Brukinsa (zanubrutinib) Gets FDA Approval for Chronic Lymphocytic Leukemia or Small Lymphocytic Lymphoma
XTalks
JANUARY 26, 2023
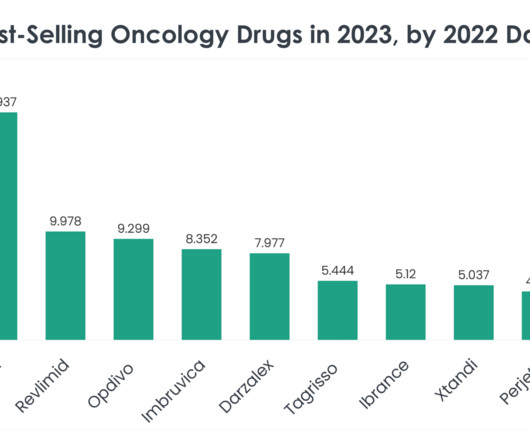
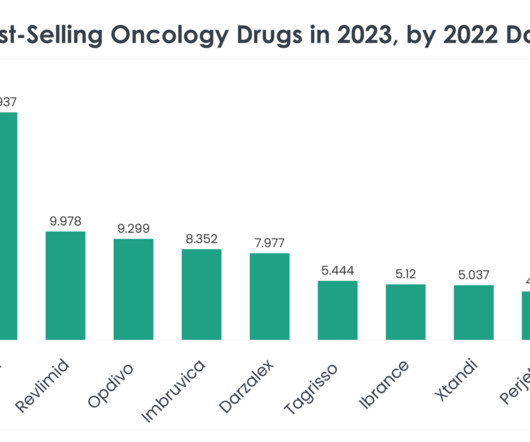
This is the fourth US FDA approval that BeiGene has received for Brukinsa since 2019. Diagnosis is confirmed by a differential complete blood count and genetic testing. Treatment selection is determined by age, disease severity, comorbidities and the presence or absence of certain genetic mutations.




























Let's personalize your content