growth Hormone Deficiency , commonly known as dwarfism, can constitute short stature and complications with metabolism, which arise due to inadequate production of growth hormones by the pituitary gland inside the body.
XTalks
FEBRUARY 5, 2024
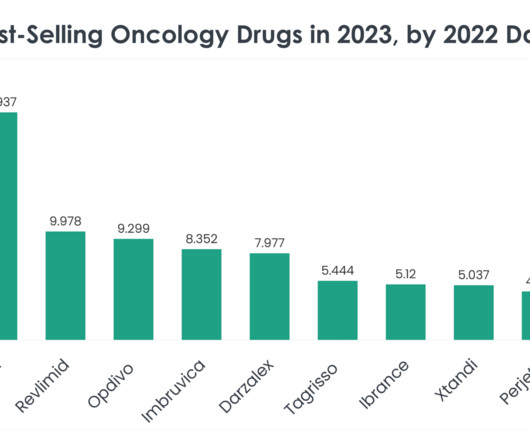
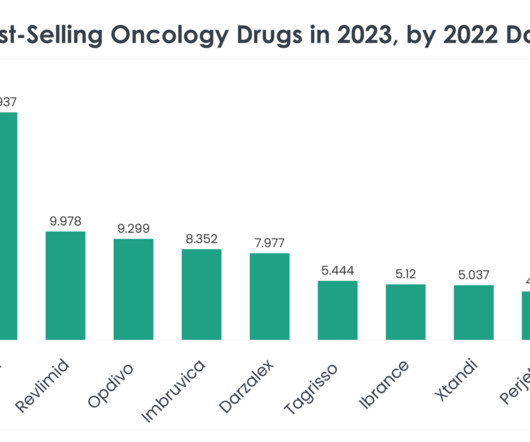
As we delve into the specifics of these best-selling oncology drugs, we uncover the underlying factors contributing to their market dominance, from clinical efficacy and safety profiles to strategic market positioning and patient accessibility. Price of Revlimid: A supply of 28 oral 2.5 mg capsules is $24,576.
XTalks
FEBRUARY 3, 2023
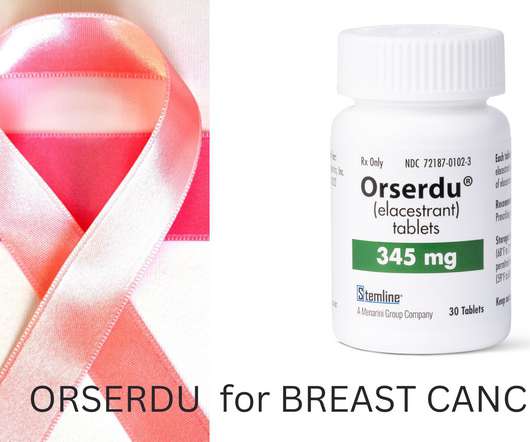
Until now, fulvestrant was the only other selective estrogen receptor degrader (SERD) therapy approved for ER+ breast cancer, delivered via intramuscular injections. Current treatments include hormonal therapies that focus on blocking the function of the estrogen receptor, curbing its production or degrading the receptor itself.

XTalks
MARCH 8, 2024
The biosimilar injections, Jubbonti (denosumab-bddz) and Wyost (denosumab-bddz), are approved as interchangeable biosimilars to Prolia and Xgeva, respectively. Despite the approvals, the market launches of Jubbonti and Wyost are uncertain due to ongoing patent litigation between Sandoz and Amgen.

Delveinsight
AUGUST 2, 2020
The dynamics of the cancer cachexia market is expected to gain momentum as several companies are testing the waters, advancing cancer cachexia pipeline. Cachexia is observed as a result of underlying diseases, including cancer, AIDS, tuberculosis, chronic heart failure, hormonal deficiency, and others.
XTalks
NOVEMBER 10, 2021
Related: GSK’s Dostarlimab Wins FDA Approval for dMMR Endometrial Cancer. Darbepoetin alfa is a synthetic version of the hormone erythropoietin, which is made in the kidneys and stimulates the production of red blood cells (RBC), or erythrocytes. Market Size and Regulatory Pathway. What is Daprodustat?
XTalks
OCTOBER 11, 2023
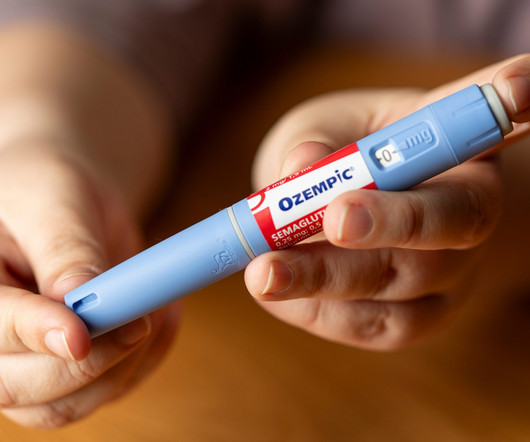
Rybelsus is Novo’s third semaglutide product on the market, a tablet form of the drug used for the treatment of type 2 diabetes. They mimic the action of GLP-1, a hormone that helps regulate blood sugar levels by enhancing insulin secretion.
XTalks
FEBRUARY 5, 2024
As we delve into the specifics of these best-selling oncology drugs, we uncover the underlying factors contributing to their market dominance, from clinical efficacy and safety profiles to strategic market positioning and patient accessibility. Price of Revlimid: A supply of 28 oral 2.5 mg capsules is $24,576.

pharmaphorum
DECEMBER 23, 2020
2020 also saw some of the first “tumour agnostic” cancer drugs get to market, with Bayer’s Vitravki (larotrectinib) getting funding in the UK for tumours with confirmed neurotrophic tyrosine receptor kinase (NTRK) gene fusions.”.

pharmaphorum
JANUARY 26, 2023
In Europe, approximately 531,000 breast cancer patients are diagnosed annually, with around 141,000 deaths, and around half of all cases would fall under the HER2-low classification, split between patients with both hormone receptor (HR) positive and HR negative disease. months to 23.4

The Pharma Data
DECEMBER 16, 2020
Food and Drug Administration (FDA) approval and the launch of Aminocaproic Acid Tablets USP, 500mg. market for this product is approximately $12.7 “This is ANI’s seventh generic product launch in 2020 and reaffirms our commitment to increasing the pace of market introductions for our products.
STAT News
DECEMBER 5, 2022
Food and Drug Administration is taking a harder line on its program that fast-tracks drug approvals based on preliminary evidence, spurring GSK, Roche, and others to remake plans for their drugs or pull them from the market , The Wall Street Journal notes. Supreme Court decision overturning Roe v. Food and Drug Administration.

Pharmaceutical Technology
SEPTEMBER 13, 2022
Hormone receptor-positive/human epidermal growth factor negative (HR+/HER2-) breast cancer accounts for approximately 70% of all breast cancers, with close to 40,000 new cases diagnosed each year worldwide. The post ESMO 2022: shrinking market for Trodelvy, with fierce ADC competition appeared first on Pharmaceutical Technology.

Delveinsight
DECEMBER 7, 2020
Banyan Biomarkers is one company to be able to market a blood-based diagnostic test in the US market, as it utilizes tech to aid the detection of traumatic brain injuries and concussions. In February 2018, the San Diego-based company was granted a de novo request from FDA for the Banyan Brain Trauma Indicator.

XTalks
FEBRUARY 19, 2021
Evofem Biosciences announced the launch of their new national campaign called, “Get Phexxi,” designed to raise awareness about the company’s non-hormonal birth control method. In May 2020, the US Food and Drug Administration (FDA) approved Phexxi , a non-hormonal contraceptive for women.

XTalks
NOVEMBER 9, 2022
Glucagon-like peptide-1 (GLP-1) receptor agonists are a newer class of diabetes drugs that have the potential to double as weight loss drugs, widening their lucrative market potentials. Novo’s obesity version of semaglutide (at a higher dose) is marketed as Wegovy and was approved in 2021. billion in 2021 to $61.6

XTalks
MARCH 15, 2023
FDA-approved ADHD-specific non-stimulants are a relatively newer class of medication designed to treat ADHD that have much fewer side effects than stimulants. Norepinephrine is a hormone and neurotransmitter that helps in alertness and maintaining control in high-stress situations. In 2017, Strattera’s total revenue was $618.2

XTalks
MAY 17, 2023
Astellas Pharma recently announced the US Food and Drug Administration (FDA) approval of their new medication, Veozah (fezolinetant) for the treatment of moderate to severe hot flashes and night sweats due to menopause. Hormone replacement therapy is thought to be one of the most effective treatments for vasomotor symptoms.

XTalks
SEPTEMBER 17, 2020
Symptoms and Etiology: Caused by an overproduction of growth hormone, acromegaly primarily affects adults. By reducing the amount of growth hormone in the blood, progressive enlargement of bones should be slowed. By reducing the amount of growth hormone in the blood, progressive enlargement of bones should be slowed.

XTalks
APRIL 29, 2022
Tirzepatide is a novel investigational obesity treatment that contains mimetics of two hormones that are involved in regulating appetite in a single peptide: a GIP (glucose-dependent insulinotropic polypeptide) receptor agonist and a GLP-1 (glucagon-like peptide-1) receptor agonist. percent and 22.5 percent, respectively, compared to placebo.
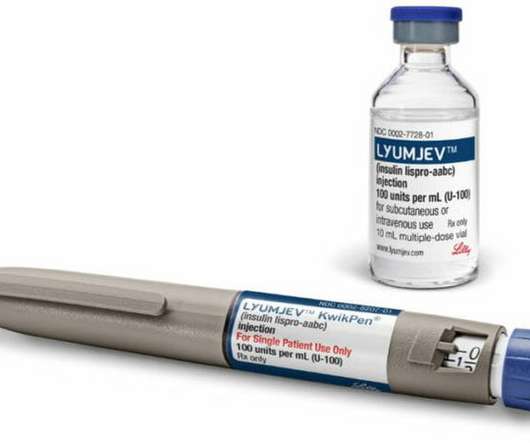
The Pharma Data
AUGUST 17, 2021
and in several global markets, including Japan and the European Union, where it is also approved for use in insulin pumps. Diabetes is a chronic disease that occurs when the body does not properly produce or use the hormone insulin. Lyumjev is available in the U.S.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content