Regeneron expands in gene editing with Mammoth deal
Bio Pharma Dive
APRIL 25, 2024
“With each passing year, we're more committed to becoming a serious player in the genetic medicine space,” a Regeneron executive said.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Bio Pharma Dive
APRIL 25, 2024
“With each passing year, we're more committed to becoming a serious player in the genetic medicine space,” a Regeneron executive said.
Worldwide Clinical Trials
NOVEMBER 27, 2023
Casgevy, the commercial product formerly known as exa-cel, is administered by taking stem cells out of a patient’s bone marrow and editing a gene in the cells in a laboratory, with the modified cells then infused back into the patient after conditioning treatment to prepare the bone marrow. In June 2023, the U.S.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
BioSpace
MARCH 24, 2024
Ubiquitous potential, possible safety advantages and the recent growth of cell and gene therapy are driving investment in a different type of genetic editing.

Pharmaceutical Technology
FEBRUARY 23, 2023
Moderna has entered a strategic research and development partnership with ElevateBio’s Life Edit Therapeutics to discover and develop new in-vivo mRNA gene editing therapies. Our novel editing systems have the potential to precisely modify gene targets for both in vivo and ex vivo therapeutic development.”

Bio Pharma Dive
OCTOBER 3, 2023
The longtime partners believe that, by combining their technologies, they can create “in vivo” genetic medicines for nervous system and muscular disorders.

pharmaphorum
FEBRUARY 15, 2024
Gene-editing biotech Prime Medicine has priced its IPO, hoping to raise $140m to advance its pipeline of one-shot therapies for severe genetic disorders.

Drug Discovery World
FEBRUARY 22, 2024
This eBook explores the latest innovations in gene editing and CRISPR, the possibilities of new editing techniques and where the greatest potential for the technology might lie in the future. The post Gene editing & CRISPR: Advances in applications and opportunity appeared first on Drug Discovery World (DDW).
Bio Pharma Dive
JUNE 15, 2023
Lilly will pay the genetic medicine specialist $60 million upfront to work together on a treatment targeting a risk factor known as Lp(a).

Bio Pharma Dive
FEBRUARY 28, 2024
The acquisition of Proof Diagnostics, which was formed to develop a COVID-19 test, gives Ginkgo a library of novel enzymes that can be used to make genetic medicines.

pharmaphorum
JANUARY 29, 2021
Cutting edge’ is, for once, a truly apt description when it comes to gene editing – both because the field is pushing medicine into areas we might never have dreamed possible, and because these technologies involve literally cutting DNA at a specific point in the genome. Zinc fingers.

STAT News
FEBRUARY 22, 2023
Moderna is aiming to build a gene editing franchise powered by some of the same technologies used in its COVID-19 vaccines. The Cambridge biotech company announced Wednesday that it will partner with Life Edit Therapeutics to develop potentially permanent treatments for rare genetic diseases and other conditions.

Drug Discovery World
DECEMBER 6, 2023
Gene editing company Eligo Bioscience has announced a successful $30 million Series B funding round, led by Sanofi Ventures. “We We feel this reflects the strong support for our vision and confirms the potential of Eligo to create a novel class of transformative genetic medicines.”

Drug Discovery World
FEBRUARY 22, 2024
Following the first regulatory approval of a CRISPR-based drug in late 2023, over ten years since the CRISPR-Cas9 system was elucidated, there is considerable optimism about the future potential for gene editing technologies. appeared first on Drug Discovery World (DDW).

Bio Pharma Dive
NOVEMBER 20, 2020
Through a deal with Precision Biosciences, the pharma aims to develop therapies for genetic disorders, including Duchenne muscular dystrophy.
XTalks
DECEMBER 14, 2022
Expanding upon the CRISPR-Cas9 gene editing system, researchers at MIT have designed a new technique called PASTE gene editing that can cut out defective genes and replace them with new genes in a safer and more efficient way. The PASTE gene editing technique was recently published in Nature Biotechnology.
BioSpace
MARCH 21, 2024
Tegoprubart, Eledon Pharmaceuticals’ experimental anti-CD40L antibody, was part of the immunosuppressive treatment regimen used following the first-ever transplant of a kidney from a genetically modified pig to a human.

Drug Discovery World
NOVEMBER 30, 2023
Scientists have used CRISPR gene editing to target and rapidly destroy glioblastoma cells in an approach that could apply to other highly mutated cancers. The Gladstone team realised that these mutated cells have a unique genetic signature that could be targeted. “We

Bio Pharma Dive
JUNE 15, 2021
A deal with biotech startup Capsida, which recently raised $140 million, gives CRISPR access to a Caltech technology that aims to improve the delivery of genetic medicines.

Pharmaceutical Technology
MAY 25, 2023
ElevateBio has raised $401m in a Series D financing round for advancing its technology platforms to expedite the design, production and development of cell and gene therapies. The technology platforms include the Life Edit gene editing platform, an RNA, cell, protein, vector engineering and induced pluripotent stem cells (iPSCs) platform.
BioPharma Reporter
FEBRUARY 23, 2023
The messenger RNA (mRNA) specialist Moderna has teamed up with ElevateBio-owned Life Edit Therapeutics to develop gene editing therapies that are delivered into patients in vivo.

Camargo
JULY 27, 2021
How and When to Incorporate PK Design into Your Gene Therapy Development Plan. Gene therapy, which was in its infancy around 30 years ago, is now becoming a more prominent treatment method in many therapeutic areas, from personalized therapy to mass vaccinations against COVID-19. Gene Therapy Definition.

pharmaphorum
SEPTEMBER 28, 2022
Vertex Pharma and partner CRISPR Therapeutics will start a rolling marketing application in the US for their gene-editing drug for sickle cell disease (SCD) and beta thalassaemia later this year. The time places exa-cel in pole position to become the first drug developed based on CRISPR/Cas9 gene-editing technology to reach the market.

XTalks
MARCH 7, 2024
In a pivotal move for the food industry, the US Food and Drug Administration (FDA) has unveiled industry guidance for genome edited foods derived from plants. Genome editing stands as a cutting-edge technique that ingeniously introduces new traits into plants.

pharmaphorum
OCTOBER 4, 2022
AstraZeneca’s rare disease firm Alexion is set to expand its genomic medicine portfolio with the acquisition of gene editing specialist LogicBio Therapeutics, in a deal worth approximately $68 million. The post AstraZeneca pays record 660% premium for gene editing company LogicBio appeared first on.

Scienmag
MAY 2, 2022
WINSTON-SALEM, NC – May 2, 2022 — Wake Forest Institute for Regenerative Medicine (WFIRM) scientists working on CRISPR/Cas9-mediated gene editing technology have developed a method to increase efficiency of editing while minimizing DNA deletion sizes, a key step toward developing gene editing therapies to treat genetic diseases.

Worldwide Clinical Trials
OCTOBER 24, 2023
Global Genes is a rare disease umbrella organization dedicated to eliminating the burdens and challenges of rare diseases for patients and families globally. Roughly 80% of rare diseases are due to a known genetic driver. Over the years, I have attended several Global Genes Summits.

pharmaphorum
OCTOBER 13, 2022
Gene therapies and research into them have grown immensely in recent years, offering more novel tools in regenerative medicine to fight disease, including rare diseases and genetic disorders. Beta-thalassemia is a rare blood disorder caused by a genetic defect in hemoglobin. Novartis also recently inked an up-to $1.5

pharmaphorum
AUGUST 24, 2021
Vertex Pharma has ramped up its involvement in gene-editing medicines for the third time in a matter of months, agreeing a partnership with CRISPR specialist Arbor Biotechnologies that could be worth up to $1.2 If approved, it could mount a challenge to bluebird bio’s gene therapy Zynteglo, which is already approved in Europe.
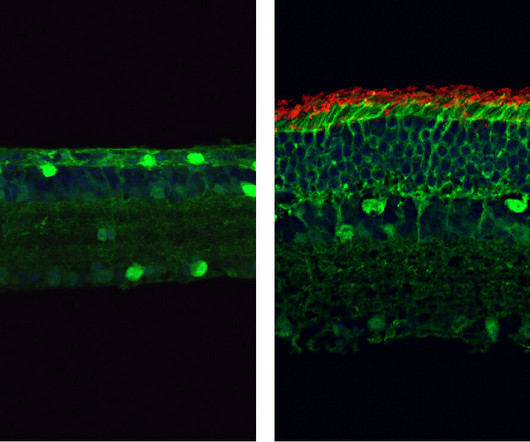
Medical Xpress
MARCH 17, 2023
The study, to be published March 17 in the Journal of Experimental Medicine, uses a new, highly versatile form of CRISPR-based genome editing with the potential to correct a wide variety of disease-causing genetic mutations.

pharmaphorum
FEBRUARY 15, 2024
Gene-editing biotech Prime Medicine has priced an initial public offering, hoping to raise $140 million to advance its pipeline of one-shot therapies for severe genetic disorders. The Cambridge, Massachusetts-based company is offering 19.2 million shares at $6.25

BioTech 365
MARCH 30, 2021
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Prime editing enables precise gene editing without collateral damage.The latest gene editing technology, prime editing, expands the ‘genetic toolbox’ for more precisely creating disease models and correcting … (..)

Scienmag
JANUARY 12, 2021
Martins Scientists have used gene-editing advances to achieve a tenfold increase in the production of super-bug targeting formicamycin antibiotics. Credit: Dr Dino J. The John Innes Centre researchers used the technology to create a new strain of Streptomyces formicae bacteria which over-produces the medically promising molecules.

STAT News
MARCH 1, 2023
Tessera Therapeutics chief scientist Michael Holmes had just finished presenting a much-anticipated peek at a technology the company had previously said could “ revolutionize genetic medicine ” and “cure nearly any genetic disease.”
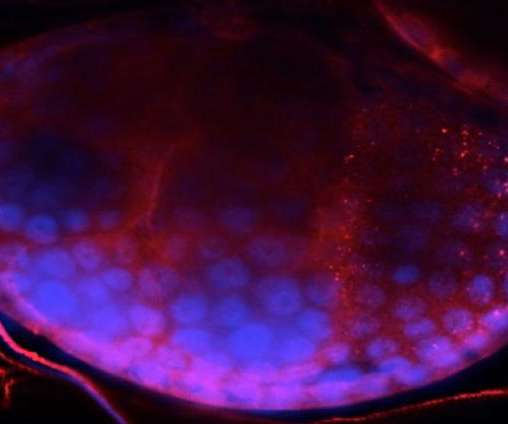
Scienmag
AUGUST 18, 2020
Credit: Rasgon Lab, Penn State Researchers who developed an improved method of gene editing for the study of arthropods will expand the technology for use in vertebrate species such as mice, fish and birds after receiving new funding from the National Science Foundation.

pharmaphorum
OCTOBER 8, 2020
Scribe Therapeutics, a start-up focusing on gene-editing using CRISPR/Cas9, has burst onto the biotech scene with a $415 million deal with Biogen. Further back in development are drugs that will be administered to edit genes within the body, but the first of these candidates are now in clinical trials. Jennifer Doudna.

Drug Discovery World
NOVEMBER 16, 2023
The Medicines and Healthcare products Regulatory Agency (MHRA) has authorised the world’s first gene therapy for sickle-cell disease (SCD) and transfusion-dependent β-thalassemia (TDT). Casgevy is designed to work by editing the faulty gene in a patient’s bone marrow stem cells so that the body produces functioning haemoglobin.

BioTech 365
DECEMBER 4, 2020
Biotechnology, Pharma and Biopharma News – Research – Science – Lifescience ://Biotech-Biopharma-Pharma: Advancing gene editing with new CRISPR/Cas9 variant.Using a new variant to repair DNA will improve both safety and effectiveness of the much-touted CRISPR-Cas9 tool in genetic research, … Continue reading →

Pharmaceutical Technology
MARCH 28, 2023
Under the terms of the deal, the company will receive non-exclusive rights to CRISPR/Cas9, a gene-editing technology of CRISPR Therapeutics, for the development of potentially curative T1D cell therapies. The gene-editing technology allows for precise, directed changes to genomic DNA.

Scienmag
NOVEMBER 19, 2020
Media contacts: Jennifer Kuzma, jkuzma@ncsu.edu Khara Grieger, kdgriege@ncsu.edu Mick Kulikowski, News Services, 919.218.5937 or mick_kulikowski@ncsu.edu Researchers at North Carolina State University call for a coalition of biotech industry, government and non-government organizations, trade organizations, and academic experts to work together to (..)
XTalks
JANUARY 4, 2024
Pfizer has kickstarted the new year with its first-ever gene therapy approval, awarded by Health Canada to the company’s Beqvez (fidanacogene elaparvovec) for the treatment of hemophilia B. There is a significant focus on developing gene therapies as longer-term solutions for the disease.
STAT News
MARCH 9, 2023
LONDON — Scientists at this year’s genome editing summit spent Tuesday showing the world just how far CRISPR -based medicines for treating human diseases have come in a decade.

BioTech 365
MAY 1, 2021
Editas Medicine to Present Preclinical Data Demonstrating Advancements in In Vivo Gene Editing Approach for the Treatment of Genetic Ocular Diseases at the Association for Research in Vision and Ophthalmology Annual Meeting Editas Medicine to Present Preclinical Data Demonstrating Advancements … Continue reading →
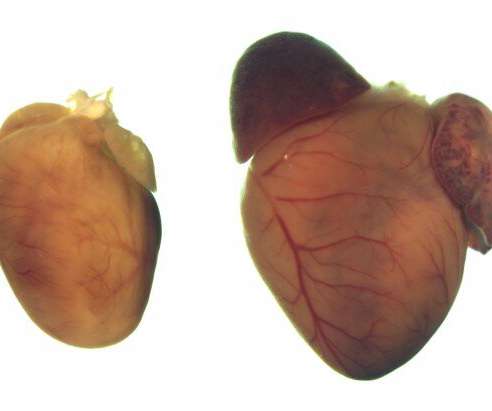
Medical Xpress
DECEMBER 21, 2022
Using the CRISPR-Cas9 gene editing system, UT Southwestern researchers corrected mutations responsible for a common inherited heart condition called dilated cardiomyopathy (DCM) in human cells and a mouse model of the disease.

Scienmag
MAY 26, 2022
A line of powerful tools has been developed from the popular CRISPR-Cas9 to cure genetic diseases. ITHACA, N.Y. — CRISPR has ushered in the era of genomic medicine. However, there is a last-mile problem – these tools need to be effectively delivered into every cell of the patient, and most Cas9s are too big to be fitted […].
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content